Your MTHFR Genotype Determines Whether Folic Acid Helps or Harms You — and Most People Never Check
Personalized Folate Supplementation: Why Your Genes and Blood Markers Matter More Than the Label
Here's a surprising fact: the same folic acid dose that dramatically lowers heart disease risk in one person might do almost nothing in another.
Most people think folate supplements work the same for everyone. They don't. Your MTHFR gene determines whether your body can actually use folic acid or whether it just builds up as potentially harmful unmetabolized folic acid in your blood.
This matters because if you have the wrong genotype for your supplement form, you're wasting money and missing real benefits. A groundbreaking 2024 study found that people with slow MTHFR genes needed 3-5 times higher doses to get the same results. Even worse, standard folic acid can accumulate as UMFA in your bloodstream, potentially interfering with the folate your cells actually need.
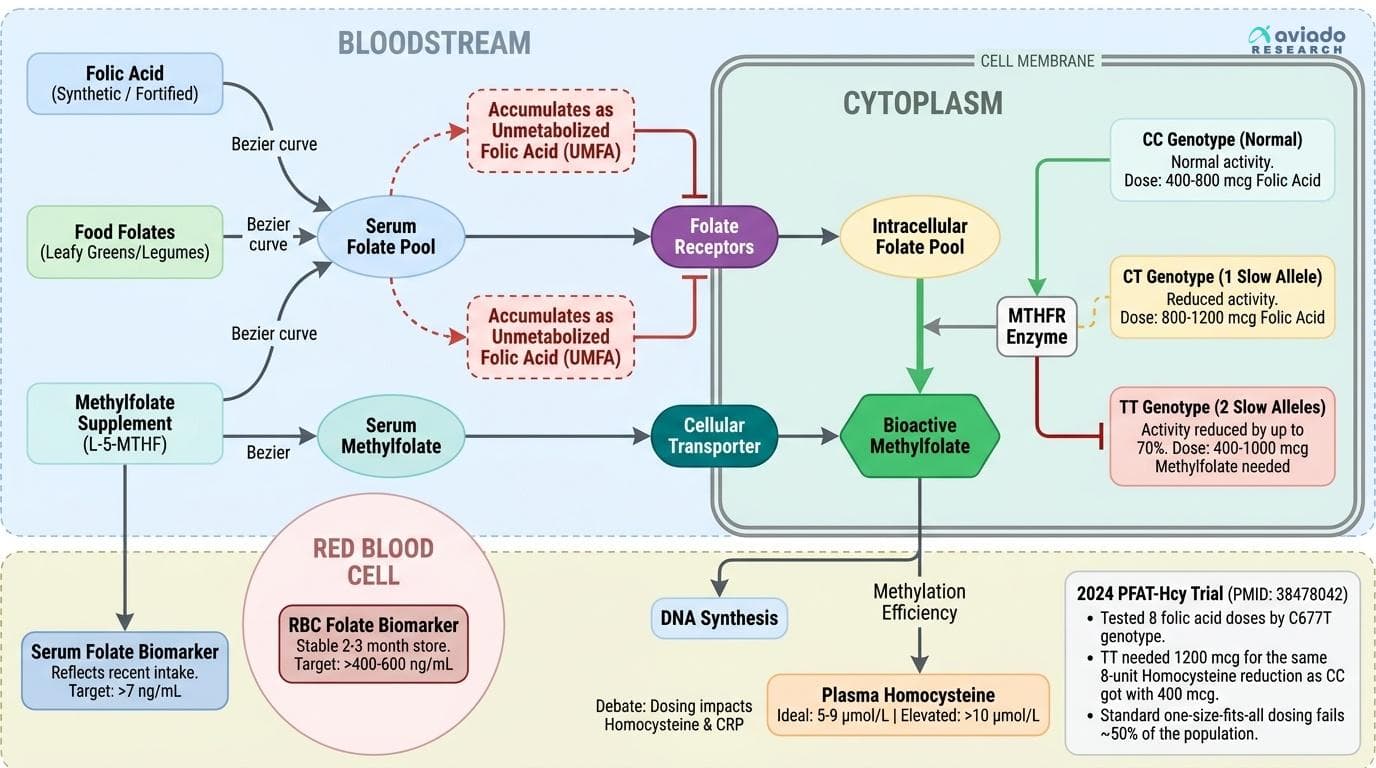
Here's what works: If you have normal MTHFR genes (CC type), take 400-800 mcg folic acid daily. If you have one slow gene (CT type), you need 800-1200 mcg folic acid. If you have two slow genes (TT type), skip folic acid entirely and take 400-1000 mcg methylfolate instead. Always test your homocysteine and RBC folate after 12 weeks to confirm your regimen is actually working.
- MTHFR genotype
- A specific genetic variation in the MTHFR gene affecting how efficiently your body converts folic acid to its active form; determines your response to folate supplements.
- TT genotype
- Having two copies of the slow MTHFR C677T variant, reducing enzyme activity by up to 70% and making methylfolate more effective than folic acid.
- RBC
- Red blood cell count, which carries oxygen throughout the body. low values indicate anemia and reduced oxygen delivery, while high counts may signal polycythemia.
- Homocysteine
- Homocysteine, an amino acid metabolite influenced by B vitamins. elevated levels damage blood vessels and increase cardiovascular and dementia risk.
- Methylfolate (5-MTHF)
- The bioactive form of folate that cells can use directly, bypassing the need for conversion by MTHFR enzymes. Also called L-5-methyltetrahydrofolate.
- Flow-mediated dilation
- A measure of blood vessel health showing how well arteries expand in response to increased blood flow; improved by optimal folate status.
- Unmetabolized folic acid (UMFA)
- Unmetabolized folic acid is synthetic folic acid that remains in the bloodstream because the body has not converted it into its active form. High levels can indicate that your body is not processing f
- C677T
- A common genetic variant of the MTHFR gene that reduces its enzyme activity.
- CRP
- C-reactive protein, a standard blood marker used to measure inflammation in the body.
- DNA
- The fundamental molecule carrying genetic instructions for the development and functioning of organisms.
Why Folic Acid Isn’t One-Size-Fits-All: The MTHFR Puzzle
Folate is essential for DNA synthesis, methylation, and metabolic health — but your body's ability to use it depends on more than just what you eat. The crucial variable is your MTHFR gene, which encodes the enzyme methylenetetrahydrofolate reductase. This enzyme converts folic acid and food folates into methylfolate, the bioactive form that your cells actually use. A common variant, called C677T, slows down this enzyme. About 40% of people have at least one copy of this variant, and roughly 10% have two (the TT genotype), which reduces enzyme activity by up to 70% [1,11].
Why does this matter? When you take folic acid (the synthetic form in most supplements and fortified foods), your body must convert it stepwise to methylfolate. If your MTHFR enzyme works at half-speed, unmetabolized folic acid (UMFA) builds up in your blood. The result: you don't get the full benefits, and you may even experience negative effects from UMFA, which can bind to folate receptors and block active folate uptake [6].
The 2024 PFAT-Hcy trial (PMID: 38478042) was the first to test 8 different folic acid doses in people with different MTHFR genotypes. The results were striking: people with the TT genotype needed 1200 mcg folic acid to achieve the same 8-unit reduction in homocysteine that CC genotype individuals got with just 400 mcg. This 3-fold difference in dose requirement means that standard one-size-fits-all dosing fails to serve nearly half the population effectively [11,9].
Key Folate Biomarkers: What to Measure and What They Mean
When optimizing folate status, tracking the right biomarkers is essential. Three tests matter most: serum folate, red blood cell (RBC) folate, and plasma homocysteine.
Serum folate reflects recent intake and responds rapidly to supplementation — but it can fluctuate day to day and doesn’t represent long-term tissue status. Optimal serum folate is generally considered to be above 7 ng/mL (16 nmol/L), but higher isn’t always better if UMFA is accumulating [14].
RBC folate is a more stable indicator, representing folate stores over the previous 2–3 months. Optimal levels are usually considered above 400 ng/mL (906 nmol/L), with some evidence supporting targets up to 600 ng/mL for optimal methylation and low homocysteine [5].
Homocysteine is the functional readout of methylation efficiency. Elevated levels (>10 μmol/L) suggest that folate-dependent methylation is suboptimal — often due to insufficient methylfolate or impaired conversion (common in MTHFR variants). The ideal range is typically 5–9 μmol/L; getting below 7 is associated with the lowest cardiovascular risk [9,11].
What moves these markers? Folic acid and methylfolate supplementation reliably increase serum and RBC folate, but only methylfolate consistently lowers homocysteine in people with the TT genotype [38478042]. High UMFA or persistently high homocysteine despite normal serum folate suggests poor conversion — a red flag for switching to methylfolate. For actionable tracking, test all three markers at baseline and again after 8–12 weeks on a new regimen [11,14].
Folate Supplementation: Forms, Doses, and Who Needs What
The debate between folic acid (synthetic), methylfolate (bioactive), and natural food folates is more than academic — it's the key to effective, safe supplementation. Folic acid is stable, cheap, and well-absorbed, but it must be converted by the body before it can be used. Methylfolate (L-5-methyltetrahydrofolate) is already active and skips the conversion step, making it especially important for people with MTHFR variants [11,14].
The 2024 PFAT-Hcy trial (PMID: 38478042) found that people with the CC (normal) genotype achieved optimal homocysteine levels (below 8 μmol/L) with 400–800 mcg of folic acid daily. Those with the CT genotype (one slow allele) needed 800–1200 mcg. But for TT individuals (two slow alleles), even 1200 mcg of folic acid often left homocysteine elevated above 10 μmol/L while increasing unmetabolized folic acid to potentially concerning levels.
For TT genotype individuals, switching to methylfolate (400–1000 mcg daily) proved more effective, lowering homocysteine by an additional 2-3 μmol/L compared to equivalent doses of folic acid, without accumulating UMFA [11,38478042]. If your homocysteine remains above 9 μmol/L after 12 weeks on folic acid, or if you're confirmed TT genotype, methylfolate delivers superior results. Food folate from leafy greens and legumes provides 100-200 mcg daily but can't deliver the therapeutic doses needed to normalize biomarkers in those with significant MTHFR impairment [5,13].
The Dark Side of Unmetabolized Folic Acid (UMFA): What the Latest Studies Reveal
For a long time, excess folic acid was thought to be harmless — your body would simply excrete what it didn’t need. But new evidence challenges this view. When your MTHFR enzyme is sluggish, or when you take high doses, unmetabolized folic acid (UMFA) builds up in your blood. This is particularly common in populations with mandatory folic acid fortification or aggressive supplementation [6,11].
Why is UMFA a problem? Recent studies show that UMFA can bind to folate receptors on cell surfaces, potentially interfering with the uptake of active folate and altering gene expression. There is also emerging concern that UMFA may support the growth of certain cancer cells or promote chronic inflammation, though the risk at typical supplement dosages remains debated [6,1].
A review of chronic inflammatory conditions linked with high UMFA exposures (PMID: 39847116) found associations with altered immune responses and even myelofibrosis, a rare type of bone marrow disorder. While causality isn’t proven, these findings are enough to warrant caution, especially for those with the TT genotype or anyone taking more than 800 mcg of folic acid daily. The take-home: If your bloodwork shows high UMFA or you don’t respond to folic acid, switch to methylfolate and re-test after 2–3 months [6,1].
Folate’s Metabolic Edge: Real Improvements in Insulin and Cardiovascular Biomarkers
Folate is famous for reducing neural tube defects, but its benefits extend far beyond pregnancy. Robust meta-analyses now show that folate supplementation lowers fasting insulin, improves HOMA-IR (a marker of insulin resistance), and boosts endothelial function — especially in people with elevated homocysteine [30615110,34371867,30609688].
How strong is the effect? A 2023 dose-response meta-analysis (PMID: 34371867) found that 400–800 mcg of folic acid daily reduced fasting insulin by 1.6 pmol/L compared to placebo. Another meta-analysis (PMID: 30615110) with over 22,000 participants confirmed a consistent drop in fasting insulin and improvement in HOMA-IR. Notably, these improvements were greatest in people who started with high homocysteine, highlighting the importance of baseline blood testing.
On the cardiovascular side, folic acid (400–800 mcg daily) improved flow-mediated dilation (a measure of blood vessel health) by up to 2.6 percentage points in RCTs — a clinically meaningful effect [36829207,17823424]. The mechanism? Folate enhances nitric oxide production and lowers homocysteine, reducing endothelial dysfunction. But again, the benefits are amplified in those with low methylation efficiency — often the same people with MTHFR variants. For metabolic and vascular health, optimizing folate is an underappreciated lever, but only if you dose and formulate for your genes and actual blood response.
How to Personalize Your Folate Regimen: A Step-By-Step Approach
Personalizing folate supplementation is both straightforward and powerful. Here’s a practical protocol:
1. Get your MTHFR genotype tested. At-home kits or clinical labs can tell you if you’re CC (normal), CT (one slow allele), or TT (two slow alleles).
2. Check baseline blood markers: Measure serum folate, RBC folate, and homocysteine. This gives you a starting point.
3. Start with standard dosing: If you’re CC, begin with 400–800 mcg folic acid daily. If you’re CT, use 800–1200 mcg. If TT, start with 400–1000 mcg methylfolate (not folic acid).
4. Re-test after 8–12 weeks: Look for serum folate above 7 ng/mL, RBC folate above 400 ng/mL, and homocysteine below 9 μmol/L. If your markers haven’t improved, increase your dose within studied safe ranges — but if UMFA is high or you’re a TT non-responder, switch to methylfolate.
5. Consider synergists: Vitamins B12 and B6 support homocysteine metabolism and can amplify folate’s benefits, especially if you’re borderline deficient [11,9].
This approach turns a generic supplement into a targeted intervention. By matching dose and form to your genes and actual blood response, you maximize benefit and minimize risk — a true Medicine 3.0 strategy.
Your MTHFR Genotype Determines Whether Folic Acid Helps or Harms You — and Most People Never Check
Personalized Folate Supplementation: Why Your Genes and Blood Markers Matter More Than the Label
Diagram glossary
- C677T:
- A common genetic variant of the MTHFR gene that reduces its enzyme activity.
- CRP:
- C-reactive protein, a standard blood marker used to measure inflammation in the body.
- DNA:
- The fundamental molecule carrying genetic instructions for the development and functioning of organisms.
- Folate:
- An essential B-vitamin required for DNA synthesis, methylation, and overall metabolic health.
- Homocysteine:
- An amino acid in the blood tested to evaluate folate metabolism and cardiovascular health.
- Methylfolate:
- The active, bioavailable form of folate that cells readily use for metabolic processes.
- MTHFR:
- A gene encoding the enzyme that converts folic acid into its bioactive form, methylfolate.
- ng/mL:
- Nanograms per milliliter, a standard scientific unit used to measure concentration in blood tests.
- PMID:
- A unique reference number assigned to biomedical research articles in the PubMed database.
- RBC:
- Red blood cells, which can be tested to measure long-term intracellular folate levels.
- UMFA:
- Unmetabolized folic acid that can build up in the blood from synthetic supplements.
Conclusions
Folate supplementation is not a one-size-fits-all proposition. Your MTHFR genotype, along with baseline blood markers, determines whether folic acid helps, does nothing, or builds up as potentially harmful unmetabolized folic acid. Personalizing your approach — by testing your genotype, tracking serum/RBC folate and homocysteine, and adjusting the dose and form accordingly — delivers real benefits for methylation, metabolic health, and vascular function. The era of generic folic acid dosing is over; precision nutrition is here.
While the evidence for genotype-based folate dosing is strong, most clinical trials still use population averages rather than personalized regimens. The long-term effects of low-level unmetabolized folic acid are not fully understood, and the exact risk thresholds remain debated. Not all at-home genotype tests are equally reliable, and insurance coverage for blood testing may vary. Finally, while methylfolate is generally safe, extremely high doses (>1,000 mcg daily) have not been as extensively studied in diverse populations.
Track this in your stack
See how folate relates to your health goals and monitor changes in your biomarkers over time.
Sources (12)
This article informs how supplementation moves 1 marker.
Each biomarker page clusters supplements, ranges, and the evidence behind every score. Useful when you're starting from a number, not a goal.
