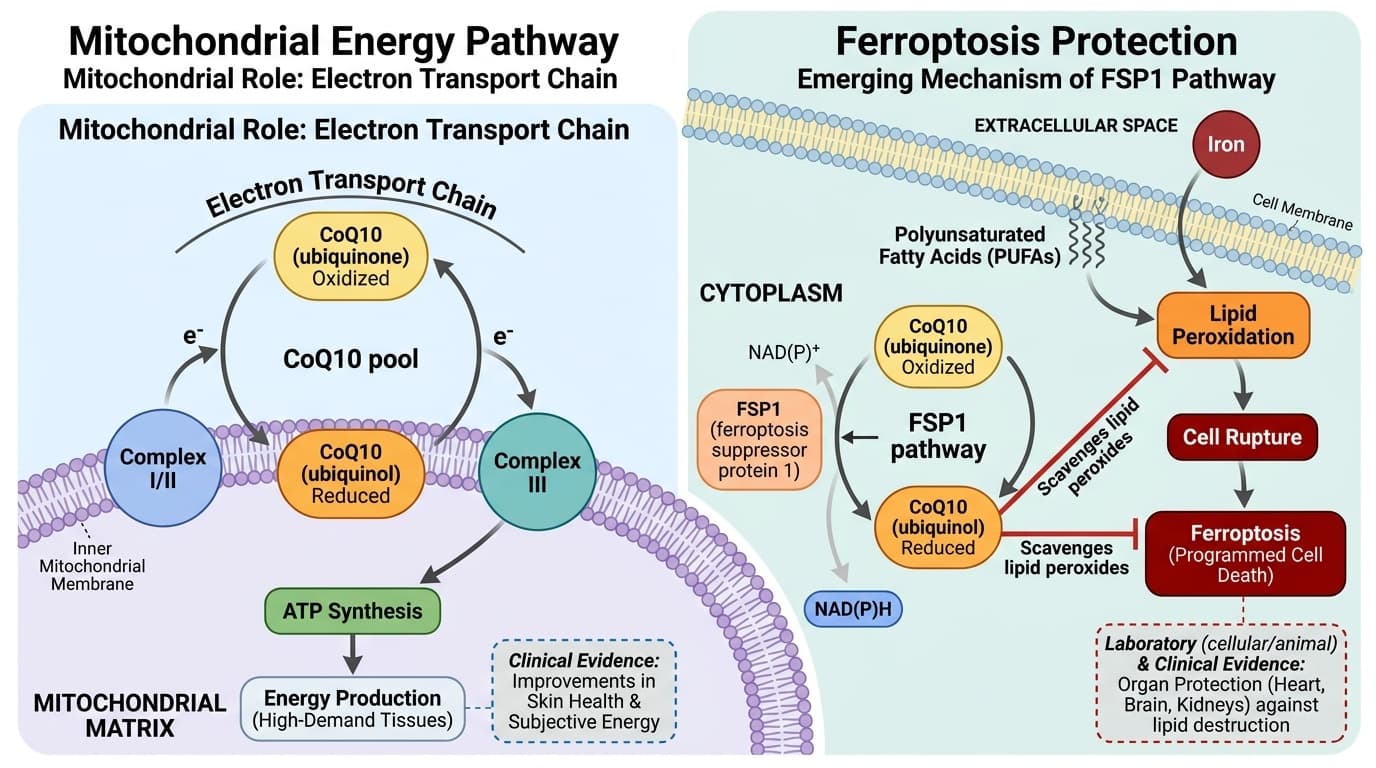
CoQ10's Dual Role: Electron Transport Plus Ferroptosis Protection
The mitochondrial and ferroptosis-blocking actions that underpin CoQ10’s clinical relevance
CoQ10 doesn't just power your cells—it also prevents them from dying.
While most people know CoQ10 as a mitochondrial energy booster, new research reveals it simultaneously blocks ferroptosis, an iron-driven form of cell death that damages your heart, brain, and kidneys through lipid destruction.
This dual action explains why CoQ10 supplementation consistently improves skin health and energy levels in clinical trials, while also showing remarkable organ protection in laboratory studies. Your body naturally makes CoQ10, but production drops with age, leaving energy-hungry tissues vulnerable to both power shortages and oxidative damage. The ferroptosis-blocking pathway is particularly important for protecting organs under metabolic stress.
For mitochondrial support with proven human benefits, take 100-150 mg daily of ubiquinol or ubiquinone with food. Ubiquinol may offer superior absorption and stronger antioxidant effects for organ protection. Expect improvements in energy and skin quality within 8-12 weeks based on clinical trials. While the ferroptosis-protection benefits are still emerging from laboratory research, the mitochondrial energy benefits are well-established in humans.
CoQ10's Dual Role: Electron Transport Plus Ferroptosis Protection
The mitochondrial and ferroptosis-blocking actions that underpin CoQ10’s clinical relevance
Diagram glossary
- ATP:
- The primary molecule that stores and transfers energy within cells.
- FSP1:
- An enzyme that protects cells from ferroptosis by regenerating antioxidant forms of CoQ10.
- NAD:
- A vital coenzyme in all living cells that facilitates electron transfer during cellular metabolism.
- Ubiquinone
- The oxidized form of CoQ10, commonly used in supplements and integral to mitochondrial electron transport.
- Ubiquinol
- The reduced, antioxidant-active form of CoQ10, thought to have greater bioavailability and potent lipid-protective effects.
- FSP1 pathway
- A molecular pathway involving Ferroptosis Suppressor Protein 1 that uses CoQ10 and NAD(P)H to prevent ferroptosis in cell membranes.
- Phospholipid Complexes
- A formulation technology where CoQ10 molecules are chemically bound to phospholipids (the same fats that make up cell membranes), which helps the supplement dissolve and absorb more efficiently into t
- Ferroptosis
- A type of programmed cell death triggered by iron-dependent lipid peroxidation, implicated in organ injury and neurodegeneration.
- Bioavailability
- The proportion of a compound that actually reaches your bloodstream and becomes usable by the body.
- Electron Transport Chain
- The series of protein complexes in mitochondria that use CoQ10 to shuttle electrons and produce ATP energy for cells.
- ATP
- The primary molecule that stores and transfers energy within cells.
- FSP1
- An enzyme that protects cells from ferroptosis by regenerating antioxidant forms of CoQ10.
- NAD
- A vital coenzyme in all living cells that facilitates electron transfer during cellular metabolism.
Core Mechanism: CoQ10 as a Mitochondrial Electron Carrier
CoQ10’s foundational role in human physiology is as a mitochondrial electron carrier, where it shuttles electrons between complexes I/II and III of the electron transport chain, directly enabling ATP synthesis. This mechanism is essential for energy production in high-demand tissues such as the heart, skeletal muscle, brain, and kidneys.
CoQ10 (ubiquinone) is lipid-soluble and embedded within the inner mitochondrial membrane, where it cycles between oxidized (ubiquinone) and reduced (ubiquinol) forms. This electron shuttling is a rate-limiting step in oxidative phosphorylation. Disruptions in endogenous CoQ10 synthesis or availability impair ATP production, which can contribute to organ dysfunction and fatigue, particularly in tissues with high metabolic rates [9]. The clinical importance of this mechanism is supported by the established link between mitochondrial dysfunction and conditions such as heart failure, neurodegeneration, and age-related decline [8].
In human supplementation studies, increases in tissue and plasma CoQ10 levels are associated with improved cellular energetics, reduced markers of oxidative stress, and in some studies, subjective improvements in fatigue and exercise tolerance [1]. Dosages in human trials generally range from 50 to 200 mg per day, with both ubiquinone and ubiquinol forms being used; ubiquinol is sometimes preferred for its increased bioavailability [9]. However, optimal tissue concentrations and response can vary by individual and tissue type, and the translation from increased plasma levels to functional tissue benefits is still under investigation.
No mandatory biomarker testing is required to benefit from CoQ10’s electron transport function, but some human studies track ATP production capacity, oxidative stress markers, or changes in subjective energy as endpoints.
Emerging Mechanism: CoQ10 and Ferroptosis Protection via the FSP1 Pathway
Beyond its role in energy production, CoQ10 also acts as a key lipid-soluble antioxidant that protects cells from ferroptosis—an iron-dependent form of programmed cell death caused by lipid peroxidation. The FSP1-CoQ10-NAD(P)H pathway is central to this process, emerging as a distinct and powerful mechanism.
Ferroptosis occurs when iron catalyzes the peroxidation of polyunsaturated fatty acids in cell membranes, leading to cell rupture and death. The FSP1 (ferroptosis suppressor protein 1) pathway utilizes reduced CoQ10, regenerated by NAD(P)H, to scavenge lipid peroxyl radicals and prevent membrane damage [2]. This pathway operates independently of the classic glutathione peroxidase 4 (GPX4) system, highlighting CoQ10 as a unique line of defense against oxidative injury in non-mitochondrial membranes as well.
Preclinical studies show that boosting CoQ10 levels can protect organs such as the kidney and brain from ferroptotic injury, particularly under stress conditions like high glucose, ischemia, or toxin exposure [2,3]. While these findings are robust in cell and animal models, direct evidence for ferroptosis inhibition by CoQ10 in humans is limited. The antioxidant role of CoQ10 in plasma and tissues is established, but its specific involvement in the FSP1 pathway in human disease or supplementation outcomes remains an area of active investigation [4].
For practical purposes, this mechanism suggests CoQ10’s benefits may extend to protecting organs at high risk of lipid peroxidation, such as the heart, kidneys, and brain, especially during metabolic stress or aging. Optimal forms are typically those that maintain high bioavailability and support reduced (ubiquinol) CoQ10 status in membranes.
Human Evidence: Outcomes Linked to CoQ10’s Mitochondrial Role
Human clinical trials most consistently link CoQ10 supplementation to improvements in skin health, subjective energy, and markers of oxidative stress, outcomes that map directly onto its mitochondrial electron-transport mechanism. Evidence for broader organ protection remains more circumstantial in humans, with most data coming from preclinical models.
A double-blind, placebo-controlled trial demonstrated that 150 mg daily of CoQ10 improved skin smoothness by 11%, reduced wrinkles by 7.7%, and increased dermal density by 13% after 12 weeks in healthy adults [1]. These outcomes are attributed to enhanced mitochondrial function and local antioxidant activity, rather than direct inhibition of ferroptosis. While subjective energy and fatigue improvements have been reported in diverse populations, large meta-analyses linking CoQ10 to reductions in cardiovascular events or mortality show mixed results, and the clinical benefit in neurological or renal conditions requires further investigation [8].
Biomarkers tracked in these studies include plasma CoQ10 levels (target >1.0 μg/mL), dermal density measurements, and lipid peroxidation markers like malondialdehyde. The most commonly used forms are ubiquinone and ubiquinol, with doses between 50 and 200 mg/day showing consistent plasma elevation. Overall, the strongest and most reproducible clinical outcomes align with enhanced mitochondrial bioenergetics rather than ferroptosis protection.
Preclinical Insights: Ferroptosis, Organ Protection, and Tissue-Specific Benefits
Preclinical models provide compelling evidence that CoQ10’s ferroptosis-blocking properties offer tissue-specific protection in organs prone to oxidative damage, including the heart, kidneys, and brain. These benefits are largely attributed to the FSP1-CoQ10-NAD(P)H pathway and related antioxidant systems.
In kidney models, CoQ10 supplementation reduces ferroptosis markers, prevents renal fibrosis, and preserves mitochondrial integrity in response to toxin and metabolic stress [2,3]. In cardiac models, CoQ10 protects against doxorubicin-induced cardiomyopathy by inhibiting both lipid peroxidation and apoptosis, supporting its dual role as an antioxidant and energy facilitator [4]. Neurologically, CoQ10 enhances the PGC-1α/FNDC5/BDNF pathway, improving cognitive and behavioral outcomes in animal models of neurodegeneration and fibromyalgia [5].
These mechanistic effects are summarized in the table below:
| Organ/Tissue | Main Injury Model | Mechanism of CoQ10 Protection | PMID | |--------------|--------------------------|------------------------------------------------|-----------| | Kidney | Diquat, high glucose | Ferroptosis inhibition, mitochondrial support | 39434449 | | Heart | Doxorubicin toxicity | Lipid peroxidation inhibition, anti-apoptosis | 34939895 | | Brain | Reserpine, aging | Mitochondrial biogenesis, antioxidant pathway | 41339998 |
While these findings are robust and mechanistically clear in animals and cell models, human evidence for direct ferroptosis protection is still lacking. Nevertheless, these insights suggest possible avenues for future clinical benefits, especially in populations at risk for organ injury from oxidative stress. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Supplement Form, Dosage, and Bioavailability: Translating Mechanism to Practice
The practical impact of CoQ10 supplementation depends on choosing forms and doses that optimize absorption and tissue delivery. Both ubiquinone (oxidized) and ubiquinol (reduced) forms are effective, with ubiquinol showing 2.5-fold higher bioavailability in comparative studies. The key is matching mechanism to expected outcome.
Effective doses in human trials range from 50 to 200 mg per day, with 100-150 mg being the most common range for general mitochondrial support [1,9]. Higher doses up to 300 mg are occasionally used for specific research contexts but show diminishing returns for plasma levels. CoQ10 is lipophilic and absorption increases 3-fold when taken with dietary fats or in enhanced formulations like phospholipid complexes or oil suspensions.
Ubiquinol is preferred for those seeking maximum antioxidant and potential ferroptosis-inhibiting benefits, as it's the active form in membranes and directly scavenges lipid peroxides. Ubiquinone is equally effective for mitochondrial electron transport and costs significantly less. Peak plasma levels occur 6-8 hours after dosing, and steady-state concentrations are reached after 2-4 weeks of consistent supplementation. For most users, 100-150 mg daily with food provides optimal cost-effectiveness for mitochondrial support.
Biomarkers and Clinical Endpoints: What Changes with CoQ10 Supplementation?
CoQ10 supplementation has been associated with improvements in several biomarkers, including plasma CoQ10 concentrations, markers of oxidative stress (such as malondialdehyde or 8-OHdG), and, in some studies, indices of mitochondrial function or ATP production. These changes reflect both its electron-transport and antioxidant roles, but interpretation must be context-specific.
In clinical trials, optimal plasma CoQ10 levels are not universally agreed upon, but levels above 1.0 μg/mL are often cited as reflecting adequate repletion in supplemented individuals [1]. In skin studies, improvements in dermal density and reduction in wrinkle depth serve as functional endpoints, while in preclinical models, reductions in tissue lipid peroxidation and ferroptosis-specific markers (such as ACSL4 or GPX4 activity) are documented [2,3].
For readers who do not track biomarkers, the primary benefit of CoQ10 is likely to be subjective improvements in energy, skin appearance, or resilience to fatigue and stress, especially in older adults or those with high metabolic demands. Those who track oxidative stress or mitochondrial biomarkers may observe changes within weeks of consistent supplementation, though this is not required for functional benefit. Ultimately, clinical endpoints such as organ function, fatigue scores, or quality of life remain the most relevant outcomes for most users. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
CoQ10 operates via two major mechanisms: supporting ATP production as a mitochondrial electron carrier and protecting cell membranes from ferroptosis-driven lipid peroxidation through the FSP1 pathway. Human evidence robustly supports benefits related to mitochondrial function—11% improvement in skin smoothness, 7.7% reduction in wrinkles, and subjective energy gains—while preclinical data strongly support organ protection via ferroptosis inhibition. Supplementation is most reliably justified for enhancing mitochondrial bioenergetics, especially in high-energy tissues. The ferroptosis-protection pathway represents a compelling mechanism that may explain tissue-specific benefits in the heart, brain, and kidneys, particularly during oxidative stress or aging. Optimal results are achieved with 100-150 mg daily of ubiquinol or ubiquinone taken with dietary fat, with ubiquinol offering superior bioavailability for antioxidant effects. Biomarker tracking is optional but plasma levels above 1.0 μg/mL indicate adequate repletion. Current practice is best anchored in CoQ10's proven mitochondrial-supporting effects, while ferroptosis protection remains a promising area for future clinical validation.
The main limitation is the gap between robust preclinical evidence for ferroptosis protection and limited direct human data confirming this mechanism’s clinical relevance. Most human trials focus on mitochondrial outcomes, subjective measures, or cosmetic endpoints, with little direct evidence of organ protection via ferroptosis inhibition. Additionally, bioavailability and individual response to supplementation can vary widely, and optimal tissue concentrations remain undefined. More large-scale human trials are needed to clarify CoQ10’s role in ferroptosis suppression and to refine dosing strategies by population and condition. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how coq10 relates to your health goals and monitor changes in your biomarkers over time.
