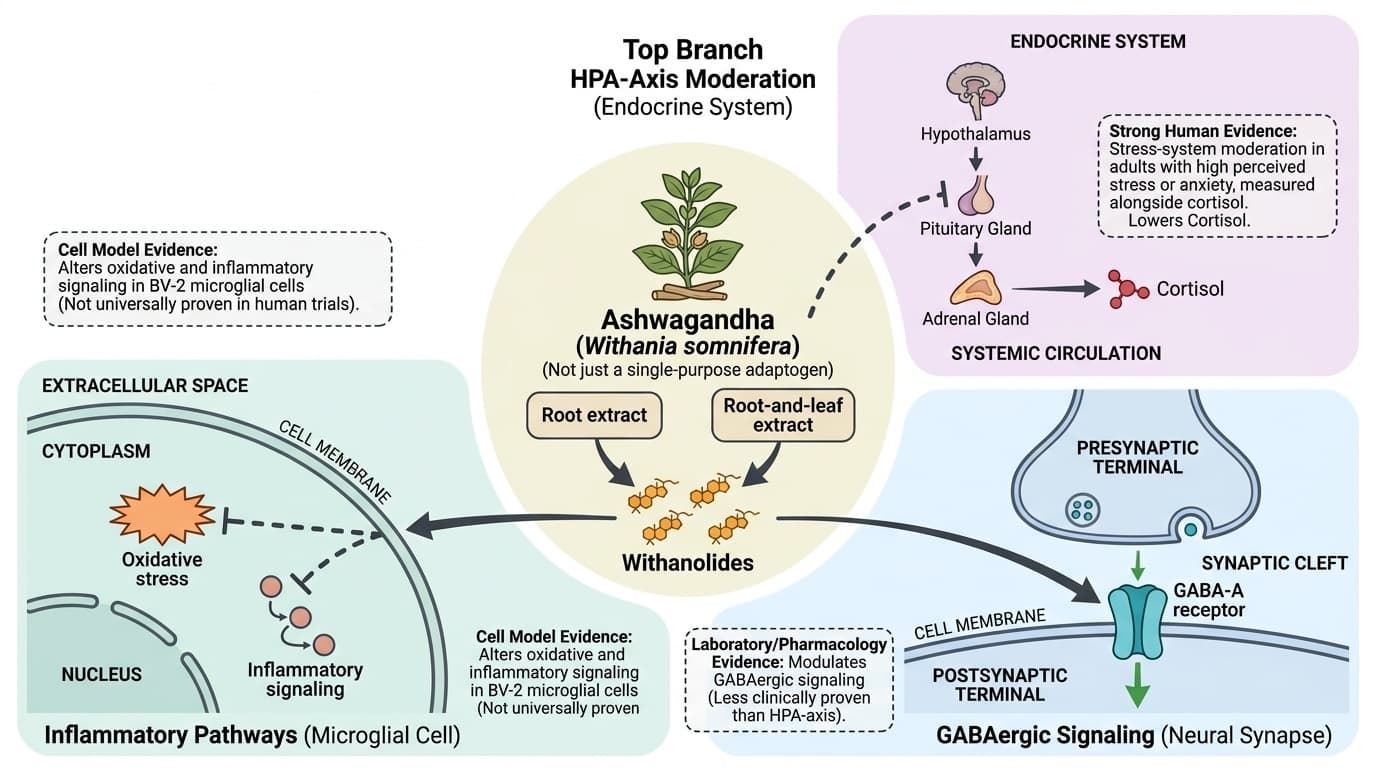
Ashwagandha Is Not Just an Adaptogen: What Its Molecules Actually Do
Withanolides and related compounds appear to work through stress, GABA, inflammatory, and endocrine pathways - not one magic stress switch.
Ashwagandha works less like a single-purpose stress pill and more like a multi-pathway botanical extract.
The best human evidence points to HPA-axis moderation, especially cortisol-related stress biology. Laboratory studies add plausible GABAergic and anti-inflammatory mechanisms, but those signals are not equally proven in people.
That distinction matters because "adaptogen" is a marketing label, not a mechanism. In trials, standardized extracts have reduced perceived stress, anxiety, cortisol, and sleep disruption in selected groups, often over 8 to 10 weeks. The same evidence does not prove that every ashwagandha product lowers cortisol, boosts testosterone, or acts as a natural sedative.
For readers who track biomarkers, cortisol, sleep metrics, and training recovery can help contextualize whether the mechanism is relevant. For readers who do not test, the practical signal is simpler: stress that carries into sleep, delayed recovery, anxious tension, or fatigue after manageable demands. Ashwagandha is most defensible when those patterns are present and the product matches the extract type used in human trials.
The key editorial caution is extract specificity. A full-spectrum root extract, a root-and-leaf aqueous extract, a high-withanolide preparation, and a piperine-combined formula should not be treated as one intervention. The mechanism is credible enough to explain why some people respond, but not precise enough to promise the same effect from every capsule.
Ashwagandha Is Not Just an Adaptogen: What Its Molecules Actually Do
Ashwagandha mechanism map: HPA-axis moderation, GABAergic signaling, extract chemistry, and inflammatory pathways.
Diagram glossary
- Adaptogen:
- A marketing label for botanical substances claimed to help the body resist stress.
- BV-2:
- An immortalized murine microglial cell line commonly used in neuroinflammation and neurological research.
- endocrine:
- Relating to the system of glands that secrete hormones directly into the bloodstream.
- GABA-A:
- A major inhibitory neurotransmitter receptor in the brain involved in reducing neuronal excitability.
- HPA:
- The hypothalamic-pituitary-adrenal axis, a major neuroendocrine system that regulates the body's stress response.
- Withanolides
- Steroidal lactone compounds used to standardize many ashwagandha extracts and linked to several proposed mechanisms.
- HPA axis
- The hypothalamus-pituitary-adrenal stress circuit that regulates cortisol and stress adaptation.
- Cortisol
- A stress hormone often measured in ashwagandha trials to assess HPA-axis activity.
- GABA-A receptor
- An inhibitory nervous-system receptor studied as one possible explanation for calming and sleep effects.
- NF-kB
- A transcription pathway involved in inflammatory signaling and oxidative stress responses.
- Root extract
- An ashwagandha preparation made from root material; not interchangeable with all root-and-leaf extracts.
- Root-and-leaf extract
- A preparation containing both plant parts, often with a different withanolide profile than root-only extracts.
- Standardization
- Chemical specification of an extract, usually by withanolide content or named marker compounds.
What does ashwagandha actually do in the body?
Ashwagandha is best understood as a multi-constituent signaling extract, not a single sedative or hormone booster. Its strongest human mechanism signal is stress-system moderation, while cell and pharmacology studies point to GABAergic, inflammatory, antioxidant, and endocrine effects that may explain why outcomes differ across people and extracts.
The marketing word "adaptogen" is too broad to be useful by itself. In the literature, ashwagandha usually means a standardized extract of Withania somnifera root, root plus leaf, or a branded preparation enriched for withanolides. Those preparations are not chemically identical. A 2022 pharmacokinetic study measured withanosides and withanolides after dosing and showed why extract chemistry matters: different molecules appear, peak, and clear differently after ingestion [8].
That chemistry explains why ashwagandha does not behave like one clean drug target. Human trials repeatedly connect it to stress and sleep outcomes, but the plausible pathway is a network: HPA-axis tone, inhibitory neurotransmission, inflammatory signaling, and possibly reproductive or androgen-related signaling. The practical read is conservative. Ashwagandha may be useful when the problem looks like stress load, poor sleep, or under-recovery, but the mechanism does not justify using it as a universal performance enhancer.
| Claim | Mechanism signal | Evidence type | How strong is it? | |---|---|---|---| | Lower stress biology | HPA-axis markers such as cortisol and DHEA-S | Human RCTs | Moderate | | Calmer neural signaling | GABA-A and GABA-rho receptor activity | Cell and receptor studies | Preliminary | | Lower neuroinflammation | NF-kB, oxidative stress, microglial markers | Cell studies | Preliminary | | Better recovery | Strength, muscle damage, testosterone-adjacent outcomes | Human RCTs | Preliminary to moderate |
Why is the HPA axis the most credible mechanism?
The HPA axis is the strongest human-backed mechanism because multiple randomized trials measured stress symptoms alongside cortisol or related hormones. The pattern is not "ashwagandha lowers cortisol in everyone." It is better framed as modulation of an activated stress system, especially in adults entering trials with high perceived stress or anxiety.
The cleanest early trial randomized 64 adults with chronic stress to high-concentration root extract or placebo for 60 days. The ashwagandha group reported lower stress scores and showed a larger cortisol reduction; the paper reported an average serum cortisol decrease of 27.9% in the treatment group, compared with 7.9% in placebo [1]. That does not prove the HPA axis is the only pathway, but it anchors the stress claim in a measured hormone.
Later trials tested different extracts and doses. Lopresti and colleagues used 240 mg/day of a standardized extract for 60 days in stressed adults and found greater reductions in morning cortisol compared with placebo, alongside changes in DHEA-S [2]. Salve and colleagues compared 250 mg/day and 600 mg/day root extract arms for eight weeks and reported dose-related improvements in perceived stress, anxiety, cortisol, and sleep quality [3].
The newest dose-response trial adds useful nuance because it tested 125, 250, and 500 mg/day of a characterized root-and-leaf extract for eight weeks in adults with chronic stress. It reported improvements in Perceived Stress Scale scores and stress-related biomarkers, including cortisol and ACTH, with no serious adverse events [11]. If you track labs, cortisol can contextualize the pathway. If you do not, the relevant real-world signs are sleep continuity, tension, recovery, and how long stress feels like it lingers.
Does ashwagandha act like a GABA drug?
Ashwagandha has GABAergic activity in laboratory systems, but it should not be described as a benzodiazepine-like drug. The best evidence shows components in aqueous root extract can activate inhibitory GABA receptor currents in experimental models, while human sleep and anxiety trials suggest effects that are compatible with calmer neural signaling.
Candelario and colleagues tested aqueous Withania somnifera root extract on mammalian ionotropic GABA-A and GABA-rho receptors using oocyte electrophysiology. The extract activated GABA-A currents with an EC50 equivalent to 4.7 mg/mL, and GABA-rho receptors were reported as 27-fold more sensitive than GABA-A receptors in that model [5]. That is direct receptor evidence, but it is not the same as demonstrating the same concentrations in the human brain.
Follow-up natural-products work complicates the story. Sonar and colleagues searched for GABA-A receptor modulators in Withania somnifera and identified ferulic acid esters and withanolide-related compounds as candidates, reinforcing that the GABA signal may come from more than one molecule [6]. This is why "withanolides equal GABA" is too simple.
Human sleep data are directionally consistent. In a 60-person insomnia and anxiety RCT, 300 mg of root extract twice daily improved sleep onset latency, total sleep time, sleep efficiency, and anxiety measures compared with placebo [4]. A separate 2021 trial in healthy stressed adults reported cognitive-function changes after root extract supplementation, which may reflect stress-system effects as much as direct nootropic action [12]. The responsible interpretation is that GABAergic signaling is plausible, not settled.
What do withanolides do to inflammatory signaling?
The inflammatory mechanism is biologically plausible but less clinically proven than the stress mechanism. Withanolides can alter oxidative stress and inflammatory pathways in cell models, including microglial systems, but human trials have not yet shown a clear, repeated inflammation-marker outcome that matches the strength of the cortisol evidence.
Sun and colleagues studied Withania somnifera and several withanolides in BV-2 microglial cells, a common model for neuroinflammatory signaling. The paper reported attenuation of oxidative and inflammatory responses and up-regulation of antioxidant response pathways [7]. This matters because chronic stress and poor sleep can interact with inflammatory signaling, but the model is not a human clinical endpoint.
The inflammatory hypothesis is useful for framing, not for overclaiming. NF-kB, oxidative stress, and microglial activation are upstream concepts; readers should not translate them into an expected drop in hs-CRP or a guaranteed reduction in neuroinflammation. If you track hs-CRP, ashwagandha is not one of the better-supported primary levers. If you do not track biomarkers, the practical signal is still indirect: less stress reactivity, better sleep, or improved recovery would be more clinically meaningful than assuming inflammation changed.
This distinction matters for AEO and editorial accuracy. Mechanism articles should teach the pathway without inflating the outcome. Ashwagandha may calm inflammatory tone in experimental systems [7], and some stress trials measure inflammatory markers as secondary endpoints [11], but the evidence does not yet support positioning it as a primary anti-inflammatory supplement.
Why do root, leaf, and extract standardization matter?
Form matters because ashwagandha is a botanical matrix, not a purified molecule. Root-only extracts, root-and-leaf extracts, high-withanolide extracts, and piperine-combined formulas can differ in active-compound profile, pharmacokinetics, dose, and clinical behavior. A dose in milligrams is incomplete unless the extract is identified.
The 2022 UHPLC-MS/MS pharmacokinetic study is especially relevant for readers comparing products. Modi and colleagues measured withanosides and withanolides from Withania somnifera and showed that multiple constituents can be tracked after dosing [8]. That supports a simple editorial rule: cite the extract used in the trial, not just the plant name.
Clinical trials also used different preparations. Chandrasekhar used a high-concentration full-spectrum root extract at 300 mg twice daily [1]. Lopresti used 240 mg/day of a standardized extract [2]. Salve compared 250 mg/day and 600 mg/day root extract arms [3]. Pandit tested 125, 250, and 500 mg/day of a characterized aqueous root-and-leaf extract [11]. Majeed used 500 mg root extract with 5 mg piperine, standardized to 2.5% full-spectrum withanolides [10].
| Extract pattern | Trial example | Dose used | Mechanism implication | |---|---:|---:|---| | Full-spectrum root extract | Chandrasekhar 2012 | 300 mg twice daily | Stress and cortisol outcomes [1] | | Standardized Shoden extract | Lopresti 2019 | 240 mg daily | HPA-axis hormone signal [2] | | Root extract dose comparison | Salve 2019 | 250 or 600 mg daily | Dose-related stress and sleep signal [3] | | Root plus leaf aqueous extract | Pandit 2024 | 125, 250, or 500 mg daily | HPA biomarkers and dose response [11] | | Root extract plus piperine | Majeed 2024 | 500 mg plus 5 mg piperine | Mood and serotonin signal [10] |
How do clinical effects map onto these pathways?
The clinical pattern fits a stress-sleep-recovery network better than a single target. Cortisol and anxiety outcomes map most directly to the HPA axis, sleep outcomes may involve GABAergic and stress-system effects, and strength or recovery outcomes may reflect lower stress load plus endocrine or anti-inflammatory influences.
The stress pathway has the clearest human signal. Trials in stressed adults show improvements in perceived stress or anxiety with measured cortisol changes [1][2][3]. The sleep pathway is also clinically visible. Langade's insomnia trial found improvements in sleep measures after 10 weeks of 300 mg twice-daily root extract [4], while Gopukumar's 2021 trial suggests cognition changes may track with stressed-adult physiology rather than isolated brain enhancement [12].
Recovery outcomes are promising but less direct mechanistically. Wankhede and colleagues randomized 57 men beginning resistance training to 300 mg root extract twice daily or placebo for eight weeks. The treatment group improved bench press and leg extension strength more than placebo and had greater reductions in exercise-induced muscle damage markers [9]. That does not prove a muscle-specific mechanism, but it shows that the stress-recovery story may extend beyond mood.
Endocrine-adjacent findings need even more care. A small thyroid trial in subclinical hypothyroidism reported TSH, T3, and T4 changes after 600 mg/day root extract [13], and a perimenopause trial reported symptom changes after root extract supplementation [14]. Those results show why endocrine pathways belong in the mechanism map, but they do not make ashwagandha a broad hormone intervention.
| Outcome domain | Human signal | Likely pathway | Editorial confidence | |---|---|---|---| | Stress and anxiety | Multiple RCTs and meta-analyses | HPA-axis moderation | Moderate | | Sleep quality | RCTs in insomnia/anxiety and pooled sleep analyses | HPA plus GABAergic signaling | Moderate | | Mood symptoms | RCT with serotonin increase | Serotonergic and stress pathways | Preliminary | | Strength and recovery | One notable resistance-training RCT | Recovery, endocrine, inflammation | Preliminary | | Inflammation markers | Mostly cell and secondary-trial signals | NF-kB and oxidative stress | Preliminary |
Who is most likely to feel a mechanism-level effect?
The best candidate is not "everyone." The strongest rationale is for people whose daily pattern already looks stress-loaded: tension, poor sleep continuity, anxious rumination, slow workout recovery, or fatigue after otherwise manageable demands. Biomarker tracking can add context, but it is not required to understand the evidence.
If you track cortisol, sleep metrics, or training recovery, the mechanism hypothesis predicts the biggest change when those signals are strained at baseline. If you do not track anything, the same logic applies through lived patterns: whether sleep feels more continuous, whether stress resolves faster, and whether training soreness or mental tension stops carrying into the next day.
The responder idea is supported by trial design as much as by individual biomarker data. Many positive studies enrolled stressed, anxious, sleep-impaired, or training-specific populations rather than universally healthy adults [1][2][3][4][9]. A trial in mild to moderate depression and anxiety also used a specific root extract plus piperine and reported increased serotonin alongside symptom improvement over 90 days [10]. Those findings should not be generalized to every product or every person.
The non-responder case is just as important. If stress, sleep, mood, and recovery are already stable, the expected mechanism-level gain is smaller. If the product is unstandardized, old, poorly labeled, or using a different extract than the trials, the biological premise weakens. And if the goal is a targeted biomarker like ApoB, HbA1c, or hs-CRP, ashwagandha is not the first supplement to consider.
Where does the mechanism story overreach?
The mechanism story overreaches when ashwagandha is treated as a universal cortisol blocker, testosterone booster, sleep drug, or anti-inflammatory. The literature supports a more specific claim: certain standardized extracts can improve stress-related outcomes in selected populations, with plausible but uneven support for GABAergic, inflammatory, and endocrine pathways.
Several limitations keep the evidence from becoming "strong." Many trials are small, short, and product-specific. Some are industry-funded. The active chemistry varies between root-only, root-and-leaf, and enhanced-bioavailability preparations [1][2][3][4][8][10][11]. The clinical domains also differ: stress, sleep, cognition, thyroid, perimenopause, and resistance training are not interchangeable outcomes [9][12][13][14].
There are also safety and boundary issues. Ashwagandha is usually well tolerated in short trials, but the mechanism touches endocrine and sedative-adjacent pathways, so caution is reasonable for pregnancy, thyroid disease, autoimmune disease, liver disease history, sedative use, or hormone-sensitive contexts. This mechanism article is not the dedicated safety article in the brief, but the pathway itself explains why "natural" is not the same as inert.
The bottom line is useful but restrained: ashwagandha is a biologically active botanical with the strongest human evidence around stress-system modulation. Its molecules may also interact with GABA receptors, inflammatory signaling, and endocrine pathways, but those mechanisms should guide better questions, not louder claims.
Conclusions
Ashwagandha's mechanism is real, but it is narrower and messier than the word "adaptogen" suggests. The most defensible human claim is HPA-axis moderation in stressed or anxious adults, supported by randomized trials that measured cortisol or related stress outcomes [1][2][3][11]. Sleep improvements fit the same stress-system story and may overlap with GABAergic signaling [4][5][6].
The broader mechanism map is preliminary. Withanolides and related compounds can influence inflammatory and antioxidant pathways in cell models [7], and pharmacokinetic work confirms that multiple Withania constituents are measurable after dosing [8]. But clinical outcomes vary by population, extract chemistry, dose, and baseline state.
The practical conclusion is restrained: use ashwagandha as a targeted stress-sleep-recovery tool, not a universal hormone or inflammation supplement. It belongs in the library as a mechanism-of-action article because the pathway story is clinically relevant, but the next most useful article for this entity page is the dose-response brief, because extract chemistry and dose are inseparable from mechanism.
Most ashwagandha trials are short, small, and tied to specific branded extracts. A result from 240 mg/day Shoden, 600 mg/day full-spectrum root extract, or 125-500 mg/day root-and-leaf extract should not be automatically transferred to an unlabeled powder. Several mechanisms come from cell or receptor studies rather than human endpoint trials. Clinical studies also enroll different populations: stressed adults, people with insomnia or anxiety, resistance-training beginners, subclinical hypothyroid patients, and perimenopausal women. That makes generalization difficult. Many trials also rely on symptom scales, which are useful but not interchangeable with objective pathway proof. Long-term safety, pregnancy safety, liver-signal risk, thyroid medication interactions, sedative-adjacent use, and autoimmune contexts need their own safety-focused review.
Track this in your stack
See how ashwagandha relates to your health goals and monitor changes in your biomarkers over time.
