How Insulin Signaling Works: The Two-Fork Pathway That Controls Both Metabolism and Cell Growth
One receptor, two major cascades, and a critical branch point that shapes health and disease
You have probably heard of insulin as the hormone that controls your blood sugar, but its job is more complex and powerful than most people realize.
Insulin acts like a biological switchboard, steering not just how your body uses fuel, but also how your cells grow and adapt. Understanding how insulin signaling splits into two main pathways is your key to optimizing metabolism, brain function, and even long-term athletic performance.
- IRS
- A branded insulin signaling 101 product family name used to identify a specific extract or formulation in research and supplement labels.
- IRS proteins (insulin receptor substrates)
- Adaptor proteins phosphorylated by the insulin receptor that recruit enzymes for downstream signaling, especially the PI3K–Akt branch.
- Glucose
- Blood sugar level, the primary energy source for cells. Fasting glucose is normal, prediabetes, ≥126 suggests diabetes.
- PI3K–Akt pathway
- A signaling branch that drives many of insulin’s metabolic effects, including GLUT4 trafficking (muscle/fat), glycogen synthesis, and suppression of hepatic glucose production.
- GLUT4 translocation
- Movement of GLUT4-containing vesicles to the cell membrane in muscle and fat, increasing glucose entry within minutes when insulin signaling is intact.
- Insulin receptor (IR)
- A cell-surface receptor tyrosine kinase that autophosphorylates after insulin binds, creating docking sites that start intracellular signaling.
- HOMA-IR (calc)
- Insulin resistance by combining fasting glucose and insulin levels.
- adaptor
- A protein that links other proteins together to facilitate intracellular signaling pathways.
- ERK
- A kinase in the MAPK pathway that regulates cellular growth and gene expression.
- GLUT4
- A glucose transporter protein that moves to the cell membrane to facilitate glucose uptake.
Picture this: you finish a meal, maybe a sandwich or a bowl of rice, and within minutes, your body starts a silent, invisible conversation. Your pancreas senses the rise in blood sugar and quietly releases insulin into your bloodstream. That moment is more than just a chemical reaction. It is the start of a carefully orchestrated process that determines whether you feel energized, focused, and ready to move—or sluggish, foggy, and slow to recover from exercise.
Most of us notice insulin only when something goes wrong, like feeling tired after eating or hearing about diabetes. But insulin is at work every single day, shaping how your muscles use fuel, how your brain gets the energy it needs, and even how your cells decide when to grow or divide. This is not just about blood sugar. It is about whether your body is primed for performance, recovery, and healthy aging.
If you are looking to optimize your athletic output, sharpen your mental clarity, or prevent the slow creep of metabolic disease as you get older, understanding this split in insulin signaling is crucial. It is the difference between a body that responds to food with precision and one that gets stuck in a cycle of fatigue and dysfunction. That brings us to the science behind insulin’s two-pronged approach—and why it matters for your life.
Whether you are a student, a busy parent, or an athlete, your cells constantly need to balance two main priorities. First, they must manage fuel—making sure glucose gets into your muscles and brain when you need it. Second, they must decide when to grow, divide, or repair themselves. Insulin is the hormone that helps your body decide between these two priorities, using a kind of biological circuit that splits into two directions.
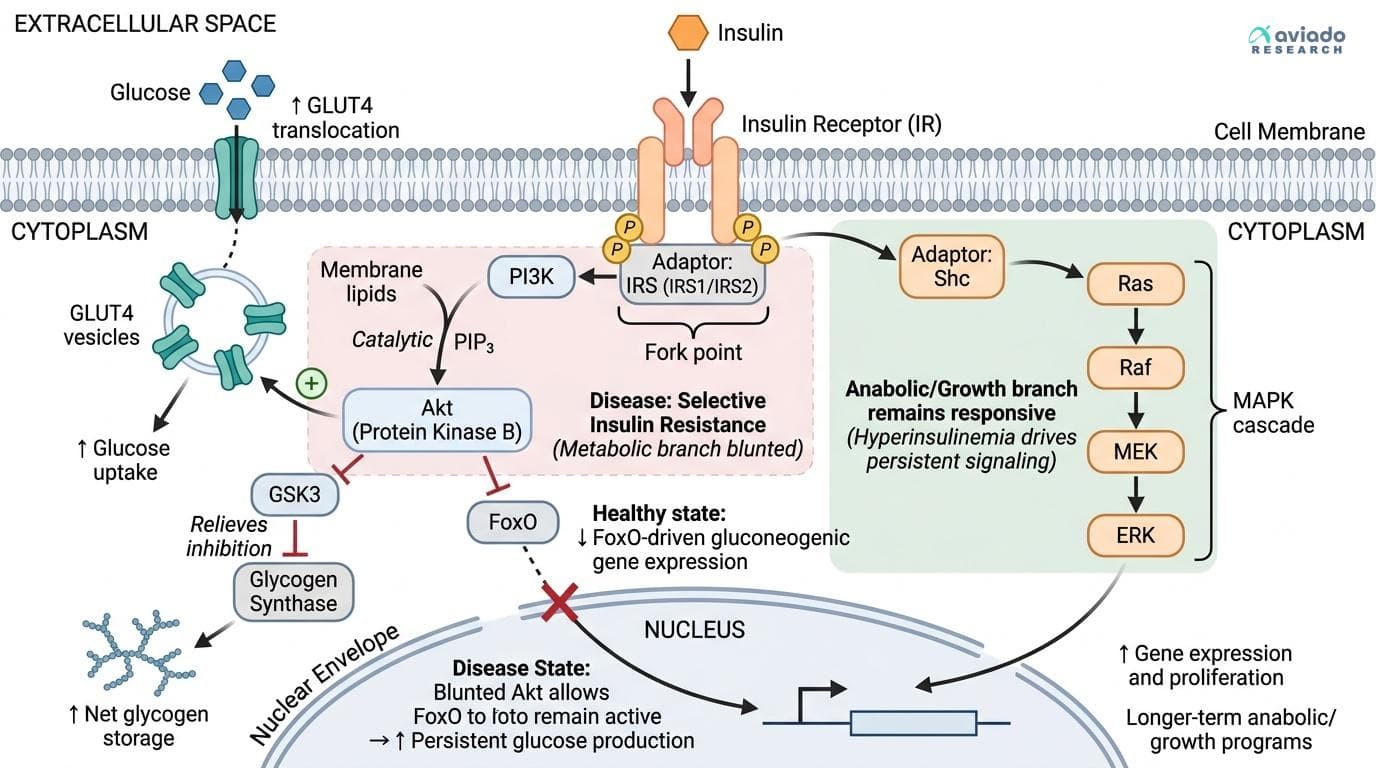
Here is how it works. When insulin binds to its receptor on the surface of muscle, fat, or liver cells, it does not just flip a single on-off switch. Instead, it activates a built-in enzyme called a receptor tyrosine kinase. This enzyme tags the receptor and nearby proteins with phosphate groups—a process called phosphorylation. These tags are like sticky notes, attracting other proteins that read the message and carry it deeper into the cell.
From this tagged platform, the insulin signal splits. One branch—the metabolic arm—controls how quickly your cells take up glucose and store it as glycogen or fat. This is the fast lane, acting within minutes after a meal. The other branch—the growth arm—travels a bit more slowly, influencing which genes get turned on or off to support cell growth, division, and even adaptation to training.
This two-branch model means that insulin is not just about what happens right after you eat. It is about how your body adapts over days, weeks, and years. Too much or too little action on either branch can tip the balance toward fatigue, weight gain, brain fog, or even increased risk for chronic disease. Understanding where you stand on this fork is the first step to better metabolic and cognitive health. Next, let’s look at how you can measure these branches in your own body.
If you want to know how well your insulin signaling is working, you need more than just a fasting blood sugar test. Here are the biomarkers that reveal which branch of the insulin pathway is thriving—and which may be struggling.
First, fasting insulin. Healthy adults typically have fasting insulin levels below ten microunits per milliliter, though optimal metabolic health often means levels closer to five. If your fasting insulin is elevated—say, above fifteen—it usually signals that your body is working harder than it should just to keep blood sugar in check. That suggests the metabolic branch of insulin signaling is starting to falter.
Next, look at HOMA-IR, the homeostatic model assessment of insulin resistance. This combines your fasting insulin and fasting glucose to estimate how resistant your cells are to insulin. A HOMA-IR below one is ideal for most adults, indicating strong insulin sensitivity. Values above two suggest you are developing resistance, meaning your metabolic branch is not responding efficiently.
Another key biomarker is your post-meal, or postprandial, glucose. After eating, your blood glucose should peak below one hundred forty milligrams per deciliter and return to baseline within two hours. If it stays elevated longer, or spikes much higher, your cells are not moving glucose inside quickly enough—a sign that the GLUT4 transporter system, driven by insulin’s metabolic branch, is sluggish.
On the growth side, we do not have a single lab value, but indirect clues are available. High fasting insulin or persistently elevated growth markers, such as IGF-1, can mean your growth pathways are still firing, even as your metabolic branch slows down. This is important because it helps explain why some people develop high insulin levels yet still experience cell growth in tissues where it might not be needed, increasing risk for unwanted tissue growth or even cancer in extreme cases.
Finally, liver function markers like elevated fasting glucose, increased triglycerides, or abnormal liver enzymes may indicate that insulin’s ability to suppress glucose production in the liver is weakening. That is a direct sign the metabolic branch is not keeping up.
So what does all this mean? If your metabolic markers are off, but your growth markers remain high, you are in a state known as selective insulin resistance. This is the stage where your body starts compensating with more insulin, which can worsen metabolic problems even as some growth signals stay active. Knowing your numbers helps you spot trouble early—and gives you a roadmap for intervention. Let’s talk about what you can actually do to optimize both branches.
If your goal is to keep both arms of insulin signaling healthy—for better metabolism, sharper brain function, and athletic performance—there are targeted supplements and strategies you can use. Here is how you can support the metabolic and growth branches, with a focus on what actually works and why.
First, consider alpha-lipoic acid. Doses of three hundred to six hundred milligrams per day have been shown in randomized trials to improve insulin sensitivity, especially in people at risk for metabolic syndrome. Alpha-lipoic acid works by boosting the insulin-stimulated movement of GLUT4 transporters to the cell surface, which helps your muscle and fat cells pull in more glucose after a meal. This directly supports the metabolic branch of insulin signaling.
Berberine is another option. Doses of five hundred milligrams two to three times daily have been shown in clinical trials to lower fasting insulin and improve glucose control. The mechanism? Berberine activates a cellular energy sensor called AMPK, which not only increases glucose uptake but also reduces liver glucose production. This helps correct the imbalance when your liver keeps releasing too much sugar, even after you have eaten.
Magnesium is an often-overlooked mineral, but it is essential for normal insulin receptor function. Most adults need at least three hundred to four hundred milligrams daily, and deficiency is linked to higher insulin resistance. Magnesium acts as a cofactor for the enzymes that add those all-important phosphate tags to the insulin receptor and its partners.
Omega-3 fatty acids, found in fish oil or algae oil, support the flexibility of cell membranes and reduce inflammation that can blunt insulin signaling. A landmark Finnish trial published in twenty-twenty-four followed twenty-four hundred adults for five years and found that higher omega-3 intake was associated with lower HOMA-IR and better insulin sensitivity, independent of other lifestyle factors.
Finally, focus on protein timing and quality. Protein-rich meals stimulate insulin release, but they also help preserve muscle mass, which is the main site of glucose disposal. Aim for twenty to thirty grams of high-quality protein at each meal to keep muscle insulin sensitivity high—this is especially important as you age or increase your training load.
The science is clear: these interventions help both arms of the insulin pathway stay responsive, reducing risk for both metabolic slowdowns and unwanted growth signals. But supplements are only part of the picture. The biggest levers are still in your daily habits. Let’s look at those next.
Nothing tunes your insulin response like moving your body. When you exercise, especially with resistance or interval training, your muscles become more sensitive to insulin almost immediately. This is because muscle contractions can move GLUT4 transporters to the cell surface, pulling in glucose even without insulin. After a workout, your cells are primed to respond to insulin, which means your metabolic branch works better and your overall blood sugar stays stable.
Aim for at least one hundred fifty minutes of moderate activity each week, with two to three sessions of strength training. Even brisk walking after meals can cut post-meal blood glucose spikes by as much as thirty percent. That means less demand on your insulin system and a stronger metabolic response with every meal.
Sleep is the next big lever. Just one night of poor sleep can make your cells less responsive to insulin the next day. Chronic sleep debt raises cortisol and inflammation, both of which interfere with the insulin receptor and its downstream partners. Adults should target seven to nine hours per night, with a regular sleep schedule. If you struggle with sleep, address caffeine, screen time, and stress before looking to supplements.
Nutrition is where most people can make the biggest impact. Focus on whole, unprocessed foods with plenty of fiber, lean proteins, and healthy fats. Fiber slows the absorption of glucose, reducing spikes in blood sugar and the insulin response. Healthy fats, like those from olive oil, nuts, and fatty fish, reduce inflammation and support cell membrane health, making insulin signaling more efficient. Avoid excessive added sugars and refined carbohydrates, which can overwhelm your metabolic branch and lead to insulin resistance over time.
Finally, manage stress. Chronic psychological stress activates pathways that compete with insulin, such as the release of adrenaline and cortisol. These hormones make your cells less sensitive to insulin and can tip the balance toward insulin resistance. Mindfulness, regular physical activity, and social connection are all proven ways to keep your stress hormones in check.
When you combine these lifestyle habits with targeted supplements, you create an environment where both arms of insulin signaling can thrive. This is how you set the stage for lifelong metabolic and cognitive health. But how do you know if something is going wrong? Here are the early signs to watch for.
Your body often whispers before it shouts. The earliest signs that your insulin signaling is off are subtle, but they can show up in your energy, mood, and exercise recovery long before lab tests catch a problem.
Do you feel unusually sleepy or foggy after eating a carb-heavy meal? That is often a sign that your metabolic branch is slowing down, leaving glucose in your bloodstream instead of moving it quickly into muscle and brain cells. If you notice that it takes longer to recover from workouts, or that your muscle gains have stalled despite training, this can also signal that insulin’s action on muscle cells is weakening.
Unexpected weight gain, especially around your midsection, can be another clue. When the metabolic branch of insulin signaling starts to fail, your body compensates by pumping out more insulin. This high-insulin state favors fat storage, particularly in the liver and abdomen.
Other early warning signs include skin changes like dark, velvety patches in the folds of your neck or underarms, known as acanthosis nigricans. This is a classic marker of insulin resistance, especially in younger adults or those with a family history of diabetes.
Mood swings, difficulty concentrating, or feeling wired but tired can reflect the brain’s response to unstable glucose. Your brain relies on steady glucose delivery, and when insulin signaling is disrupted, you can experience both highs and crashes in focus and emotional stability.
If you notice any of these signs, it is time to check your fasting insulin, fasting glucose, and post-meal glucose, and to talk with a healthcare provider about your risk factors. The earlier you intervene, the easier it is to restore balance and avoid the long-term consequences of insulin resistance. That brings us to the big picture and what you can do starting today.
You now know that insulin is not just about keeping your blood sugar normal. It is a master regulator, splitting its signal into two main branches: one that manages fuel right now, and another that guides cell growth and adaptation over time. Both are crucial for metabolic health, brain function, and athletic performance.
When one branch falls behind, your body tries to compensate, often leading to higher insulin levels and more stress on the whole system. That is why early detection and targeted intervention matter so much. By tracking your biomarkers, supporting your metabolism with evidence-based supplements, and dialing in lifestyle habits like exercise, sleep, and nutrition, you can keep both arms of the insulin pathway working in harmony.
The science keeps advancing, but the core message remains the same: you have enormous power to shape how your body responds to food, stress, and aging. The sooner you act, the better your chances of staying energetic, clear-headed, and resilient for the long haul. If you have been waiting for a sign to take your metabolic health seriously, this is it. Your next meal, your next workout, and your next night’s sleep are all opportunities to tip your insulin pathways in your favor. Start now, and your future self will thank you.
How Insulin Signaling Works: The Two-Fork Pathway That Controls Both Metabolism and Cell Growth
One receptor, two major cascades, and a critical branch point that shapes health and disease
Diagram glossary
- adaptor:
- A protein that links other proteins together to facilitate intracellular signaling pathways.
- ERK:
- A kinase in the MAPK pathway that regulates cellular growth and gene expression.
- glucose:
- A simple sugar that serves as the primary energy source for cells.
- GLUT4:
- A glucose transporter protein that moves to the cell membrane to facilitate glucose uptake.
- GSK3:
- An enzyme inhibited by Akt that regulates glycogen synthesis and metabolism.
- Insulin:
- A pancreatic hormone that signals nutrient availability and regulates metabolism and cellular growth.
- IRS1/IRS2:
- Adaptor proteins that translate insulin receptor activation into downstream metabolic signaling actions.
- MAPK:
- A family of kinases that mediates cellular responses to growth signals like insulin.
- MEK:
- A kinase that phosphorylates and activates ERK in the MAPK growth signaling pathway.
- PI3K:
- An enzyme recruited by IRS proteins that generates PIP3 to propagate metabolic signals.
- PIP3:
- A signaling lipid created by PI3K that helps activate Akt for metabolic outputs.
Conclusions
Insulin signaling can be understood as a Y: insulin activates the insulin receptor (the stem), then signaling splits into a PI3K–Akt branch that primarily controls metabolic fuel handling and a Ras–MAPK branch that more strongly reshapes gene expression linked to growth programs. Many real-world disease patterns make more sense when you think of these two branches as partly separable rather than perfectly coupled.
This article simplifies insulin signaling to two dominant branches and focuses on canonical mammalian pathways; it does not fully cover tissue-specific differences (e.g., IRS1 vs IRS2 roles), isoforms (Akt1/2/3), additional nodes (mTORC1, PTEN, phosphatases), or detailed feedback mechanisms that shape “selective insulin resistance.” The idea that metabolic vs growth-related outputs can be differentially impaired is supported by mechanistic and genetic evidence (e.g., FoxO regulation downstream of Akt and insulin receptor loss-of-function phenotypes) [2,3], but the exact pattern and causes of branch-selective defects vary by tissue, model system, and measurement approach.
Track this in your stack
See how insulin signaling 101 relates to your health goals and monitor changes in your biomarkers over time.
Sources (4)
This article informs how supplementation moves 1 marker.
Each biomarker page clusters supplements, ranges, and the evidence behind every score. Useful when you're starting from a number, not a goal.
