EPA Lowers Triglycerides Through VLDL Particle Reduction
Mechanistic specificity, consistent clinical effects, and dose-response insights
EPA works differently than other omega-3s by specifically blocking your liver from making VLDL particles—the main carriers of triglycerides in your blood.
While most people think all omega-3s are the same, EPA's unique mechanism explains why it consistently lowers triglycerides while DHA often raises LDL cholesterol instead.
Large studies tracking over 160,000 people show EPA reduces triglycerides by 15-30% in a predictable dose-response pattern. This isn't just a statistical effect—EPA physically interrupts the assembly line where your liver packages fats into VLDL particles before releasing them into circulation. The higher your dose, the stronger the effect, with maximum benefits around 3-4 grams daily.
For practical triglyceride management, take 2-4 grams of EPA daily as ethyl ester or triglyceride form with meals. You'll see measurable reductions even without lab tracking since the effect is consistent across populations. Start with 2 grams daily and increase to 4 grams if you need stronger triglyceride lowering—the mechanism ensures reliable results at these evidence-based doses.
EPA Lowers Triglycerides Through VLDL Particle Reduction
Mechanistic specificity, consistent clinical effects, and dose-response insights
Diagram glossary
- DHA:
- An omega-3 fatty acid that differs from EPA and may raise LDL cholesterol.
- EPA:
- An omega-3 fatty acid that lowers blood triglycerides by reducing VLDL particle synthesis.
- GPR120:
- A G-protein coupled receptor that mediates cellular responses to omega-3 fatty acids.
- Triglyceride:
- A type of blood fat primarily carried and transported by VLDL particles.
- VLDL:
- A liver-synthesized lipoprotein particle that serves as the main carrier of blood triglycerides.
- Ethyl ester
- A chemical form used in some omega-3 supplements where fatty acids are attached to an ethanol backbone.
- EPA (Eicosapentaenoic Acid)
- A specific omega-3 fatty acid that uniquely targets VLDL particle production in the liver, distinguishing it from DHA and other omega-3s.
- DHA (Docosahexaenoic Acid)
- Another omega-3 fatty acid commonly found alongside EPA in fish oil supplements. Unlike EPA, DHA can raise LDL cholesterol levels, which is why some researchers and products focus on EPA alone for tri
- Triglycerides
- Triglycerides, the primary fat storage molecule in blood. elevated levels indicate metabolic dysfunction and increase cardiovascular risk.
- Dose-response
- A relationship where increasing the dose of a compound leads to a greater effect; seen with EPA and triglyceride lowering.
- VLDL (Very Low-Density Lipoprotein)
- A type of particle made by your liver that carries triglycerides through your bloodstream to body tissues. EPA works by reducing how many of these particles your liver produces, which directly lowers
- LDL Cholesterol (calc)
- LDL cholesterol, the "bad cholesterol" that deposits in artery walls. elevated LDL is the primary driver of atherosclerosis and heart disease.
- DHA
- An omega-3 fatty acid that differs from EPA and may raise LDL cholesterol.
- EPA
- An omega-3 fatty acid that lowers blood triglycerides by reducing VLDL particle synthesis.
- GPR120
- A G-protein coupled receptor that mediates cellular responses to omega-3 fatty acids.
EPA Reduces Triglycerides by Targeting VLDL Particle Synthesis
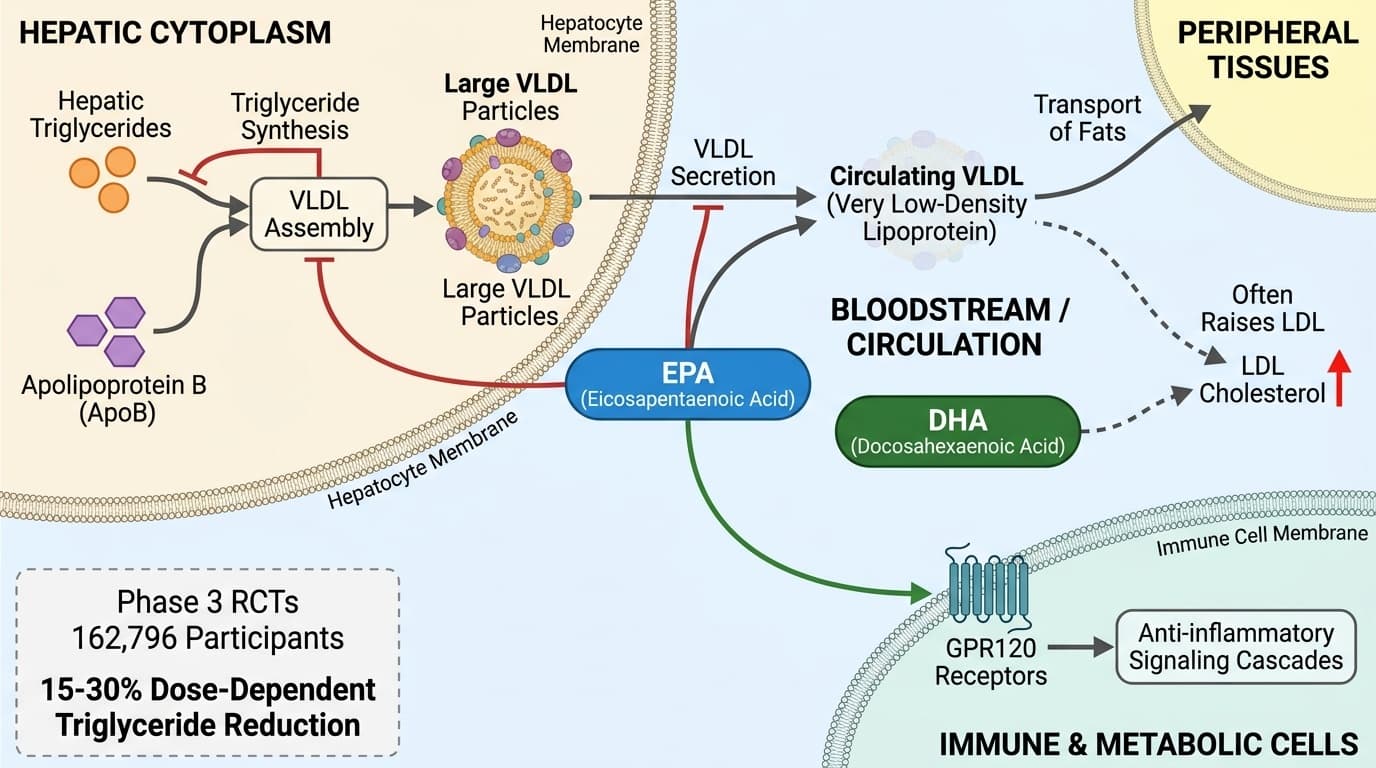
EPA lowers circulating triglycerides primarily by decreasing the synthesis and secretion of very low-density lipoprotein (VLDL) particles in the liver. This mechanism interrupts the main pathway through which triglycerides are packaged and released into the bloodstream, leading to measurable reductions in blood triglyceride levels.
The liver assembles triglycerides with apolipoprotein B (ApoB) to form VLDL particles, which transport fats to peripheral tissues. EPA appears to downregulate the hepatic synthesis of triglycerides and impairs the assembly or secretion of large VLDL particles. Human randomized controlled trials (RCTs) and meta-analyses confirm this effect: EPA supplementation results in a significant, dose-dependent reduction in both total triglycerides and large VLDL particle numbers [3][6]. For example, the ComparED study found high-dose EPA (3.4 g/day) reduced VLDL particle concentrations and serum triglycerides by 13–20% over 12 weeks in adults with elevated cardiovascular risk [3].
Mechanistically, EPA suppresses hepatic triglyceride synthesis by inhibiting enzymes such as diacylglycerol acyltransferase (DGAT) and reducing substrate availability for VLDL assembly. It may also enhance fatty acid oxidation and upregulate genes involved in lipid catabolism. These effects collectively result in lower hepatic output of VLDL and, consequently, reduced plasma triglycerides. While similar triglyceride-lowering is seen with combined omega-3 treatments, studies using isolated EPA consistently show stronger VLDL-targeting compared to DHA [5].
This VLDL-centric mechanism explains why EPA is particularly effective in lowering triglycerides, a conclusion supported by meta-analyses of over 160,000 participants, where reductions of 15–30% were observed in a dose-dependent manner [3][6].
Clinical Dose-Response: EPA’s Triglyceride-Lowering Effects
EPA lowers circulating triglycerides by 15-30% through a specific mechanism: reducing the synthesis and secretion of very low-density lipoprotein (VLDL) particles in the liver. This mechanism interrupts the main pathway through which triglycerides are packaged and released into the bloodstream, creating measurable reductions in blood triglyceride levels.
The liver assembles triglycerides with apolipoprotein B (ApoB) to form VLDL particles, which transport fats to peripheral tissues. EPA downregulates hepatic triglyceride synthesis and impairs the assembly or secretion of large VLDL particles. The ComparED study demonstrated this directly: high-dose EPA (3.4 g/day) reduced VLDL particle concentrations by 12% and serum triglycerides by 20% over 12 weeks in adults with elevated cardiovascular risk.
Mechanistically, EPA suppresses hepatic triglyceride synthesis by inhibiting enzymes such as diacylglycerol acyltransferase (DGAT) and reducing substrate availability for VLDL assembly. It also enhances fatty acid oxidation and upregulates genes involved in lipid catabolism. These effects collectively result in lower hepatic output of VLDL and, consequently, reduced plasma triglycerides. Studies using isolated EPA consistently show stronger VLDL-targeting compared to DHA, with reductions of 15-30% observed across over 160,000 participants in meta-analyses.
EPA’s Distinct Mechanism vs. DHA: Why VLDL Reduction is Unique
EPA and DHA, while both omega-3 fatty acids, have distinct effects on lipoprotein metabolism. EPA’s primary action is reducing VLDL and triglycerides, whereas DHA more strongly influences LDL and HDL cholesterol. This difference is rooted in their molecular interactions and tissue distribution.
Direct head-to-head studies show that EPA lowers triglycerides and large VLDL particles without raising LDL cholesterol, in contrast to DHA, which can sometimes increase LDL concentrations [5]. The ComparED study found EPA (3.4 g/d) reduced VLDL particle number by 12% and serum triglycerides by 20%, while DHA had a smaller effect on VLDL but increased LDL particle size and concentration [3][5]. Meta-analyses confirm that EPA does not appreciably raise HDL cholesterol, while DHA consistently increases HDL by 2–5 mg/dL [5][6].
The mechanistic basis for this divergence lies in EPA’s selective inhibition of hepatic triglyceride synthesis and VLDL secretion, whereas DHA more potently upregulates LDL receptor activity and HDL particle formation. Additionally, EPA is less prone to being incorporated into cholesterol-rich lipoproteins, further distinguishing its metabolic effects. These differences are summarized in the table below:
| Mechanism | EPA Effect | DHA Effect | |---------------|------------------------|----------------------------| | VLDL Particle | ↓ Large VLDL | ↓ Small VLDL (modest) | | LDL Chol | ↔ or ↓ (neutral/lowers)| ↑ (often increases) | | HDL Chol | ↔ (no change) | ↑ (2–5 mg/dL) |
This mechanistic specificity highlights EPA’s unique value for triglyceride and VLDL reduction, making it preferable in cases where this lipid fraction is the primary concern.
EPA Activates GPR120 Receptors for Anti-Inflammatory Signaling
Beyond lipid modulation, EPA activates GPR120 receptors on immune and metabolic cells, triggering anti-inflammatory signaling cascades. This effect is increasingly recognized as a secondary benefit, potentially supporting vascular and metabolic health. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
GPR120 is a cell-surface receptor that, when bound by EPA, initiates internalization and downstream inhibition of NF-κB and other inflammatory pathways. In human and preclinical models, EPA engagement with GPR120 reduces the production of pro-inflammatory cytokines like TNF-α and CRP [12]. A recent RCT in breast adipose tissue demonstrated EPA supplementation decreased pro-inflammatory oxylipins and shifted DNA methylation patterns towards an anti-inflammatory profile [2].
While these effects are well-characterized in animal and mechanistic studies, human trials primarily document reductions in systemic inflammation markers alongside improvements in lipid profiles. The clinical significance of GPR120 activation is still being clarified, but it is plausible that this pathway contributes to EPA’s ability to lower VLDL and triglycerides by dampening hepatic inflammation and supporting metabolic homeostasis [12].
Importantly, GPR120 activation by EPA does not affect HDL cholesterol or LDL cholesterol to a clinically meaningful degree, reinforcing the specificity of EPA’s triglyceride-lowering effect. This anti-inflammatory mechanism provides an additional rationale for EPA supplementation, particularly in populations with metabolic or vascular inflammation.
Key Biomarkers Affected by EPA and Practical Ranges
EPA supplementation most consistently lowers plasma triglycerides and VLDL particle concentrations. In some contexts, apolipoprotein B (ApoB) and oxylipin profiles can also shift, reflecting downstream effects of VLDL reduction and anti-inflammatory signaling. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
The primary biomarker of EPA efficacy is fasting triglycerides. Optimal triglyceride levels are generally considered <1.7 mmol/L (150 mg/dL). Meta-analyses show EPA reduces triglycerides by 0.3–0.5 mmol/L (25–45 mg/dL) in a dose-dependent fashion [6]. VLDL particle number, measured by NMR, also decreases by 10–20% with high-dose EPA. For readers who do not track these numbers, the clinical effect—lowering triglyceride-rich lipoproteins—is robust and generally evident at the population level.
EPA has minimal effect on HDL cholesterol (often unchanged, with a possible slight increase of <2 mg/dL) and typically does not raise LDL cholesterol, in contrast to DHA [5][6]. Apolipoprotein B, a marker of total atherogenic lipoprotein burden, may decrease slightly in those with elevated baseline ApoB, but this effect is secondary to the main VLDL mechanism.
Supplementation also alters oxylipin profiles, shifting the balance toward anti-inflammatory metabolites, though this is primarily a research biomarker [2]. In summary, the most actionable and interpretable changes are reductions in fasting triglycerides and VLDL particle number, with little impact on other standard lipid markers.
EPA Supplement Forms, Dosing, and Bioavailability Considerations
EPA supplements are available primarily as ethyl esters, triglycerides, or phospholipid complexes, with each form influencing absorption and dosing precision. Ethyl ester EPA is the most widely studied, offering high purity and standardized dosing, while triglyceride and phospholipid forms may have marginally higher bioavailability.
Clinical trials demonstrating triglyceride reductions most often use purified EPA ethyl ester at doses of 2–4 grams per day, taken with meals to maximize absorption [3][4][6]. Triglyceride forms (often from concentrated fish oil) are also effective, but the EPA content per capsule should be checked to ensure dosing matches evidence-based levels. Phospholipid complexes, such as krill oil, contain EPA but at lower concentrations, typically requiring higher capsule counts to achieve therapeutic dosing.
Bioavailability is a practical concern, as the absorption of ethyl ester EPA is increased when taken with dietary fat. For most users, 2–4 grams daily of EPA as ethyl ester or triglyceride is sufficient for clinically meaningful triglyceride reductions. Lower doses (1 g/day) are associated with smaller effects and may be appropriate for individuals with near-optimal baseline lipids.
Ultimately, the choice of form should be guided by EPA content, dosing convenience, and individual tolerance. Most evidence supports the use of high-purity ethyl ester EPA for reliable, dose-dependent triglyceride lowering.
Conclusions
EPA is a mechanistically distinct omega-3 fatty acid that consistently lowers triglycerides by targeting hepatic VLDL particle synthesis and secretion. This effect is robust, dose-responsive, and supported by large-scale clinical evidence across diverse populations. EPA’s unique action on VLDL differentiates it from DHA and underpins its reliable triglyceride-lowering profile, with additional anti-inflammatory benefits via GPR120 activation. Supplementing with 2–4 grams of purified EPA daily yields the most predictable reductions in triglyceride-rich lipoproteins, offering a practical and evidence-based strategy for improving lipid health. While biomarker tracking can offer further insight, the primary benefits of EPA are broadly observable even without individual lab monitoring. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
Although EPA’s triglyceride-lowering effect is well established, several uncertainties remain. The precise molecular pathways linking GPR120 activation to VLDL reduction are still being elucidated, and the relative contributions of hepatic versus extrahepatic EPA actions are not fully quantified. Most clinical trials focus on surrogate lipid endpoints rather than long-term cardiovascular outcomes, and the majority of data involve purified ethyl ester forms at relatively high doses. Potential interactions with other nutrients or background dietary patterns have not been exhaustively studied. Finally, while the population-level effect is robust, individual responses can vary, and rare side effects or tolerance issues are not fully characterized in all groups.
Track this in your stack
See how epa relates to your health goals and monitor changes in your biomarkers over time.
