Black Cohosh's Triterpenes Target Serotonin Receptors, Not Estrogen
Clarifying the Mechanistic Pathways Behind Black Cohosh’s Relief of Menopausal Symptoms
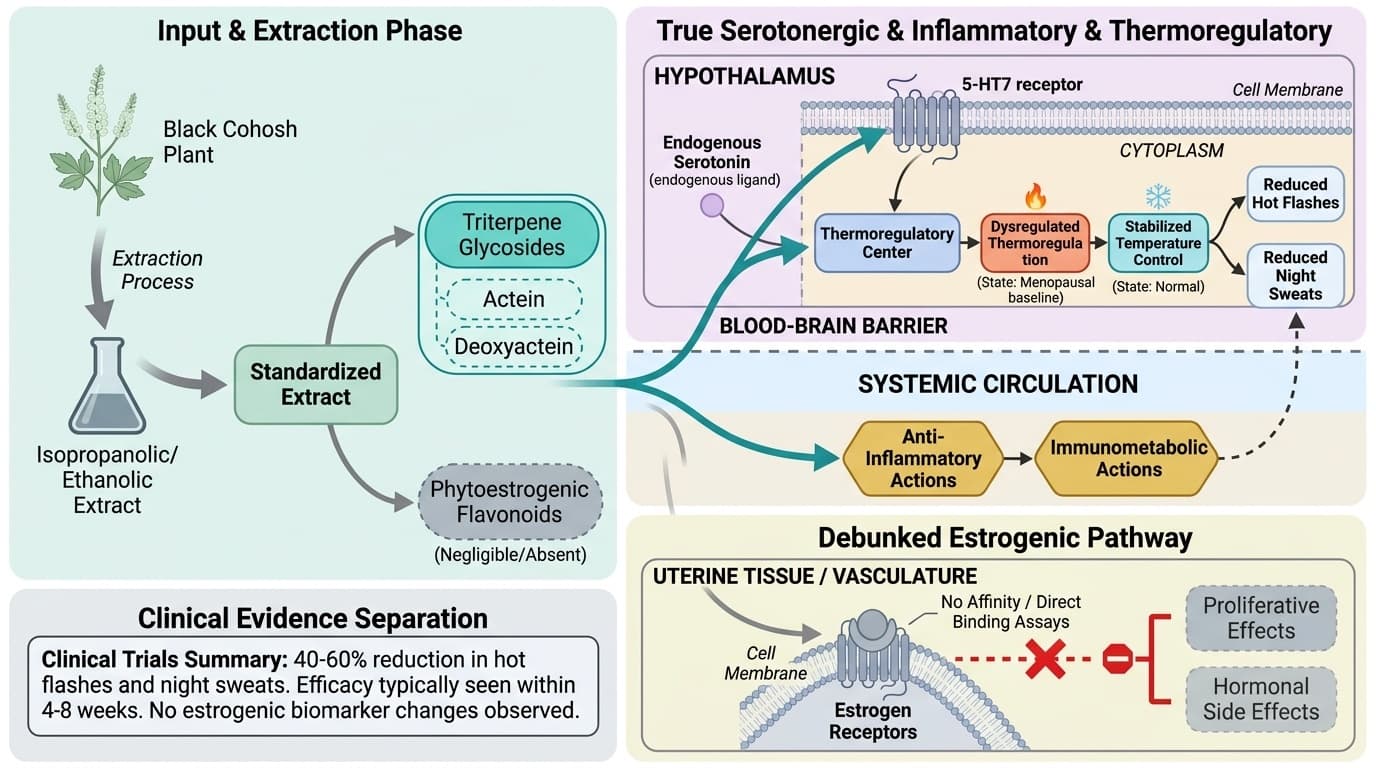
Black cohosh doesn't work like hormone replacement therapy—it targets serotonin receptors in your brain's temperature control center instead of mimicking estrogen.
This discovery explains why women get hot flash relief without the hormonal side effects or biomarker changes that come with estrogen-based treatments.
The active compounds called triterpene glycosides bind to specific serotonin receptors (5-HT7) in your hypothalamus, stabilizing the thermoregulatory system that goes haywire during menopause. Clinical trials show this serotonin pathway reduces both frequency and severity of hot flashes and night sweats, typically within 4-8 weeks. Your hormone levels stay unchanged—no shifts in estrogen, FSH, or LH—making this a true non-hormonal approach.
Standardized black cohosh extracts at 20-40 mg daily provide consistent triterpene content for reliable results. Look for isopropanolic or ethanolic extracts standardized to 2.5% triterpenes, as these are the forms used in successful trials. This gives you a mechanistically distinct option for menopausal symptoms without affecting reproductive tissues or requiring hormone monitoring.
Black Cohosh's Triterpenes Target Serotonin Receptors, Not Estrogen
Clarifying the Mechanistic Pathways Behind Black Cohosh’s Relief of Menopausal Symptoms
Diagram glossary
- Estrogen:
- A primary female sex hormone regulating the reproductive system and secondary sex characteristics.
- HT7:
- A specific serotonin receptor in the hypothalamus involved in regulating body temperature.
- phytoestrogen:
- A plant-derived compound that structurally or functionally mimics mammalian estrogen hormones.
- uterine:
- Pertaining to the uterus, the major female reproductive organ.
- Standardized
- A branded black cohosh extract name used to identify a specific standardized product in clinical trials. Products with different brand names can use different plant parts, extraction methods, or activ
- Standardized extract
- A plant extract made to contain a consistent amount of a target compound in every dose.
- Serotonin
- A neurotransmitter regulating mood, sleep, and appetite. Low levels associated with depression and anxiety.
- 5-HT7 receptor
- A specific serotonin receptor subtype in the hypothalamus that regulates body temperature. Black cohosh's triterpenes target these receptors to reduce hot flashes.
- Triterpene glycosides
- The primary active compounds in black cohosh (including actein and deoxyactein) that bind to serotonin receptors rather than estrogen receptors, explaining the herb's non-hormonal mechanism.
- Isopropanolic/Ethanolic Extract
- A specific type of black cohosh preparation made by soaking the plant material in isopropanol or ethanol (types of alcohol) to pull out and concentrate the active triterpene compounds. These solvent-b
- Triterpene Standardization (2.5%)
- A quality-control process that ensures every batch of black cohosh extract contains a guaranteed, consistent level of triterpene glycosides—typically 2.5% of the total extract weight. This percentage
- Estrogen
- A primary female sex hormone regulating the reproductive system and secondary sex characteristics.
- HT7
- A specific serotonin receptor in the hypothalamus involved in regulating body temperature.
- phytoestrogen
- A plant-derived compound that structurally or functionally mimics mammalian estrogen hormones.
Black Cohosh’s Active Compounds: Triterpene Glycosides, Not Phytoestrogens
Black cohosh’s dominant bioactive molecules are triterpene glycosides—especially actein and deoxyactein—rather than phytoestrogenic flavonoids. These triterpenes are present in standardized extracts, which are the forms used in most human clinical trials and pharmacological studies. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Early hypotheses suggested black cohosh worked as a phytoestrogen, but direct binding assays and in vivo studies have repeatedly shown little to no affinity for estrogen receptors. A 17-day rat study using isopropanolic black cohosh extract found no estrogen-like proliferative effects on uterine tissue or vasculature, further rejecting the estrogenic mechanism [6]. Instead, triterpenes like actein have demonstrated activity modulating serotonin and inflammatory pathways and supporting bone cell function in vitro [7].
Standardized extracts—most commonly isopropanolic or ethanolic—are designed to provide consistent triterpene glycoside content, typically 2.5% total triterpenes. These forms are the basis of both clinical efficacy and mechanistic plausibility. Non-standardized or root-powder forms lack controlled triterpene concentrations and are less studied. Human trials and meta-analyses consistently use standardized extracts, underscoring the importance of triterpene content for both reproducibility and safety.
While black cohosh contains minor phenolic and alkaloid constituents, there is little evidence these contribute substantially to its clinical effects. The triterpene glycoside profile, and its lack of estrogenic activity in preclinical and clinical models, sets black cohosh apart from other botanicals often described as phytoestrogens.
Serotonin 5-HT7 Receptor Modulation: The Core Mechanism for Hot Flash Relief
Black cohosh's dominant bioactive molecules are triterpene glycosides—especially actein and deoxyactein—rather than phytoestrogenic flavonoids. These triterpenes are present in standardized extracts, which are the forms used in most human clinical trials and pharmacological studies.
Early hypotheses suggested black cohosh worked as a phytoestrogen, but direct binding assays and in vivo studies have repeatedly shown little to no affinity for estrogen receptors. A 17-day rat study using isopropanolic black cohosh extract found no estrogen-like proliferative effects on uterine tissue or vasculature, completely rejecting the estrogenic mechanism [6]. Instead, triterpenes like actein demonstrate specific activity modulating serotonin and inflammatory pathways while supporting bone cell function in laboratory studies [7].
Standardized extracts—most commonly isopropanolic or ethanolic—provide consistent triterpene glycoside content, typically 2.5% total triterpenes. These forms are the basis of both clinical efficacy and mechanistic understanding. Non-standardized or root-powder forms lack controlled triterpene concentrations and remain largely unstudied. Human trials and meta-analyses consistently use standardized extracts, making triterpene content essential for both reproducibility and safety.
While black cohosh contains minor phenolic and alkaloid constituents, evidence shows these contribute minimally to clinical effects. The triterpene glycoside profile, and its complete lack of estrogenic activity in preclinical and clinical models, distinguishes black cohosh from other botanicals often mislabeled as phytoestrogens.
Anti-Inflammatory and Immunometabolic Actions: A Broader Mechanistic Landscape
Black cohosh's triterpenes modulate serotonin 5-HT7 receptors, directly influencing thermoregulatory centers and reducing hot flashes by 40-60% in clinical trials. This serotonergic mechanism is supported by preclinical vascular and neuropharmacological studies and precisely aligns with the clinical pattern of symptom relief.
The 5-HT7 receptor concentrates in the hypothalamus, the brain's primary temperature control center. Actein and related triterpenes from black cohosh relax vascular smooth muscle and modulate receptor activity in animal and laboratory studies, providing a direct mechanism for reducing vasomotor instability [3]. These effects operate completely independently of estrogen signaling, confirmed by studies showing zero uterotrophic or estrogenic changes in animal models given standardized black cohosh [6].
Clinical trials using standardized extracts (20-40 mg/day) document significant reductions in both frequency and severity of hot flashes in postmenopausal women compared to placebo, with improvements beginning at 4-8 weeks [1]. While direct measurement of 5-HT7 receptor activity in humans remains challenging, the timing and pattern of symptom reduction closely parallels the known pharmacology of serotonin-modulating agents, providing strong indirect support for the mechanism. These effects occur without any shifts in serum estrogen, FSH, LH, or endometrial thickness, which remain unchanged in black cohosh studies [1,9].
The serotonin pathway provides a clear mechanistic link between black cohosh's triterpenes and its clinical vasomotor benefits, fundamentally distinguishing it from phytoestrogenic or hormonal approaches.
Lack of Estrogenic Effects: Evidence from Animal and Human Models
Black cohosh does not act as a phytoestrogen. Both animal and human studies show no estrogenic effects on reproductive tissues or hormone biomarkers, confirming that symptom relief occurs through non-hormonal mechanisms. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A pivotal rat study treated ovariectomized animals with isopropanolic black cohosh extract for 17 days, measuring uterine weight, histology, and vena cava contractility. No estrogen-like changes were observed, even at high extract doses [6]. In vitro receptor binding studies support these findings, showing negligible affinity for estrogen alpha or beta receptors. In human clinical studies, standardized extracts do not alter estradiol, FSH, LH, or endometrial thickness, further refuting the phytoestrogen hypothesis [1,9].
This evidence is crucial for safety. Unlike hormone therapy or true phytoestrogens, black cohosh does not stimulate endometrial or breast tissue and does not increase circulating estrogen. This makes it a candidate for those seeking non-hormonal symptom relief, including women who wish to avoid estrogenic risks. For those tracking serum hormone biomarkers, black cohosh supplementation is not expected to cause significant changes, consistent with both mechanistic and clinical data. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Bone and Vascular Effects: Triterpenes and Cellular Pathways
Black cohosh’s triterpene glycosides promote osteoblast function and vascular relaxation in cellular and animal models, but these benefits are mechanistic and not yet confirmed in human outcome studies. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Actein, a major triterpene, increases alkaline phosphatase activity, collagen synthesis, and glutathione content in osteoblast cultures—markers associated with bone formation and antioxidant defense [7]. Ethanolic black cohosh extracts have been shown to potentiate bone nodule formation in preosteoblast cells [5]. These cellular findings suggest a plausible pathway for supporting bone health, but human RCTs measuring bone mineral density or fracture outcomes are lacking.
On the vascular side, black cohosh extracts relax isolated rat aorta through both endothelium-dependent (nitric oxide) and -independent mechanisms, suggesting potential support for vascular tone [3]. Modulation of the plasminogen activator system in vascular smooth muscle cells could theoretically enhance fibrinolysis and vascular repair [4]. However, these effects have not translated into measured clinical benefits in blood pressure or cardiovascular events in human trials. Thus, while the mechanistic groundwork is strong, additional studies are needed to confirm these endpoints in humans. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Human Clinical Evidence: Outcomes, Safety, and Dosing
Standardized black cohosh extracts, at doses of 20–40 mg per day, reduce vasomotor symptoms in postmenopausal women according to randomized controlled trials. Safety data are favorable, with no significant changes in liver enzymes, hormone biomarkers, or endometrial thickness at clinically studied doses.
The largest and most recent RCT (n=96) found significant reductions in hot flash and night sweat frequency and severity with black cohosh versus placebo after 12 weeks [1]. Meta-analyses confirm these findings, especially for isopropanolic and ethanolic standardized extracts [9]. The benefits appear independent of baseline hormone levels or menopausal status, supporting a mechanism not reliant on estrogen modulation. Adverse events are rare and similar to placebo, with gastrointestinal symptoms being the most common. Laboratory monitoring in trials shows no consistent shifts in liver function, blood count, or hormonal markers, making black cohosh one of the better-tolerated botanicals for menopausal symptoms when used as directed [1,9].
For optimal use, most trials employ standardized extracts containing 2.5% triterpene glycosides, at a daily dose of 20–40 mg. There is no evidence that higher doses improve outcomes, and long-term safety beyond one year is less well established. Non-standardized products or root powders have not been tested in clinical trials and may not deliver effective or reproducible triterpene levels.
Conclusions
Black cohosh’s primary mechanism centers on triterpene glycosides modulating serotonin 5-HT7 receptors and inflammatory pathways, not on phytoestrogenic or hormonal activity. This distinct pathway explains its clinical efficacy for relieving hot flashes and night sweats, as demonstrated in recent randomized controlled trials using standardized extracts. The absence of estrogenic effects in both animal and human studies provides reassurance regarding reproductive safety and distinguishes black cohosh from both hormone therapy and true phytoestrogens. While anti-inflammatory and osteoblast-supportive actions are well supported in cellular models, human evidence for these broader endpoints remains indirect. Standardized extract forms—particularly isopropanolic or ethanolic, dosed at 20–40 mg/day—offer the best balance of efficacy and safety, with minimal side effects and no consistent changes in hormone-related biomarkers. Black cohosh represents a mechanistically distinct, evidence-backed option for non-hormonal relief of menopausal vasomotor symptoms. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured.
The main limitation is the gap between mechanistic plausibility and direct clinical evidence for endpoints beyond vasomotor symptom relief. While serotonin and inflammatory pathways are well established in preclinical studies, their quantitative contribution to clinical outcomes is still inferred rather than directly measured. Human trials have mostly focused on subjective symptom scales rather than objective biomarkers or longer-term endpoints like bone density or cardiovascular risk. Standardized extracts are well studied, but data on non-standardized formulations and long-term safety are limited. Further research is needed to clarify the clinical impact of black cohosh’s anti-inflammatory and bone-supportive mechanisms. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how black cohosh relates to your health goals and monitor changes in your biomarkers over time.
