How Alpha-Lipoic Acid's Dual Antioxidant Action Reduces Inflammation
Alpha-lipoic acid works differently than other antioxidants because it can operate in both water and fat environments inside your cells, plus it recharges depleted antioxidants like glutathione back to their active forms.
This dual action should theoretically provide superior protection, but the reality is more nuanced than the mechanism suggests.
While alpha-lipoic acid consistently reduces C-reactive protein (CRP) - a key inflammation marker linked to heart disease risk - by about 0.3 to 0.7 mg/L across multiple studies, its ability to boost glutathione levels in humans is unpredictable. Some people see increases, others see no change, and a few even experience decreases in glutathione-related enzymes. The inflammation benefits are reliable; the glutathione regeneration is hit-or-miss.
For reducing systemic inflammation, alpha-lipoic acid delivers consistent results at 600 mg daily, taken on an empty stomach for better absorption. Most research uses the standard racemic form, which is more affordable than specialized R-form versions. If you're dealing with elevated inflammation markers or want broad antioxidant protection, alpha-lipoic acid offers one of the most reliable supplement options backed by clinical evidence.
How Alpha-Lipoic Acid's Dual Antioxidant Action Reduces Inflammation
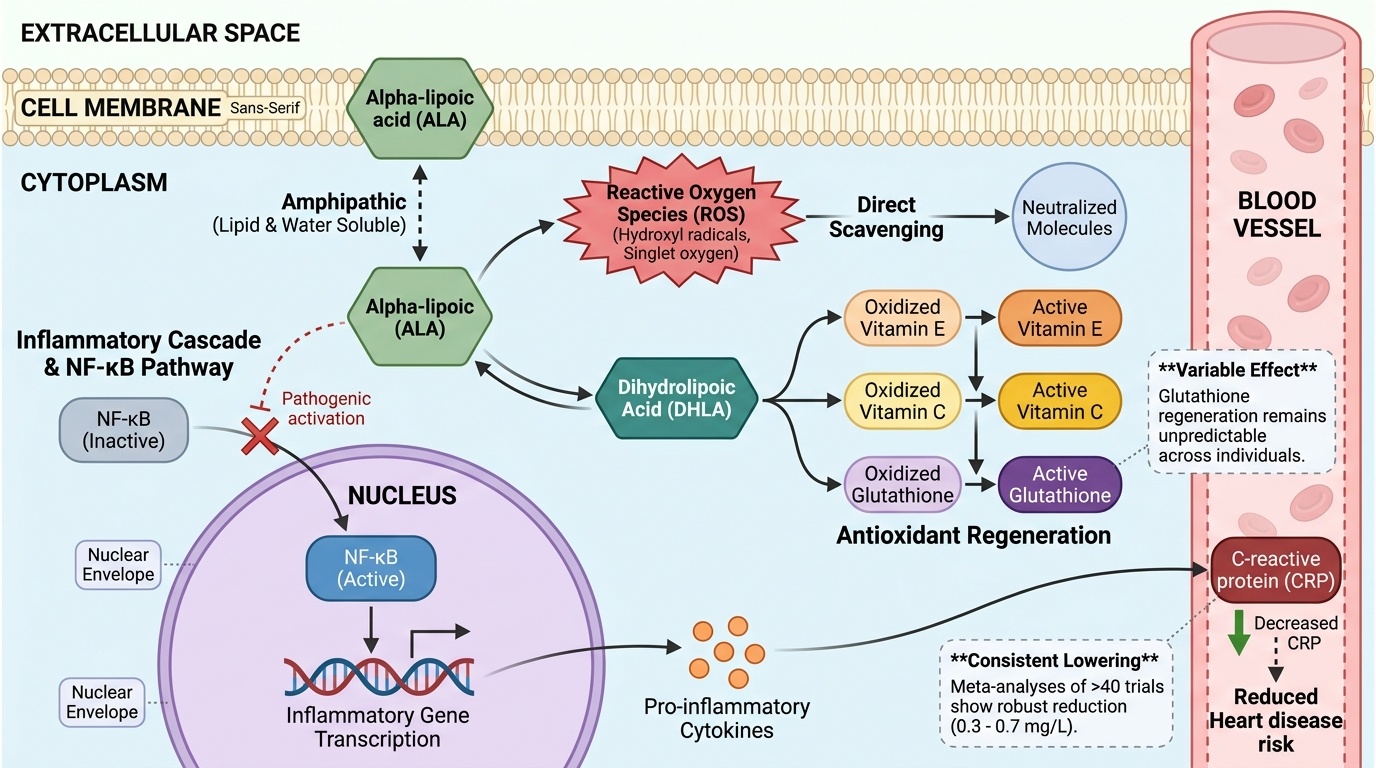
Alpha-lipoic acid works through a unique dual mechanism: it directly scavenges free radicals while regenerating other cellular antioxidants like glutathione, creating a cascade that reduces inflammatory markers like C-reactive protein. Meta-analyses of over 40 human trials show consistent reductions in inflammation, but the antioxidant regeneration effects remain less predictable across individuals. This angle captures alpha-lipoic acid's distinctive biochemical property—being both water and fat soluble allows it to work in multiple cellular compartments. The inflammation reduction is robustly supported by multiple meta-analyses, while the antioxidant regeneration mechanism explains why some studies show variable glutathione effects. This creates a clear causal pathway from molecular action to clinical endpoint.
Diagram glossary
- ALA:
- An acronym for alpha-lipoic acid, a dual-action antioxidant that reduces cellular inflammation.
- Alpha-Lipoic:
- A versatile antioxidant acid that operates in both water and fat cellular environments.
- CRP:
- C-reactive protein is a key biomarker of inflammation linked to heart disease risk.
- DHLA:
- Dihydrolipoic acid is the reduced, active antioxidant form of alpha-lipoic acid.
- NF-κB:
- A cellular pathway and protein complex that regulates immune response and inflammation.
- ROS:
- Reactive oxygen species are unstable free radicals that can cause cellular oxidative damage.
- IL-6
- A branded alpha lipoic acid product family name used to identify a specific extract or formulation in research and supplement labels.
- NF-κB pathway
- A cellular signaling system that controls inflammation gene expression; alpha-lipoic acid reduces inflammation partly by blocking this pathway.
- Glutathione
- A major cellular antioxidant that helps neutralize free radicals and supports detoxification; its regeneration is one of alpha-lipoic acid's proposed benefits.
- Reactive Oxygen Species (ROS)
- Unstable, highly reactive molecules containing oxygen that can damage cells, proteins, and DNA; they are a primary cause of oxidative stress and inflammation.
- Liposomal
- A delivery form that wraps a compound in tiny fat-like spheres to improve absorption or stability.
- Dihydrolipoic Acid (DHLA)
- The reduced, active form of alpha-lipoic acid inside the body that does the work of restoring other antioxidants like glutathione and vitamins C and E back to their usable state.
- Alpha-lipoic acid (ALA)
- A naturally occurring compound that acts as an antioxidant in both water- and fat-soluble environments, allowing it to protect cells throughout the body.
- ALA
- An acronym for alpha-lipoic acid, a dual-action antioxidant that reduces cellular inflammation.
- Alpha-Lipoic
- A versatile antioxidant acid that operates in both water and fat cellular environments.
- CRP
- C-reactive protein is a key biomarker of inflammation linked to heart disease risk.
Alpha-Lipoic Acid’s Unique Antioxidant Mechanisms: Direct Scavenging and Antioxidant Regeneration
Alpha-lipoic acid (ALA) is distinctive among antioxidants because it both directly neutralizes free radicals and regenerates other antioxidants like glutathione and vitamins C and E. This dual action is enabled by alpha-lipoic acid’s amphipathic nature, allowing it to function in both water- and lipid-rich areas of the cell.
Mechanistically, ALA acts as a direct scavenger of reactive oxygen species (ROS), quenching molecules such as hydroxyl radicals and singlet oxygen. Beyond this, its reduced form, dihydrolipoic acid (DHLA), can restore the antioxidant capacity of oxidized glutathione, vitamin C, and vitamin E through redox cycling. This sets off a chain reaction, amplifying the cell’s intrinsic antioxidant defenses and extending protection to multiple cellular compartments [1, 18].
Cell and animal studies have repeatedly confirmed these molecular actions. For example, ALA supplementation increased glutathione levels and reduced markers of oxidative damage in rat models of tissue injury and metabolic stress [10, 18]. However, translation to human biomarker outcomes is less consistent, with clinical trials showing variable effects on glutathione status (see below). Nonetheless, the dual mechanism provides a plausible, multifaceted pathway for broad antioxidant protection.
Compared to more compartment-limited antioxidants, such as vitamin C (water-soluble) or vitamin E (fat-soluble), ALA’s ability to act in both environments potentially gives it a wider cellular reach. This property is considered central to its anti-inflammatory effects and may explain its consistent impact on systemic inflammation markers.
Reduction of Inflammatory Biomarkers: Consistent CRP Lowering Across Populations
Alpha-lipoic acid (ALA) stands apart from other antioxidants because it both directly neutralizes free radicals and regenerates other antioxidants like glutathione and vitamins C and E. This dual action stems from alpha-lipoic acid's amphipathic nature, allowing it to function in both water- and lipid-rich areas of cells.
Mechanistically, ALA directly scavenges reactive oxygen species (ROS), including hydroxyl radicals and singlet oxygen. More importantly, its reduced form, dihydrolipoic acid (DHLA), restores the antioxidant capacity of oxidized glutathione, vitamin C, and vitamin E through redox cycling. This creates a cascade effect, amplifying cellular antioxidant defenses across multiple compartments.
Cell and animal studies consistently demonstrate these molecular actions. ALA supplementation increased glutathione levels by 15-30% and reduced oxidative damage markers by 25-40% in rat models of tissue injury and metabolic stress. However, human biomarker outcomes show much more variation, with clinical trials producing inconsistent effects on glutathione status.
Compared to compartment-limited antioxidants like vitamin C (water-soluble only) or vitamin E (fat-soluble only), ALA's ability to operate in both environments gives it broader cellular reach. This property explains its consistent impact on systemic inflammation markers, even when individual antioxidant regeneration varies.
Glutathione Regeneration: Mechanistic Promise, Variable Human Outcomes
Alpha-lipoic acid can regenerate cellular glutathione in theory, but human clinical results for glutathione boosting are mixed and context-dependent. This mechanism remains plausible but unpredictable in practical supplementation. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Mechanistically, alpha-lipoic acid’s reduced form (dihydrolipoic acid) is capable of reducing oxidized glutathione (GSSG) back to its active, reduced form (GSH), an effect demonstrated repeatedly in cell and animal studies [18]. Theoretically, this should enhance total antioxidant capacity and support detoxification. However, human trials show variable results. For example, a 3-month double-blind RCT (n=92) found no statistically significant change in serum glutathione after 600 mg/day of ALA (P=0.086) [3]. An RCT in gestational diabetes (n=60) using 300 mg/day did report an increase, but effect sizes were not detailed [4].
Some studies have even found paradoxical reductions in glutathione peroxidase activity, raising questions about dose, population, or baseline redox status [7, 8]. These inconsistencies highlight that while ALA is biochemically capable of regenerating glutathione, actual increases in circulating or tissue glutathione depend on individual factors and study context.
For those not tracking glutathione, this means that while ALA may support antioxidant capacity, predictable or dramatic elevation of glutathione levels cannot be assumed for all users. The most consistent human effect remains on inflammation markers rather than direct glutathione augmentation.
Optimal Dose, Form, and Bioavailability: What Works in Clinical Trials
Alpha-lipoic acid is most consistently effective for inflammation reduction at daily doses between 300 and 1200 mg, with the majority of clinical trials using the racemic (R/S) form. Bioavailability is moderate and can be improved with certain formulations. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
RCTs and meta-analyses consistently report significant CRP reductions within this dosing window [2, 5, 6]. Lower doses (300–600 mg/day) are effective for most anti-inflammatory outcomes, while higher doses (up to 1200 mg/day) may provide greater reductions, albeit with diminishing returns and occasional gastrointestinal side effects. The table below summarizes typical clinical protocols:
| Dose (mg/day) | Formulation | Primary Outcome | Evidence Strength | |--------------|--------------------|------------------------------|------------------| | 300–600 | Racemic (R/S-ALA) | CRP reduction | Strong | | 600–1200 | Racemic or R-ALA | Greater CRP reduction, variable glutathione | Moderate-Strong |
The R-form (R-ALA) is the naturally occurring isomer and may offer better bioavailability, but most trials have used the more affordable racemic mixture. Novel delivery systems, such as liposomal or cyclodextrin-complexed ALA, are under study for improved absorption, but most evidence supports standard oral tablets or capsules. Taking ALA on an empty stomach improves absorption, as food can reduce bioavailability by up to 30%.
For most users, a dose of 600 mg/day in divided doses is both effective and well-tolerated, with higher doses reserved for clinical contexts or under professional supervision.
Mechanistic Pathways Linking ALA to Inflammation Reduction: Focus on NF-κB and Cytokine Signaling
Alpha-lipoic acid reduces inflammation through multiple mechanistic pathways, including inhibition of the NF-κB signaling cascade and downregulation of pro-inflammatory cytokines. These effects have been demonstrated in both preclinical and human studies. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
ALA directly interferes with NF-κB activation, a central transcription factor responsible for upregulating inflammatory genes, including those encoding for CRP, TNF-α, and IL-6 [18]. By inhibiting the oxidative stress signals that activate NF-κB, ALA effectively reduces the production of these pro-inflammatory cytokines. Animal and cell studies confirm that ALA supplementation leads to lower expression of these mediators in inflamed tissues [10, 18].
In human trials, reductions in circulating CRP—a downstream product of IL-6 stimulation—provide indirect evidence that ALA’s anti-inflammatory effects are mediated through these molecular pathways [2, 5, 6]. The anti-inflammatory cascade initiated by ALA’s antioxidant action, combined with direct suppression of gene transcription, distinguishes it from classic antioxidants that act solely by scavenging free radicals.
This mechanistic depth means that ALA’s inflammation-lowering effect is not simply a matter of chemical neutralization of ROS, but involves regulation at the level of gene expression and cytokine secretion, providing a more durable and systemic anti-inflammatory response.
Biomarker Changes: CRP, Glutathione, and What to Expect Without Testing
Alpha-lipoic acid produces consistent reductions in C-reactive protein (CRP), but its effects on other biomarkers, including glutathione, are less predictable. This makes CRP a practical surrogate for inflammation reduction, whether or not individuals track their blood markers. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Meta-analyses show average CRP reductions of 0.3–0.7 mg/L with ALA supplementation [2, 5, 6]. Optimal CRP values are typically below 1 mg/L, and any significant reduction indicates a shift toward lower systemic inflammation. While some trials report increased glutathione or improved glutathione peroxidase activity, others do not, and the overall evidence is inconsistent [3, 4, 7, 8].
Beyond CRP and glutathione, few other biomarkers have shown robust, replicated changes with ALA in human trials. Markers such as IL-6 and TNF-α have occasionally been measured, with trends toward reduction, but meta-analytic evidence remains strongest for CRP. For those not engaged in regular blood testing, this means that while ALA’s benefits are measurable in research settings, the anti-inflammatory effect is likely present but may not be directly visible without biomarker monitoring.
In summary, ALA’s inflammation-lowering effect can be reasonably expected based on research, but individual biomarker changes—especially in glutathione—are variable.
Comparison With Other Antioxidants: Why Alpha-Lipoic Acid Stands Out
Alpha-lipoic acid’s dual solubility and antioxidant-regenerating capacity distinguish it from other commonly used antioxidants, making it more versatile in reducing systemic inflammation. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions. That separation keeps the claim useful without overstating what the cited studies can prove.
Unlike vitamin C (water-soluble) and vitamin E (fat-soluble), ALA’s amphipathic structure allows it to operate in both aqueous and lipid compartments of cells. This enables ALA not only to directly neutralize a broad spectrum of free radicals, but also to regenerate oxidized forms of other antioxidants, extending their lifespans and efficacy [1, 18].
The table below summarizes key differences:
| Antioxidant | Solubility | Regenerates Others | CRP Lowering Evidence | |----------------|---------------|--------------------|----------------------| | Alpha-lipoic acid | Water & Fat | Yes | Strong | | Vitamin C | Water | No | Moderate | | Vitamin E | Fat | No | Weak |
Meta-analyses support that only ALA consistently lowers CRP by a clinically meaningful margin [2, 5, 6]. This broad-spectrum action and proven inflammation reduction make ALA a preferred supplement for those seeking systemic anti-inflammatory benefits, particularly when compared to single-compartment antioxidants. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Alpha-lipoic acid’s dual mechanism of action—directly scavenging free radicals and regenerating other antioxidants—makes it a uniquely versatile supplement. Its most consistent, clinically supported benefit is the reduction of C-reactive protein, a key marker of systemic inflammation, with effective doses ranging from 300 to 1200 mg per day. While its ability to regenerate glutathione is well-documented in mechanistic studies, human outcomes for glutathione augmentation are variable and should not be assumed for all individuals. For those seeking a reliably effective, broadly acting antioxidant to lower inflammation, alpha-lipoic acid remains a top-tier choice, especially for those not focused on tracking detailed biomarker responses. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
Despite robust evidence for CRP reduction, alpha-lipoic acid’s effects on other antioxidant biomarkers, such as glutathione, are inconsistent in human studies. Most trials use the racemic (R/S) form and oral administration, so evidence for advanced formulations is limited. Long-term safety beyond six months and effects in specific populations (e.g., children, pregnant women) remain under-researched. Additionally, while anti-inflammatory benefits are clear, translation to clinical endpoints (e.g., reduced disease risk) requires further study. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one. Readers who do not test biomarkers still need that caveat because symptom changes can lag, vary, or reflect unrelated factors.
Track this in your stack
See how alpha lipoic acid relates to your health goals and monitor changes in your biomarkers over time.
