GABA's Sleep Architecture Effect: Slow-Wave Enhancement Via GABAA Receptors
How GABA Supplementation Alters Sleep Stages and Efficiency Through Receptor Mechanisms
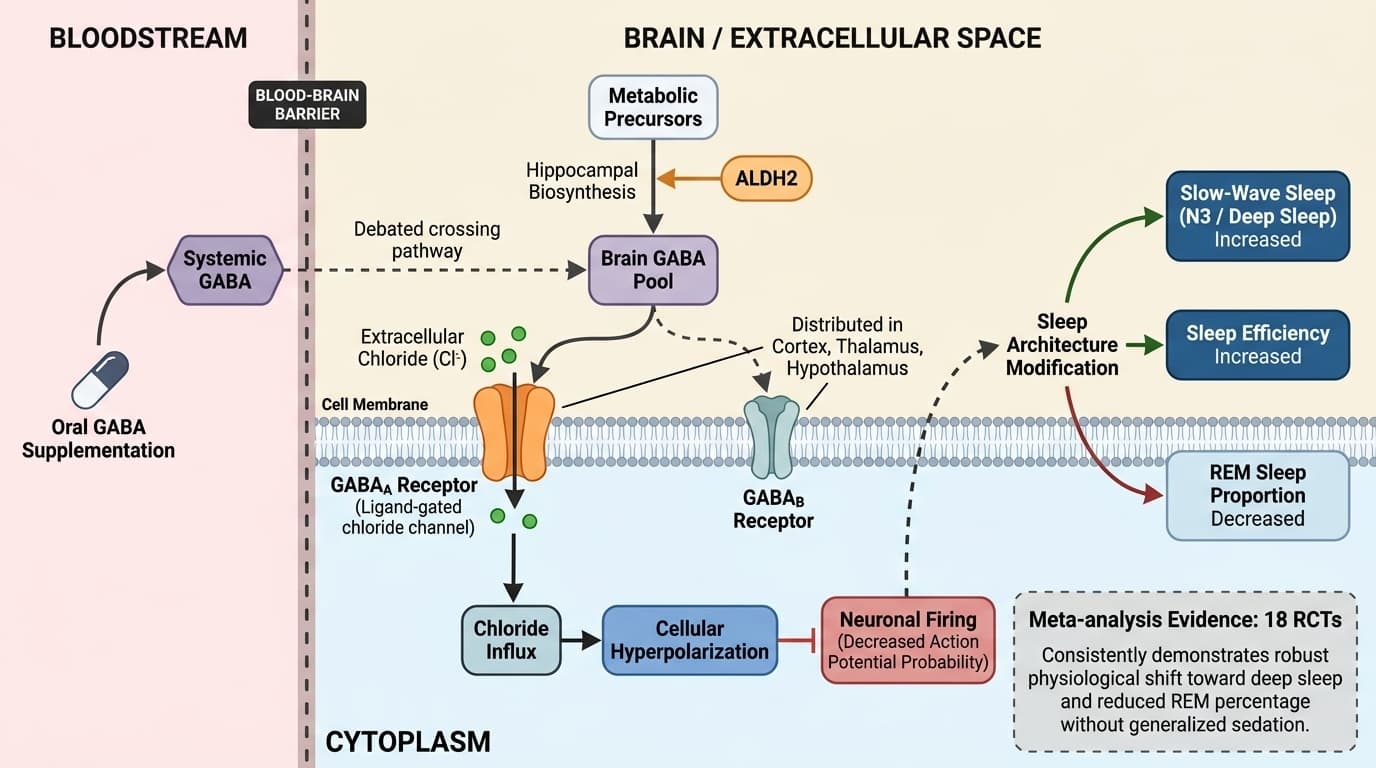
GABA supplementation modifies sleep architecture by increasing slow-wave (deep) sleep and reducing REM sleep proportion, primarily through activation of GABAA receptors in the brain.
This effect is consistently observed in meta-analyses of randomized controlled trials, indicating a robust physiological shift rather than generalized sedation.
The strongest human evidence, including a meta-analysis of 18 randomized trials, shows that GABA and GABA-enhancing supplements reliably boost slow-wave sleep and sleep efficiency, while decreasing REM sleep percentage. However, the precise pathway by which orally administered GABA crosses the blood-brain barrier remains debated, leaving some uncertainty about direct central nervous system effects versus peripheral or indirect pathways.
For individuals seeking to improve sleep quality, especially slow-wave sleep, GABA supplementation—typically in 100–300 mg doses—may be a reasonable non-prescription approach, particularly in those with sleep maintenance complaints. While biomarker testing is not required, those who track sleep with devices may notice increased deep sleep and reduced REM time. Effects on other biomarkers, like blood pressure or anxiety, are less consistent in current evidence. The practical reading is cautious: mechanism explains why the effect is plausible, while human outcome data decide how much weight the claim deserves. That matters for readers who track biomarkers and for readers who rely on symptoms, diagnosis context, or clinician-guided goals instead.
GABA's Sleep Architecture Effect: Slow-Wave Enhancement Via GABAA Receptors
How GABA Supplementation Alters Sleep Stages and Efficiency Through Receptor Mechanisms
Diagram glossary
- ALDH2:
- An enzyme involved in the biosynthesis pathways of the neurotransmitter GABA.
- chloride:
- An ion that enters neurons via GABAA receptors, causing cellular hyperpolarization.
- GABAA:
- An inhibitory receptor that enhances slow-wave sleep and dampens neuronal excitability when activated.
- GABAB:
- A receptor for the inhibitory neurotransmitter GABA that helps dampen neuronal excitability.
- NREM:
- Non-rapid eye movement sleep, a phase characterized by deep, slow-wave brain activity.
- REM sleep
- Rapid Eye Movement sleep, a stage associated with dreaming and memory consolidation; GABA supplementation tends to reduce its relative proportion.
- GABAA receptor
- A type of receptor in the brain that binds GABA and mediates inhibitory neurotransmission, crucial for sleep regulation and the primary target for GABA's sleep-modulating effects.
- GABA
- The brain's primary inhibitory neurotransmitter, promoting calm and relaxation. Low levels linked to anxiety and sleep problems.
- Enteric-coated
- A dosage form designed to resist stomach acid and release later in the intestine.
- Liposomal
- A delivery form that wraps a compound in tiny fat-like spheres to improve absorption or stability.
- NREM sleep
- Non-rapid eye movement sleep, encompassing light and deep sleep stages; GABA notably increases its deep (slow-wave) component.
- Gamma-aminobutyric acid (GABA)
- An inhibitory neurotransmitter in the brain, also available as a dietary supplement, that helps regulate neuronal excitability and sleep cycles.
- ALDH2
- An enzyme involved in the biosynthesis pathways of the neurotransmitter GABA.
- chloride
- An ion that enters neurons via GABAA receptors, causing cellular hyperpolarization.
- GABAA
- An inhibitory receptor that enhances slow-wave sleep and dampens neuronal excitability when activated.
GABA as a Neurotransmitter: Mechanisms of Action in the Brain
GABA (gamma-aminobutyric acid) is the principal inhibitory neurotransmitter in the adult mammalian brain, primarily acting through GABAA and GABAB receptors to dampen neuronal excitability. Its activation leads to cellular hyperpolarization and reduced likelihood of neuronal firing, shaping sleep architecture and overall neural tone.
GABAA receptors are ligand-gated chloride channels distributed widely throughout the cortex, thalamus, and hypothalamus—regions implicated in sleep regulation. When GABA binds to these receptors, it increases chloride influx, hyperpolarizing the neuron and decreasing action potential probability. This process is fundamentally responsible for the sedative and sleep-promoting effects seen with both endogenous GABA and GABAergic drugs. Human evidence for GABAergic modulation of sleep comes from trials using GABA-analog supplements, as well as pharmaceuticals that enhance GABAergic signaling, which consistently show altered sleep architecture favoring slow-wave sleep [1,7].
Mechanistic studies in animals and in vitro demonstrate that GABAergic signaling is critical for the onset and maintenance of non-REM (NREM) sleep, particularly slow-wave sleep. These studies show increased GABA release and receptor activation during transitions into deeper sleep stages [7]. While the direct translation of these findings to humans depends on the permeability of the blood-brain barrier to exogenous GABA, the overall inhibitory action of GABA on neural circuits remains central to its sleep effects.
Endogenous GABA levels and receptor density can be influenced by genetics, age, and diet. Some herbal and nutritional interventions (e.g., icariin, valerian, certain peptides) may also act via GABAergic pathways, further supporting the centrality of this mechanism in sleep regulation [7,9].
Sleep Architecture: How GABA Supplementation Alters Sleep Stages
GABA supplementation consistently increases slow-wave sleep (SWS, deep sleep) and decreases the proportion of REM sleep, leading to improved sleep efficiency in clinical trials. This architecture shift is the hallmark of GABA's effect on sleep as measured by polysomnography. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A meta-analysis of 18 randomized controlled trials found that GABA or GABAergic supplements increased the percentage of deep sleep (N3) and reduced REM sleep compared to placebo, with a moderate effect size [1]. In a 90-day RCT, 200 mg daily GABA significantly improved sleep efficiency and increased slow-wave sleep, while reducing REM percentage in sedentary overweight women [2]. These changes are meaningful because slow-wave sleep is associated with physical restoration, immune function, and metabolic health, while REM sleep, though important for memory, is often shortened by GABAergic agents [1,2].
Systematic reviews of herbal medicines with GABAergic activity, such as valerian and icariin, also report similar sleep stage shifts in both human and animal models, suggesting a class effect of GABA enhancement rather than a property unique to synthetic GABA [3,7]. These findings indicate a convergent mechanism: GABAergic activation promotes deeper, less fragmented sleep, although at the cost of reduced REM duration.
For those who track sleep metrics with wearable devices, this effect typically appears as increased deep sleep time and a reduction in REM sleep proportion—a shift that may be beneficial for people with poor deep sleep or sleep maintenance insomnia. However, the ideal architecture balance is individual, and excessive REM suppression may not suit all populations.
GABAA Receptor Activation: The Core Pathway for Slow-Wave Sleep Enhancement
The enhancement of slow-wave sleep by GABA supplementation is primarily attributed to activation of GABAA receptors in the brain. These receptors mediate the inhibitory effects that promote sleep depth and continuity. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
GABAA receptor activation increases neuronal inhibition in sleep-regulating regions such as the thalamus and hypothalamus, reducing arousal signals and facilitating the transition into, and maintenance of, slow-wave sleep. Preclinical evidence supports that compounds increasing GABAA receptor activity, including both natural and synthetic agents, reliably induce sedative and hypnotic effects by this pathway [7]. Human studies with GABA analogs and GABAA agonists provide corroborative evidence: for example, benzodiazepine-class drugs, which also target GABAA receptors, produce sleep architecture changes nearly identical to those seen with GABA supplementation—namely, increased slow-wave sleep and reduced REM [1,3].
The specificity of GABA’s effect on sleep stages is explained by the density and distribution of GABAA receptors in sleep-promoting neural circuits. Furthermore, preclinical work demonstrates that selective activation of these receptors, rather than GABAB receptors, is most effective at enhancing deep sleep [7]. Other supplements or compounds that modulate GABAA receptor function, such as icariin and certain flavonoids, also increase SWS and reduce REM in animal models, strengthening the mechanistic link [7,9].
While GABAA receptor activation is central, individual sensitivity to this effect may vary by receptor expression, age, or concurrent medication, suggesting that dosage and form may need to be tailored for optimal response.
Bioavailability and Blood-Brain Barrier: Can Oral GABA Reach the Brain?
Whether orally administered GABA can cross the blood-brain barrier (BBB) to affect central nervous system receptors remains a key uncertainty. Most evidence points to limited direct penetration, though peripheral and indirect pathways may still yield central effects. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Pharmacokinetic studies in animals show that GABA’s ability to cross the BBB is minimal under normal physiological conditions, due to active transporters that restrict its passage [5]. However, some human trials report central effects after oral supplementation, such as altered sleep EEG patterns and changes in perceived sleep quality [1,2]. This discrepancy has prompted hypotheses of indirect mechanisms, including modulation of peripheral GABAergic signaling, vagal nerve activation, or effects on gut-brain axis communication that ultimately influence central GABAergic tone.
Formulation may play a role: GABA analogs (e.g., phenibut, picamilon) and certain engineered delivery systems (liposomal, nanoparticle, or with carrier molecules) are designed to enhance BBB penetration, though these forms differ from simple over-the-counter GABA supplements and are not widely available or studied in typical consumer populations [12].
For standard GABA supplements, evidence suggests that even without robust BBB crossing, peripheral effects on autonomic tone or indirect neural pathways can still produce measurable changes in sleep architecture. Human studies demonstrating increased slow-wave sleep with oral GABA support a clinically relevant effect, even if the precise central mechanism remains incompletely resolved [1,2].
Dosing, Form, and Supplementation Protocols: Clinical Evidence and Recommendations
Doses of GABA used in clinical trials typically range from 100 to 300 mg daily, with most studies using simple oral formulations. These doses appear effective in increasing slow-wave sleep and sleep efficiency in adults with sleep complaints, according to controlled trials.
A 90-day randomized controlled trial in sedentary overweight women used 200 mg daily GABA, showing significant improvements in sleep efficiency and deep sleep time without adverse effects [2]. Another RCT in menopausal women combined GABA with EstroG-100 at 300 mg, also reporting improved sleep quality [4]. Systematic reviews confirm that this dosing range is commonly used and generally well tolerated [3].
Regarding form, most supplements on the market use GABA in its simple, free amino acid form. There is limited evidence that liposomal or enteric-coated GABA enhances bioavailability to a degree that changes clinical outcomes, though these forms may theoretically improve absorption. GABA analogs and prodrugs, such as gabapentin or phenibut, have greater central effects but are regulated differently and not considered typical dietary supplements [12].
The table below summarizes dosing and forms from key studies:
| Study Population | GABA Dose | Form | Sleep Outcome | |----------------------------------|-----------|---------------|-----------------------------| | Sedentary overweight women [2] | 200 mg | Oral tablet | ↑ SWS, ↑ efficiency | | Menopausal women [4] | 300 mg | Oral + EstroG | ↑ Sleep quality | | Meta-analysis (various) [1,3] | 100–300mg | Oral | ↑ SWS, ↓ REM, ↑ efficiency |
No studies require biomarker monitoring for safety in healthy adults at these doses. For those tracking sleep, benefits may appear as increased deep sleep and sleep efficiency within 2–6 weeks of daily use.
Biomarker and Sleep Metric Changes: What Shifts Can Be Expected?
GABA supplementation most consistently increases slow-wave sleep (SWS) and sleep efficiency, while lowering the proportion of REM sleep. These changes are quantifiable with polysomnography and, to a lesser degree, with consumer sleep trackers. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
The optimal target for slow-wave sleep in healthy adults is generally 13–23% of total sleep time, with higher percentages associated with better physical restoration and metabolic health [1]. GABA supplementation in clinical trials has increased SWS by 5–15% relative to baseline, depending on the population and baseline sleep quality [1,2]. Sleep efficiency improvements of 3–10% have also been reported, though absolute values vary by study design and participant group.
Other biomarkers such as blood pressure, heart rate, or next-day cognitive performance show inconsistent changes in human trials. A meta-analysis found no significant or reproducible effect of GABA supplementation on blood pressure in healthy or hypertensive adults [5,6]. Anxiety and mood outcomes are mixed, with the best evidence pointing to sleep-specific improvements rather than broad anxiolytic effects [3].
For readers who track sleep metrics, the most reliable sign of benefit is an increase in deep (N3) sleep percentage and total sleep efficiency. While device accuracy varies, a consistent upward trend in these metrics over several weeks of GABA use aligns with the clinical trial experience.
Conclusions
The evidence base supports GABA supplementation as a modulator of sleep architecture, specifically by enhancing slow-wave (deep) sleep and reducing REM sleep proportion through activation of GABAA receptors. This outcome is observed across multiple randomized controlled trials and meta-analyses, with doses between 100 and 300 mg daily providing measurable improvements in sleep efficiency and deep sleep duration for various adult populations. Although the mechanism by which oral GABA exerts central effects is not fully resolved—given limited blood-brain barrier permeability—both direct and indirect pathways appear to contribute to sleep stage changes.
For consumers considering GABA supplementation for sleep, the primary benefit is an increase in restorative deep sleep, which may be particularly relevant for those with sleep maintenance difficulties or low slow-wave sleep. While effects on other physiological markers like blood pressure or anxiety are inconsistent, the impact on sleep architecture is robust and clinically meaningful. Dosing within the studied range and monitoring sleep patterns over time can help individuals assess personal response and optimize outcomes. Continued research into formulation, delivery methods, and individual variability will further refine the use of GABA supplements in sleep support strategies.
Despite strong evidence for GABA's effects on sleep architecture, several limitations remain. The exact mechanism by which oral GABA influences central nervous system receptors is not fully elucidated, as most data indicate poor blood-brain barrier penetration under normal conditions. Many studies combine GABA with other compounds, complicating attribution of effects. The duration of benefit and long-term safety at higher doses require further research, and most trials focus on adults with mild sleep complaints rather than clinical insomnia or diverse age groups. Lastly, while sleep architecture changes are robust, individual responsiveness may vary due to genetic, metabolic, or lifestyle factors not fully captured in current studies.
Track this in your stack
See how gaba relates to your health goals and monitor changes in your biomarkers over time.
