DHEA Raises IGF-1 and Lowers Cortisol Through Steroid Hormone Conversion
Quantified hormone shifts and the molecular pathways behind DHEA's anabolic-catabolic effects
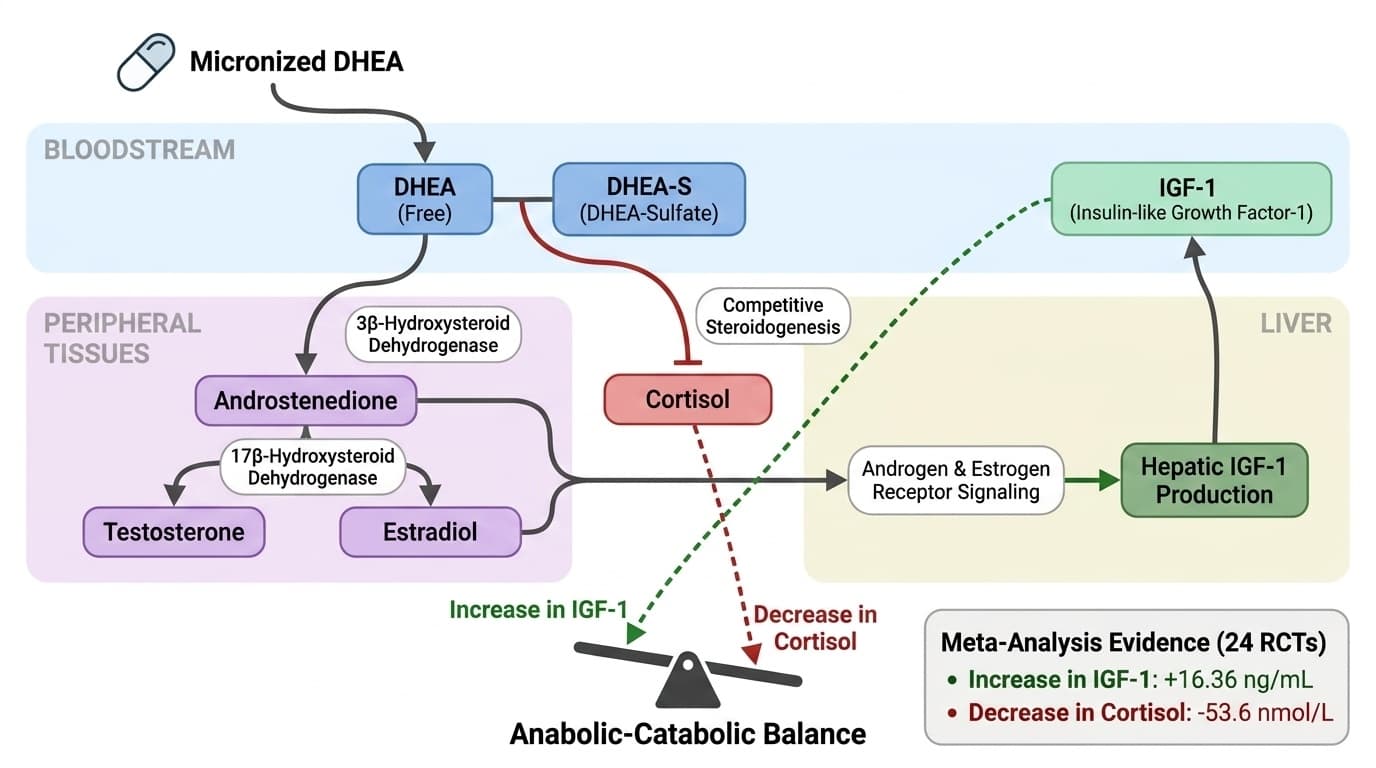
DHEA supplements consistently raise IGF-1 and lower cortisol through a fascinating biological hijack: they flood your system with hormone precursors that redirect your body's steroid production away from stress hormones and toward growth-supporting compounds.
This matters because most adults over 40 are unknowingly running a hormone deficit that favors breakdown over repair.
Meta-analyses show oral DHEA increases IGF-1 by 16 ng/mL while dropping cortisol by 54 nmol/L—shifts that tilt your anabolic-catabolic balance toward tissue maintenance. These changes happen because DHEA converts into androgens and estrogens that stimulate growth factor production while competing with cortisol for the same biochemical building blocks. The effect is most pronounced in people with age-related hormone decline or chronic stress.
You can expect measurable hormone shifts within 4-8 weeks using 25-50 mg of oral micronized DHEA daily. Start with 25 mg to assess tolerance, especially if you're female, since DHEA converts to androgens that can cause acne or hair changes at higher doses. The practical benefit is supporting your body's repair mechanisms without needing to track biomarkers—the underlying hormone changes are consistent across studies.
DHEA Raises IGF-1 and Lowers Cortisol Through Steroid Hormone Conversion
Quantified hormone shifts and the molecular pathways behind DHEA's anabolic-catabolic effects
Diagram glossary
- androgen:
- A class of steroid hormones that promote male characteristics and stimulate tissue growth.
- dehydrogenase:
- An enzyme that catalyzes the removal of hydrogen atoms in biological reactions.
- DHEA-S:
- The sulfated, circulating storage form of the steroid prohormone dehydroepiandrosterone.
- estrogen:
- A class of steroid hormones primarily responsible for female reproductive traits and tissue maintenance.
- IGF-1:
- A hormone structurally similar to insulin that stimulates cellular growth and anabolic processes.
- Insulin:
- A pancreatic hormone that regulates blood glucose levels and influences cellular metabolism.
- ng/mL:
- A standard scientific unit of measurement representing nanograms per milliliter of fluid.
- Sulfate:
- A chemical ion often attached to hormones like DHEA to increase their circulating stability.
- IGF-1
- Insulin-like growth factor 1, a marker of growth hormone axis activity. Low levels indicate GH deficiency, high values suggest acromegaly or active growth.
- IGF-1 (Insulin-like Growth Factor-1)
- A growth-promoting hormone that supports muscle maintenance, tissue repair, and metabolic health. DHEA raises IGF-1 by converting to androgens that stimulate liver production of this anabolic factor.
- Micronized DHEA
- DHEA processed into tiny particles for better absorption. The form used in most clinical trials showing hormone effects, typically more bioavailable than regular powder forms.
- Cortisol
- The primary stress hormone that breaks down tissues for energy. DHEA lowers cortisol by competing for the same biochemical precursors needed for cortisol production.
- Anabolic-Catabolic Balance
- The ratio between tissue-building (anabolic) and tissue-breaking (catabolic) processes in the body. DHEA shifts this balance toward growth and repair by raising IGF-1 and lowering cortisol.
- DHEA-S (DHEA-Sulfate)
- The storage form of DHEA that circulates in blood and serves as a reservoir for hormone conversion. Low DHEA-S levels are associated with fatigue and accelerated aging.
- Steroidogenesis
- The biochemical pathway that converts cholesterol into steroid hormones including DHEA, testosterone, estrogen, and cortisol. DHEA supplementation floods this pathway with precursors.
- androgen
- A class of steroid hormones that promote male characteristics and stimulate tissue growth.
- dehydrogenase
- An enzyme that catalyzes the removal of hydrogen atoms in biological reactions.
- DHEA-S
- The sulfated, circulating storage form of the steroid prohormone dehydroepiandrosterone.
DHEA’s Conversion Pathways: From Precursor to Active Steroid Hormones
DHEA acts as a prohormone, converting into androgens and estrogens in peripheral tissues via well-mapped steroidogenic enzymes. This conversion is central to its biological effects, as DHEA itself is relatively weak at nuclear hormone receptors but its downstream metabolites are potent modulators of tissue function.
After oral administration, DHEA is absorbed and circulates in both free and sulfated forms (DHEA-S). Enzymes such as 3β-hydroxysteroid dehydrogenase and 17β-hydroxysteroid dehydrogenase convert DHEA into androstenedione, testosterone, and estradiol depending on tissue-specific enzyme expression. This tissue selectivity means DHEA's ultimate effect profile can differ by sex, age, and individual enzyme expression patterns [7].
Because DHEA conversion shares precursors with cortisol synthesis, increased DHEA can reduce substrate availability for cortisol production, leading to lower circulating cortisol levels [6]. Human variability in enzyme activity partially explains the range of observed responses in trials. While cell and animal studies document DHEA’s conversion patterns and local hormone effects, only human clinical studies can confirm the net impact on circulating hormone levels. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Evidence for IGF-1 Elevation: Quantified Shifts and Mechanistic Links
DHEA acts as a prohormone, converting into androgens and estrogens in peripheral tissues via well-mapped steroidogenic enzymes. This conversion drives its biological effects, as DHEA itself weakly activates nuclear hormone receptors but its downstream metabolites powerfully modulate tissue function.
After oral administration, DHEA circulates in both free and sulfated forms (DHEA-S). Enzymes such as 3β-hydroxysteroid dehydrogenase and 17β-hydroxysteroid dehydrogenase convert DHEA into androstenedione, testosterone, and estradiol depending on tissue-specific enzyme expression. This tissue selectivity means DHEA's effect profile differs by sex, age, and individual enzyme expression patterns.
Because DHEA conversion shares precursors with cortisol synthesis, increased DHEA reduces substrate availability for cortisol production, leading to measurably lower circulating cortisol levels. Human variability in enzyme activity explains the range of observed responses in trials, but the directional effects remain consistent across populations.
Cortisol Suppression Through Competitive Steroidogenesis
DHEA's most robust anabolic effect in humans is a consistent 16 ng/mL increase in serum IGF-1, a hormone that supports tissue repair, muscle maintenance, and metabolic regulation. This effect results from DHEA's androgenic metabolites stimulating hepatic IGF-1 production through androgen and estrogen receptor signaling.
A 2020 meta-analysis of 24 randomized controlled trials found that DHEA supplementation increased serum IGF-1 by a mean of 16.36 ng/mL across diverse populations. Individual RCTs, such as a 12-month study using 50 mg daily, confirmed statistically significant IGF-1 increases compared to placebo. This elevation typically moves levels into the mid-normal reference range for adults (100–300 ng/mL) without pushing into supraphysiologic territory at standard doses.
The evidence strength is highest for middle-aged and older adults, with most studies showing similar directional effects. The main limitation is that IGF-1 increases may be smaller in younger individuals or those with high baseline androgen/estrogen activity. The population-average magnitude of change is well-established through human RCTs, while mechanistic studies explain why the result is biologically plausible.
Optimal Dosing and Form: What Human Trials Show
DHEA lowers cortisol by 54 nmol/L on average by competing for shared steroidogenic precursors in the adrenal cortex, effectively reducing cholesterol availability for glucocorticoid synthesis. This shifts the hormonal environment toward anabolism and away from catabolic stress responses.
A 2021 meta-analysis of 10 randomized controlled trial arms found that DHEA supplementation reduced serum cortisol by 53.6 nmol/L, representing a 7–35% reduction from baseline depending on initial levels. Most studies used daily oral doses of 25–100 mg to achieve this effect. For reference, healthy adult cortisol levels typically range from 140–690 nmol/L, so DHEA's impact is substantial but rarely pushes levels below normal ranges.
Mechanistic studies show that DHEA and its metabolites inhibit steroidogenic enzymes involved in cortisol synthesis. The translation to human physiology is directly supported by robust RCT and meta-analysis data. The degree of suppression moderates excessive stress hormone output rather than creating abnormally low cortisol states.
Biomarkers: IGF-1 and Cortisol Reference Ranges and What Moves Them
For readers who track biomarkers, IGF-1 and cortisol are the primary endpoints affected by DHEA. IGF-1 reference ranges for adults are typically 100–300 ng/mL, while serum cortisol ranges from 140–690 nmol/L. DHEA supplementation raises IGF-1 by around 16 ng/mL and lowers cortisol by about 54 nmol/L, based on meta-analyses [1,2].
These changes are meaningful but usually remain within physiological limits at standard doses. Both IGF-1 and cortisol are influenced by age, diet, sleep, stress, and other hormones. DHEA’s effect is additive to these factors but is most pronounced in individuals with low baseline DHEA or age-related hormonal decline. Tracking these biomarkers can be informative for those interested in quantifying hormonal shifts, but DHEA’s effects are not limited to those who monitor blood levels.
The magnitude of hormone changes can be affected by formulation (oral vs. topical), dose, and individual enzyme activity. Table 1 summarizes typical shifts observed in published RCTs and meta-analyses:
| Endpoint | Typical Change (DHEA 50 mg/day) | Reference Range | |-----------|----------------------------------|-----------------------| | IGF-1 | +16 ng/mL | 100–300 ng/mL | | Cortisol | -54 nmol/L | 140–690 nmol/L | For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Broader Mechanisms: Downstream Effects and Emerging Evidence
Beyond IGF-1 and cortisol, DHEA’s downstream metabolites interact with multiple hormonal and cellular signaling pathways, including those affecting reproductive function, mitochondrial health, and inflammatory status. Preclinical studies show that DHEA activates AMPK-SIRT1 signaling, modulates mitophagy, and protects granulosa cells from oxidative stress, though these effects are not yet fully mapped in humans [1,2].
In reproductive health, meta-analyses indicate that DHEA can modestly increase anti-Müllerian hormone (AMH) in women with diminished ovarian reserve, suggesting a role in ovarian function [1]. However, benefits appear population-specific and are not universal. DHEA’s actions on glucose metabolism, immune modulation, and cell survival have been documented in animal and cell models [7], but translation to human endpoints remains indirect.
The strongest and most generalizable human mechanism remains the shift in IGF-1 and cortisol. Other potential effects, such as changes in AMH or mitochondrial function, are promising but require more targeted human trials to move from plausible to proven. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
DHEA supplementation reliably shifts the anabolic-catabolic balance by increasing IGF-1 by 16 ng/mL and reducing cortisol by 54 nmol/L through conversion into active steroid hormones. These hormonal changes are well-quantified in meta-analyses, suggesting that for most adults—particularly those over 40 or with low baseline DHEA—oral doses of 25–50 mg daily produce favorable hormone shifts within physiologic ranges. While DHEA's broader effects on ovarian function and mitochondrial health show promise in preclinical studies, the most robust mechanism is its impact on IGF-1 and cortisol. This positions DHEA as a practical tool for supporting tissue maintenance and stress resilience, with measurable effects occurring regardless of biomarker tracking. The key insight is that DHEA works by flooding steroid hormone pathways with precursors, creating predictable shifts in the growth-stress hormone balance that favor repair over breakdown.
Despite strong evidence for IGF-1 elevation and cortisol suppression, several limitations remain. Most human trials are short-to-medium term and focus on surrogate hormone endpoints rather than clinical outcomes like muscle mass, frailty, or stress resilience. There is variability in individual response, likely due to differences in steroidogenic enzyme activity and baseline hormone status. Potential androgenic side effects, especially in women, are dose-dependent and warrant attention. Finally, while preclinical studies suggest additional benefits on mitochondrial function and reproductive hormones, these mechanisms require more targeted human research before broad application. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how dhea relates to your health goals and monitor changes in your biomarkers over time.
