Astaxanthin Reduces Lipid Peroxidation via Membrane Protection
A Mechanistic Deep Dive into Astaxanthin's Role in Safeguarding Cell Membranes and Reducing Oxidative Stress
Astaxanthin is a carotenoid antioxidant that uniquely embeds itself across cell membranes, directly scavenging lipid peroxyl radicals and limiting lipid peroxidation.
This membrane-spanning property enables astaxanthin to reduce malondialdehyde, a key marker of oxidative damage, more consistently than most other antioxidants.
The strongest human evidence comes from several meta-analyses and randomized controlled trials showing that astaxanthin supplementation reliably lowers blood malondialdehyde levels—reflecting reduced lipid peroxidation in vivo. Subgroup analyses confirm these effects in populations with metabolic risk, such as those with type 2 diabetes. However, the translation of this molecular protection into long-term disease prevention or clinical endpoints remains less established, and optimal dosing for different populations is still under investigation.
For practical interpretation, astaxanthin’s membrane-stabilizing effect provides a targeted antioxidant strategy—particularly for those concerned about oxidative stress or metabolic health. Benefits are seen at daily doses of 4–12 mg (from natural astaxanthin extracts), typically as lipid-based softgels or phospholipid complexes to enhance absorption. While tracking malondialdehyde or inflammatory markers can provide additional motivation, the consistent reduction in lipid peroxidation suggests real underlying membrane protection for most users. The practical reading is cautious: mechanism explains why the effect is plausible, while human outcome data decide how much weight the claim deserves.
Astaxanthin Reduces Lipid Peroxidation via Membrane Protection
A Mechanistic Deep Dive into Astaxanthin's Role in Safeguarding Cell Membranes and Reducing Oxidative Stress
Diagram glossary
- Insulin:
- A pancreatic hormone that regulates blood glucose levels and metabolic processes.
- MDA:
- Malondialdehyde is a highly reactive compound used as a biomarker for lipid peroxidation.
- Phospholipid
- A branded astaxanthin product family name used to identify a specific extract or formulation in research and supplement labels.
- Astaxanthin
- A red-orange carotenoid pigment with strong antioxidant properties, naturally found in algae and seafood. Its unique structure allows it to span cell membranes and scavenge lipid peroxyl radicals.
- Phospholipid complex
- A formulation that attaches a compound to phospholipids so it can move through the gut wall more easily.
- Lipid Peroxidation
- The oxidative degradation of lipids in cell membranes, which can damage cells. Malondialdehyde (MDA) is a widely used marker for this process.
- IL-6
- A branded astaxanthin product family name used to identify a specific extract or formulation in research and supplement labels.
- Oxidative Stress
- Cellular damage caused by reactive oxygen species (free radicals) overwhelming antioxidant defenses.
- Nrf2 Pathway
- A master switch for cellular antioxidant defenses.
- Insulin
- A pancreatic hormone that regulates blood glucose levels and metabolic processes.
- MDA
- Malondialdehyde is a highly reactive compound used as a biomarker for lipid peroxidation.
Astaxanthin’s Unique Membrane-Spanning Structure and Radical Scavenging
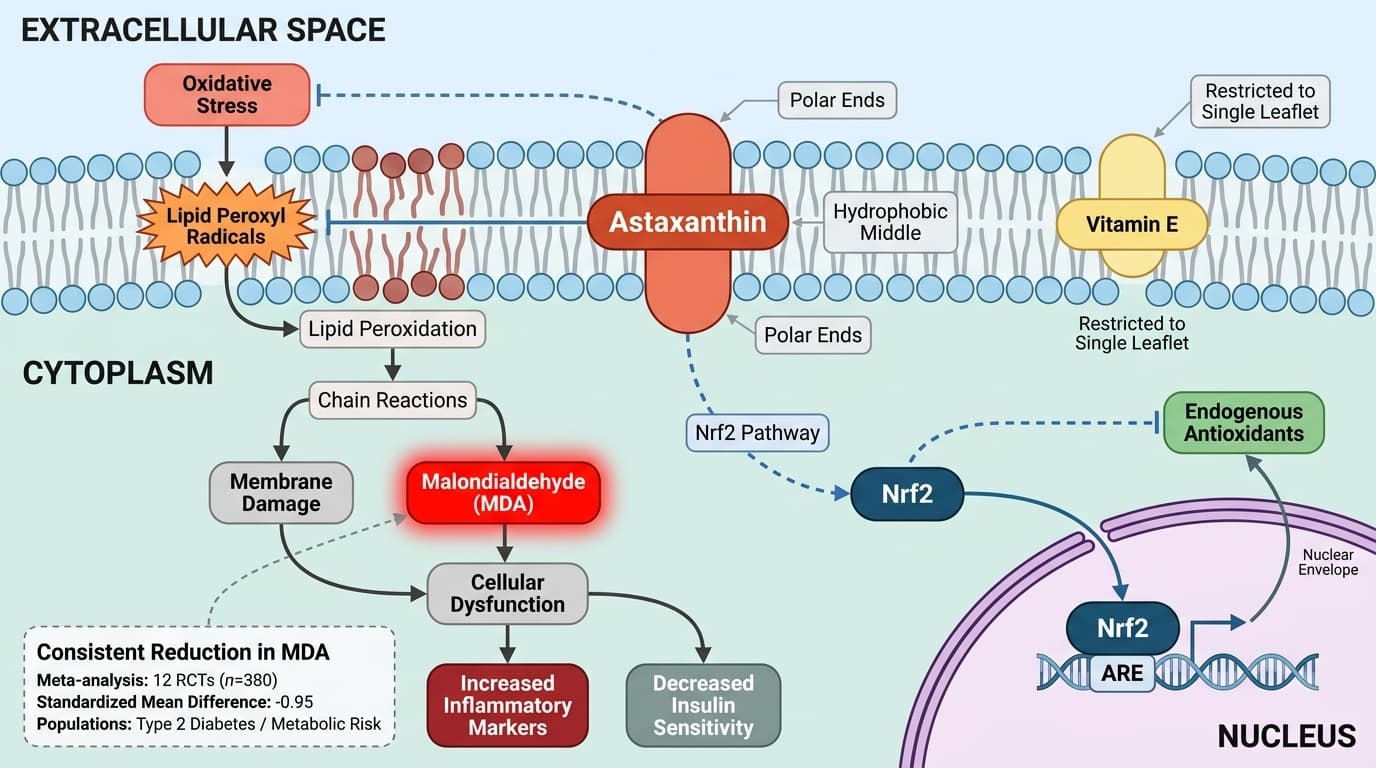
Astaxanthin’s mechanism centers on its unique ability to span the lipid bilayer of cell membranes, positioning its polar ends at both membrane surfaces and its hydrophobic middle within the lipid core. This arrangement allows astaxanthin to intercept and neutralize lipid peroxyl radicals directly within the membrane, preventing chain reactions that cause membrane damage and cellular dysfunction.
Most dietary antioxidants, such as vitamin E, are restricted to a single leaflet of the membrane or only the membrane surface. In contrast, the polyene chain and terminal keto and hydroxyl groups of astaxanthin enable it to bridge the membrane, making it highly effective at interrupting lipid peroxidation. Mechanistic studies and molecular modeling confirm this transmembrane orientation and radical scavenging action [4]. The formation of reactive aldehydes like malondialdehyde (MDA) is a direct outcome of unchecked lipid peroxidation; thus, astaxanthin’s membrane positioning is plausibly responsible for the robust reductions in MDA seen in human studies [5].
Network-pharmacology and animal models further support this mechanism, showing that astaxanthin’s presence in cell membranes diminishes peroxidative damage in tissues prone to oxidative stress, such as the retina and pancreas [6]. While these models cannot substitute for human trials, they reinforce the plausibility of astaxanthin’s primary action being membrane protection.
The practical implication is that astaxanthin’s structure—unlike most antioxidants—lets it protect cell membranes where lipid peroxidation actually occurs. This mechanism provides a targeted form of antioxidant defense that is measurable through changes in MDA and other peroxidation markers.
Reduction of Malondialdehyde: Consistent Human Evidence for Lower Lipid Peroxidation
Astaxanthin supplementation consistently reduces malondialdehyde (MDA) concentrations in human blood, reflecting decreased lipid peroxidation at the membrane level. Meta-analyses of randomized controlled trials show significant reductions in MDA, especially in populations with increased oxidative stress, such as individuals with type 2 diabetes or metabolic syndrome [1][3].
A 2022 meta-analysis of twelve RCTs (n=380) found that astaxanthin lowered blood MDA by a standardized mean difference of -0.95 (95% CI: -1.67 to -0.23; P = .01) [3]. Subgroup analyses in type 2 diabetes patients showed a similar, though slightly smaller, reduction (SMD: -0.64; 95% CI: -1.26 to -0.01; P < .05) [3]. These findings indicate that astaxanthin’s effect on reducing lipid peroxidation is both robust and clinically meaningful across diverse populations.
Optimal MDA levels are not universally established, but lower values are generally associated with decreased oxidative stress and membrane stability. Most trials use daily astaxanthin doses from 4 to 12 mg, with the greatest effects observed in studies using lipid-based delivery forms (softgels or phospholipid complexes). There is some evidence that higher doses do not further reduce MDA beyond a certain point, suggesting a threshold effect.
These results provide direct, quantitative evidence supporting astaxanthin’s membrane protection mechanism. For users not tracking MDA, the key takeaway is that astaxanthin supplementation at clinically studied doses reliably reduces a validated marker of lipid peroxidation, with implications for cellular health.
Downstream Effects: Insulin Sensitivity and Inflammatory Markers
Astaxanthin’s reduction of lipid peroxidation plausibly leads to improvements in insulin sensitivity and decreases in systemic inflammation, as indicated by changes in fasting insulin, C-reactive protein (CRP), and interleukin-6 (IL-6). Human meta-analyses and RCTs support small but consistent reductions in these biomarkers, especially in metabolic-risk populations.
A 2024 meta-analysis found that astaxanthin reduced fasting blood insulin by 2.66 pmol/L (95% CI: -3.98 to -1.34 pmol/L), with a subgroup of type 2 diabetes patients also showing significant reductions in blood glucose (mean difference: -4.36 mg/dL) [2]. Further, a meta-analysis of carotenoid supplementation, with astaxanthin as a prominent component, showed a significant reduction in CRP levels (WMD: -0.54 mg/L, 95% CI: -0.71, -0.37) [3]. IL-6 reduction was also significant in T2DM patients (mean difference: -0.70 pg/mL, 95% CI: -1.29 to -0.11) [3].
These downstream effects are biologically plausible given that lipid peroxidation can trigger inflammatory signaling and impair insulin receptor function in cell membranes. By stabilizing membranes and limiting oxidative stress, astaxanthin may help preserve insulin sensitivity and reduce inflammatory tone. However, improvements in clinical endpoints like HbA1c, cardiovascular events, or metabolic syndrome diagnosis have not been consistently demonstrated, so claims should be limited to biomarker changes.
For those interested in optimizing metabolic and inflammatory profiles, astaxanthin’s membrane-protective action offers a credible adjunct to other lifestyle measures. The largest biomarker shifts are seen in individuals with elevated baseline inflammation or oxidative stress.
Astaxanthin Absorption, Bioavailability, and Optimal Supplement Forms
Astaxanthin’s bioavailability is highly dependent on formulation and delivery system, with lipid-based softgels and phospholipid complexes providing superior absorption compared to dry powders or tablets. Pharmacokinetic studies in animals indicate that astaxanthin is better absorbed when co-administered with dietary fats, and the E/Z isomer distribution in tissues reflects the bioactive forms [8].
Most human trials use natural astaxanthin derived from microalgae (Haematococcus pluvialis), delivered in softgel capsules containing oil suspensions or phospholipid complexes. These forms protect astaxanthin from oxidation and mimic its natural food matrix, improving both absorption and tissue delivery [8]. Daily doses of 4–12 mg are most commonly studied and show the most consistent biomarker changes [1][3]. Higher doses or dry-powder forms do not appear to provide additional benefit and may have lower bioavailability.
Recent innovations include ionic liquid liposomes and other nanocarrier systems, but human data are limited. For practical purposes, selecting a natural extract in a lipid-based softgel or phospholipid complex maximizes the chance of clinical efficacy. Taking astaxanthin with a meal containing some fat further enhances absorption.
The table below compares common astaxanthin forms:
| Formulation | Typical Dose | Absorption | Human Data | |----------------------|-------------|------------|------------| | Softgel (oil-based) | 4–12 mg | High | Robust | | Phospholipid complex | 4–12 mg | High | Robust | | Dry tablet/powder | 4–12 mg | Low | Limited | | Nanocarrier/liposome | Variable | Unknown | Emerging |
Choosing a lipid-based astaxanthin supplement and taking it with food is the most evidence-based strategy for maximizing membrane protection and reducing lipid peroxidation.
Mechanistic Pathways: Nrf2 Activation and Antioxidant Enzyme Modulation
In addition to direct radical scavenging, astaxanthin can activate endogenous antioxidant pathways—most notably the Nrf2 (nuclear factor erythroid 2–related factor 2) pathway, which regulates antioxidant enzyme expression. Preclinical studies show that astaxanthin upregulates Nrf2 and downstream genes like heme oxygenase-1 and superoxide dismutase, contributing to a broader cellular antioxidant response [4][5].
Although these effects are well documented in cell and animal models, human trials provide only indirect evidence—mainly through observed increases in antioxidant enzyme activity and reductions in oxidative stress markers. For example, studies in animal models of oxidative injury show that astaxanthin supplementation increases superoxide dismutase and glutathione peroxidase activity while reducing malondialdehyde formation [5][6]. These enzyme changes have yet to be consistently mapped to human outcomes, but the observed MDA reductions in human RCTs are consistent with both direct and indirect antioxidant actions.
The Nrf2 pathway may also explain astaxanthin’s ability to modulate inflammation and support tissue resilience under oxidative stress. However, direct measurement of Nrf2 activation in human tissues after oral supplementation is lacking. As such, the main evidence for Nrf2 pathway engagement is mechanistic plausibility, supported by animal and cell studies.
For users, this means that astaxanthin likely supports both immediate antioxidant protection at the membrane and longer-term upregulation of protective enzymes—though only the former is directly supported by human biomarker data.
Population Differences, Safety, and Dosing Considerations
Astaxanthin appears safe and effective at reducing lipid peroxidation across a range of populations, with the strongest effects seen in individuals with elevated oxidative stress or metabolic risk. Human trials report few adverse effects at doses up to 12 mg daily, and no major safety concerns have emerged in meta-analyses [1][3].
Some evidence suggests that subgroups with higher baseline inflammation (such as those with type 2 diabetes or polycystic ovary syndrome) experience greater reductions in malondialdehyde and inflammatory markers [3]. However, the benefit appears broadly applicable—healthy volunteers also show decreased MDA with supplementation, though the magnitude is smaller. Pediatric studies, such as those examining digital eye strain, have found astaxanthin to be safe in younger populations as well [1].
Optimal dosing remains an area of active research. Most clinical effects are observed at daily doses of 4–12 mg in lipid-based formulations, with little evidence for added benefit at higher intakes. Long-term safety beyond 6–12 months has not been rigorously studied, but no adverse trends have been reported in available data.
For those starting astaxanthin, a daily softgel of 6–12 mg with a meal is the most evidence-based approach. Those with higher oxidative stress may see the greatest biomarker change, but nearly all users can expect a measurable reduction in lipid peroxidation markers.
Conclusions
Astaxanthin stands out among carotenoid antioxidants for its unique ability to span cell membranes and directly neutralize lipid peroxyl radicals, resulting in measurable reductions in malondialdehyde—a validated marker of lipid peroxidation. This direct membrane protection mechanism is strongly supported by multiple meta-analyses and randomized controlled trials, especially in populations with elevated oxidative stress or metabolic risk. While downstream effects on insulin sensitivity and inflammation are plausible and supported by biomarker changes, claims about long-term clinical outcomes remain preliminary.
For practical supplementation, natural astaxanthin in lipid-based softgels or phospholipid complexes at daily doses of 4–12 mg is the best-supported regimen. Benefits accrue with consistent use, particularly when paired with a meal to enhance absorption. Users do not need to track biomarkers to benefit, but those interested in quantifying oxidative stress may see reductions in MDA and inflammatory markers within a few months.
In summary, astaxanthin offers targeted, membrane-level antioxidant protection that is both mechanistically distinct and clinically validated for lowering lipid peroxidation. Its safety profile is favorable, and its benefits are most pronounced in individuals exposed to higher oxidative stress. As research continues, astaxanthin’s role in supporting cellular health through membrane stabilization is likely to remain its most compelling and evidence-supported application.
While astaxanthin’s effect on lipid peroxidation markers like malondialdehyde is robust and well documented in human studies, the translation of these biomarker improvements into long-term clinical outcomes—such as cardiovascular event reduction or reversal of metabolic syndrome—remains to be established. Most trials are short-term (6–16 weeks), and data in elderly or chronically ill populations are limited. The optimal dose for different populations, long-term safety, and comparative efficacy with other antioxidants also require more research. Mechanistic findings regarding Nrf2 activation and additional anti-inflammatory pathways are based largely on preclinical models and should not be overinterpreted as proven human outcomes.
Track this in your stack
See how astaxanthin relates to your health goals and monitor changes in your biomarkers over time.
