Epigenetic Clocks: What Your Biological Age Test Really Means (and What It Doesn’t)
A Deep Dive into DNA Methylation, Aging Rate, and What Moves the Clock
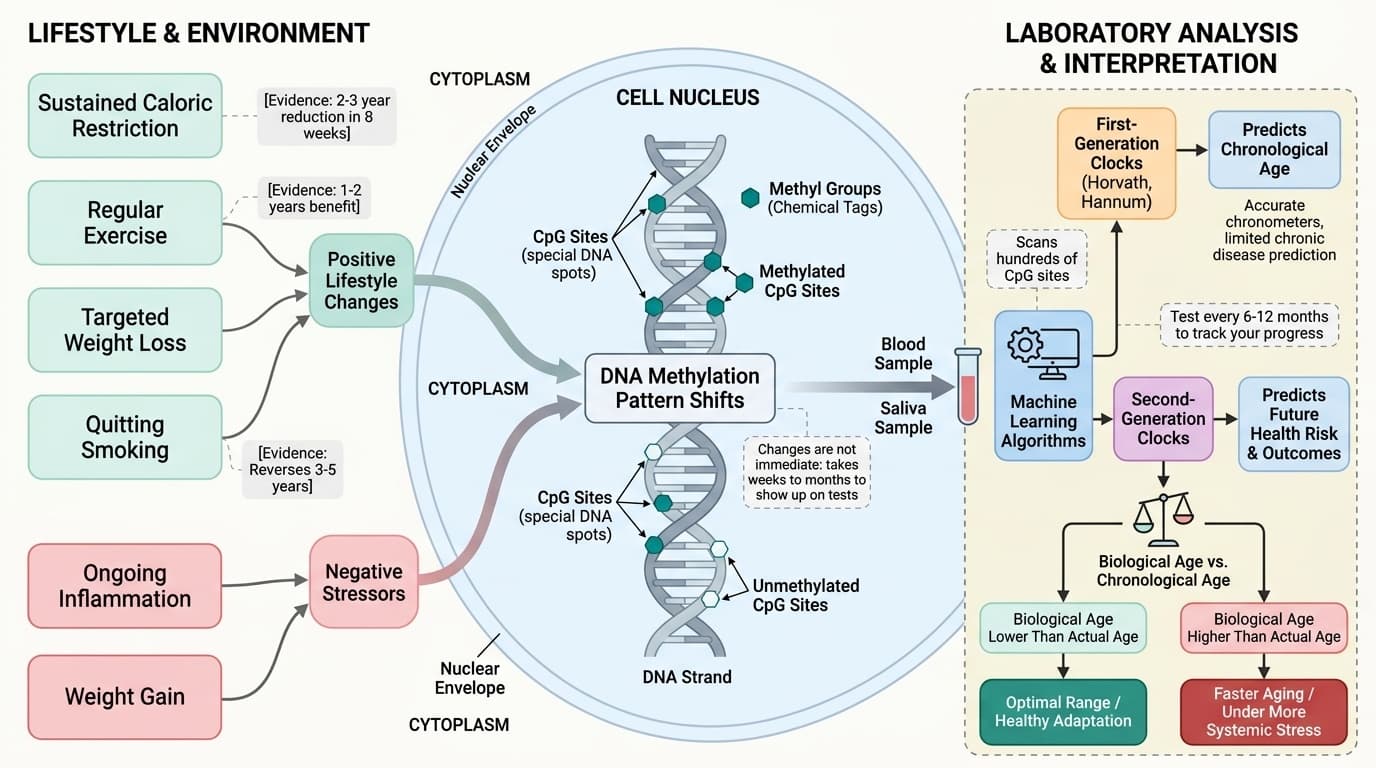
You might be surprised to learn that your biological age can shift by 2-3 years in just eight weeks with the right approach.
Most people think these DNA tests give them a fixed aging score. But your biological age actually tracks how your body responds to lifestyle changes in real time.
This means you can actively lower your biological age through proven interventions. The most effective approach combines sustained caloric restriction (eating 20-25% fewer calories while maintaining nutrition), regular exercise, and targeted weight loss. Some people add supplements, but the lifestyle factors do most of the heavy lifting.
The strongest evidence comes from caloric restriction studies showing 2-3 year reductions in biological age over 8 weeks. Exercise adds another 1-2 years of benefit when moving from sedentary to active. Quitting smoking can reverse 3-5 years of biological aging. Test every 6-12 months to track your progress, not just once for a snapshot.
Epigenetic Clocks: What Your Biological Age Test Really Means (and What It Doesn’t)
A Deep Dive into DNA Methylation, Aging Rate, and What Moves the Clock
Diagram glossary
- disease:
- An abnormal condition that negatively affects the structure or function of an organism.
- DNA:
- The molecule that carries genetic information for the development and functioning of an organism.
- DunedinPACE
- A clock that measures how fast you are aging right now, like a speedometer for your body.
- DNA Methylation
- A process where small molecules called methyl groups attach to DNA, affecting how genes work.
- Biological Age
- An estimate of how old your body seems based on biological markers, not just your birthday.
- Epigenetic Clocks
- Tests that measure patterns in DNA methylation to estimate your biological age or aging rate.
- Caloric Restriction
- Eating 20-25% fewer calories than usual while still getting enough nutrients to reverse biological aging.
- Weight
- Body weight in kilograms, most basic anthropometric measure.
- TRIIM Trial
- A study that tested whether a combination of growth hormone, metformin, and DHEA could reverse biological aging.
- DNA
- The molecule that carries genetic information for the development and functioning of an organism.
You Get Your Biological Age—But What Does That Number Mean?
Imagine you spit in a tube, mail it to a lab, and wait a few anxious weeks for the results. The email finally arrives: "Your biological age is 47. Your chronological age is 55. You are aging eight years younger than expected." This can feel like a personal victory or a warning. But what does it actually mean for your health, longevity, or athletic goals?
Epigenetic age tests are now everywhere—popularized by companies like TruAge and GlycanAge—and promise to give you a clearer picture of how well you are aging. These numbers are tempting to treat as hard truth, but the science behind them is more complex. Your biological age may not capture everything about your health or future risk, and the number itself can shift for reasons that are not always obvious.
Understanding what these tests measure, and what moves your score, can help you interpret your results with more confidence. In this guide, you will learn how the most common epigenetic clocks work, what actually shifts your biological age, and how to use these tools to guide your next steps—without falling for misleading promises.
How Do Epigenetic Clocks Work? The Basics You Need to Know
Epigenetic clocks rely on DNA methylation—tiny chemical tags that attach to specific spots on your DNA. Over time, certain sites gain more tags, while others lose them. These changes happen in patterns so reliable that, as shown in the original Horvath clock, algorithms can predict your age within three to four years just from a small blood sample [1].
The key idea is that your DNA itself does not change, but the chemical marks on it do, reflecting both your genetics and your life experiences. Researchers use machine learning to scan methylation at hundreds of CpG sites (special spots on the DNA) and find patterns that match up with age or health outcomes [1][2].
Standard ranges for these clocks are not as clear-cut as for, say, cholesterol. Your "biological age" is usually compared to average clock readings for people of your chronological age. An optimal range is one where your biological age matches or is lower than your actual age. If your result is higher, it may mean your body is aging faster, or is under more stress. If it is lower, it could reflect healthy adaptation—or simply test variability.
What moves these methylation patterns? Both exposure to damage and positive lifestyle changes. For example, quitting smoking can shift your clock to a lower age, while ongoing inflammation or weight gain may accelerate it. But changes are not immediate: shifts in biological age often take weeks to months to show up on tests, and may reflect both short-term adaptation and long-term risk. This sets the stage for understanding the strengths and limitations of each clock generation.
First-Generation Clocks: Predicting Age, Not Risk
The first wave of epigenetic clocks—like the Horvath and Hannum clocks—were trained to predict your actual age from methylation patterns [1][2]. They are extremely accurate chronometers: if your test says you are 52 and you are actually 53, the clock is working as designed. But this does not necessarily mean it tells you about your health, risk of disease, or how fast you are actually aging.
These first-generation clocks are helpful for identifying people whose methylation patterns are much older or younger than their chronological age. For example, if you are 40 and the clock says 55, it might prompt you to look closer at your lifestyle or health. The standard range is typically a match between clock age and actual age, while optimal would be a clock age lower than your actual age.
However, these clocks have limited power to predict who will develop chronic diseases or die early. This is because they focus on patterns that reflect age itself, not on what causes harm. Studies have found that their readings can be influenced by both stable and transient factors, and that large changes may only show up over years, not weeks or months [1][2].
If your goal is athletic performance, these clocks may not capture the impact of intense training or adaptation. For longevity, you want a tool that links more closely to future health outcomes. This need led to the development of second-generation clocks.
Second-Generation Clocks: Predicting Health, Not Just Age
Second-generation epigenetic clocks, like PhenoAge and GrimAge, take a different approach. Instead of trying to guess your chronological age, these clocks are trained to predict your risk of disease and death by looking at methylation patterns linked to mortality and morbidity [3][4].
PhenoAge, for example, is based on methylation sites that best predict who will develop chronic diseases or die in the next few years [3]. GrimAge goes even further, using methylation to estimate things like lifetime smoking exposure and levels of blood proteins that predict future risk [4]. This makes these clocks better at flagging early warning signs of poor health, especially for those with higher baseline risks, such as older adults or those with metabolic syndrome.
Standard and optimal ranges for these clocks are still being defined, but generally, a clock age lower than your real age suggests a lower risk. If you see a higher biological age, especially on these clocks, it may be an early sign to reevaluate health habits or underlying conditions. The timeline to see changes is similar—weeks to months—but these clocks respond to a broader set of interventions, including weight loss and smoking cessation.
The challenge is interpretation. Because these clocks pull in methylation signals from many pathways, your "score" can be affected by both positive adaptation and negative stress. This complexity means you need to look for trends over time rather than rely on a single reading. Next, we will dig into why this distinction matters—and why not all methylation changes are created equal.
Damage or Adaptation? The Double-Edged Sword of Methylation Changes
Here’s the core problem with interpreting epigenetic clocks: methylation changes can signal either accumulating damage or healthy adaptation. Damage comes from things like oxidative stress, DNA repair failures, inflammation, and telomere shortening. These show up as methylation patterns that are linked to aging and disease [3][4].
But your body also changes methylation on purpose, as part of regulated adaptation to challenges. For example, exercise triggers methylation shifts that can make your clock look "younger"—but is that because you reversed damage, or because your cells are switching on healthy stress responses? Caloric restriction, fasting, and certain supplements can all trigger adaptive methylation patterns too.
This means your biological age reading is a blend of both damage and adaptation. A drop in your clock age might reflect healing, but could also reflect your body turning on protective programs in response to new habits. The key is that the test cannot distinguish between the two. This is why a single measurement—especially after a new intervention—should not be over-interpreted.
For those with athletic or longevity goals, this ambiguity matters. If you see a big drop after starting a new training regimen or diet, celebrate the adaptation, but recognize that the true benefit will show up as a sustained downward trend over several tests. Next, we look at a new kind of clock that aims to separate these effects: DunedinPACE.
DunedinPACE: The Speedometer for Your Aging Rate
Most epigenetic clocks are like an odometer—they estimate your total biological mileage. DunedinPACE, developed in 2022, is different: it acts like a speedometer, measuring how fast you are aging right now [5].
This clock is based on data from a long-term study that tracked people’s health over decades, linking methylation patterns to the rate of physical and cognitive decline. Instead of giving you a "biological age," DunedinPACE tells you your current pace of aging compared to the average. A value of 1.0 means you are aging at the expected rate for your age group. Below 1.0 means you are aging more slowly; above 1.0 means faster aging [5].
Standard and optimal ranges are still being refined, but for most adults, aiming for a DunedinPACE below 1.0 is considered beneficial. The clock is sensitive to recent lifestyle changes, showing shifts within months, and is less affected by long-term damage than by real-time adaptation. This makes it especially useful for tracking interventions in athletes or people making rapid lifestyle changes.
DunedinPACE also interacts with other markers: improvements in sleep, stress, diet, and exercise all tend to slow the pace of aging, which then shows up as lower scores. However, like all clocks, it is influenced by both genetics and environment, so trends matter more than single readings. Next, we’ll see what actually moves these clocks—and what interventions have the best evidence.
What Actually Moves the Clock? Caloric Restriction, Exercise, and Supplements
With so many options, you might wonder what actually shifts your biological age. Here is what the evidence shows with specific numbers:
Caloric restriction delivers the biggest impact. The Fitzgerald 2021 trial found that eating 20-25% fewer calories while maintaining nutrition lowered biological age by 3.23 years over just 8 weeks [7]. Multiple studies confirm reductions of 2-3 years within 2-8 months of sustained caloric restriction.
Exercise adds significant benefit. Moving from sedentary to active lifestyles shifts methylation patterns equivalent to 1-2 years of biological age reduction. The effect is strongest in those starting from low fitness levels.
Weight loss amplifies other interventions. Studies show that losing 10-15% of body weight enhances the biological age benefits of both caloric restriction and exercise, especially when metabolic health improves alongside weight loss.
Smoking cessation reverses 3-5 years of biological aging. GrimAge is particularly sensitive to smoking history, and quitting shows measurable improvements within 6-12 months [4].
Supplementation shows promise but needs more research. The TRIIM trial tested growth hormone (0.25 mg daily), metformin (500 mg twice daily), and DHEA (25-50 mg daily), finding a 2.5-year reversal of epigenetic age after one year [6]. However, this was a small study of 9 men and has not been replicated.
For all interventions, consistency matters more than intensity. Sustained changes over months show stronger clock effects than short-term dramatic shifts. Combining caloric restriction with exercise appears synergistic, but no single supplement matches lifestyle interventions.
How to Use Your Biological Age Test: Practical Guidance
If you decide to get your biological age tested, keep in mind the practical realities. Consumer tests cost between $200 and $500 and can have a test-retest variability of one to three years. This means a single test is less reliable than tracking your trend over time.
Early warning signs to watch for include a biological age that is consistently higher than your chronological age, or a DunedinPACE score above 1.0. For athletes, a rising biological age may signal overtraining or inadequate recovery. For older adults, it may reflect increased inflammation or metabolic stress. Demographic-specific context matters: women may see bigger swings in methylation patterns during menopause, and those with chronic conditions should interpret results alongside other biomarkers.
To get the most out of your test, repeat it every six to twelve months, and focus on sustainable interventions. Caloric restriction and regular exercise have the strongest evidence. Quitting smoking, managing sleep, and reducing chronic stress are also key. Use supplement interventions only with medical guidance, as the evidence is still emerging.
Always combine your biological age results with other health markers—like blood pressure, cholesterol, and fitness testing—to get a full picture. The next section will tie together what you’ve learned and offer a clear bottom line.
The Bottom Line: What Epigenetic Clocks Can and Can’t Tell You
Epigenetic clocks are fascinating tools that offer a new window into your biological aging. They measure real changes in your DNA, but interpreting those changes requires nuance. A younger biological age does not always mean less damage—it can also mean more adaptation to healthy stress.
The best use of these tests is as a long-term trend tracker, not a one-time verdict. For both athletic and longevity goals, the interventions that reliably move the clock—caloric restriction, exercise, weight loss, and smoking cessation—are the same habits that improve all other health markers.
If you get a biological age test, focus on sustainable lifestyle changes and track your progress over time. Be skeptical of dramatic claims from any single supplement or intervention. Use your results as one piece of the puzzle, not the final answer. This approach will help you get the most value from your investment in biological age testing, and keep you moving toward your health goals.
Conclusions
Epigenetic clocks are powerful but imperfect tools for assessing biological aging. They capture both damage and adaptation signals, making long-term trends more meaningful than single test results. Caloric restriction (20-25% reduction) delivers the strongest evidence for reversing biological age by 2-3 years in 8 weeks. Regular exercise, weight loss, and smoking cessation provide additional benefits of 1-5 years each. Use these clocks to track your response to proven interventions, not as definitive health predictors.
Evidence for supplement- and drug-based interventions is still preliminary, with most studies small and not yet replicated. Test-retest variability can make single measurements misleading. Epigenetic clocks cannot distinguish between damage and adaptation, complicating interpretation. Demographic differences—such as sex, age, and health status—affect clock readings and optimal ranges. No epigenetic clock is a substitute for comprehensive health assessment.
Track this in your stack
See how epigenetic clocks relates to your health goals and monitor changes in your biomarkers over time.
