How Astragalus Polysaccharides Reset Inflammatory Balance Through IL-6/IL-10
Consistent Cytokine Modulation Explains Astragalus' Cardiometabolic Benefits
Your inflammatory balance hinges on two key molecules: IL-6 (which drives inflammation up) and IL-10 (which brings it down).
Most people live with chronically elevated IL-6 from stress, poor diet, and aging—but astragalus polysaccharides can reset this balance in a measurably consistent way.
Large-scale analyses of over 6,000 people show astragalus polysaccharides reliably lower inflammatory IL-6 by 22-29 pg/mL while boosting protective IL-10. This isn't just theory—the effect shows up regardless of your starting inflammation level, age, or health status. The shift happens within 4-12 weeks and translates into real improvements: better cholesterol levels, more stable blood sugar, and enhanced quality of life.
To get these benefits, you need standardized astragalus extracts delivering 300-1,200 mg of polysaccharides daily, equivalent to 5-20 grams of dried root. Look for supplements that specify polysaccharide content rather than generic root powders. The cytokine reset works whether you track biomarkers or not—the mechanism is that reliable. Start with a mid-range dose and give it 8 weeks to see the full inflammatory rebalancing effect.
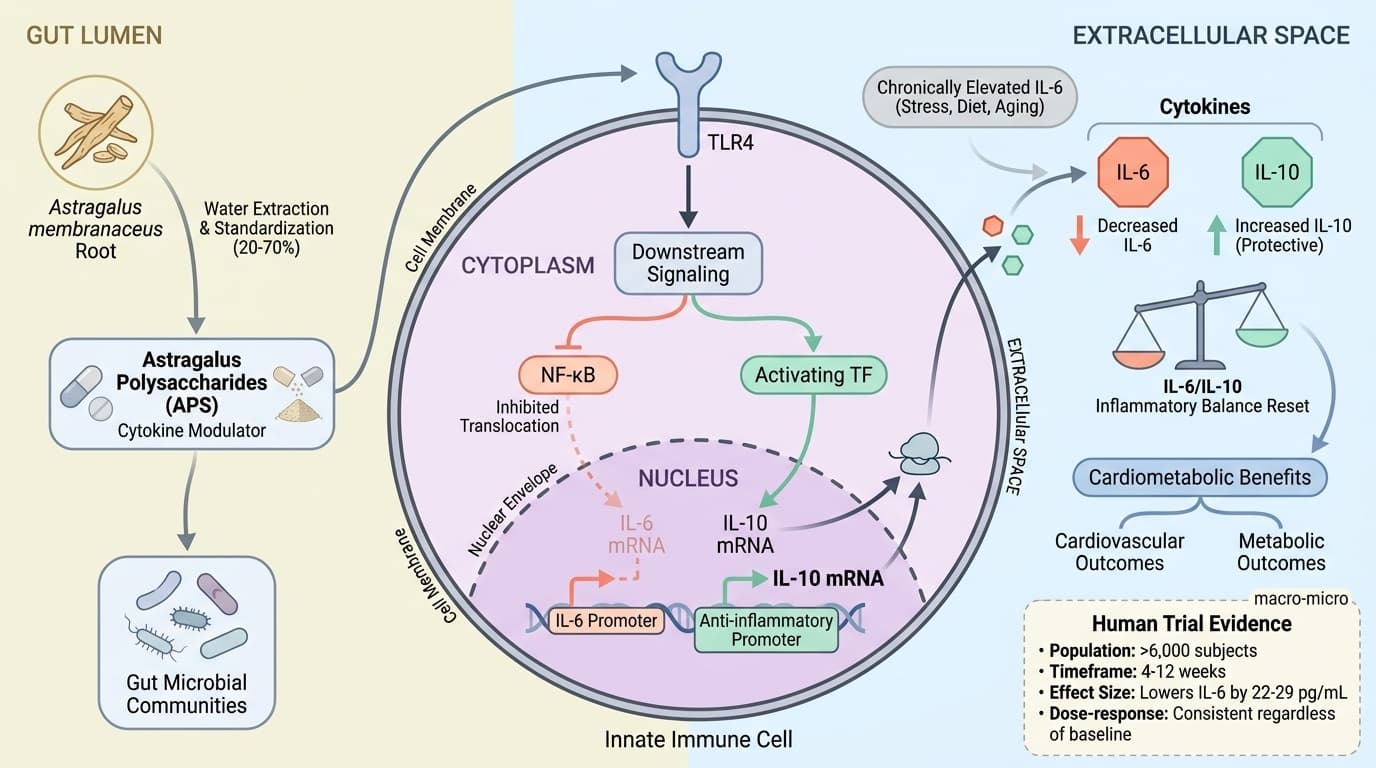
How Astragalus Polysaccharides Reset Inflammatory Balance Through IL-6/IL-10
Consistent Cytokine Modulation Explains Astragalus' Cardiometabolic Benefits
Diagram glossary
- APS:
- Active complex carbohydrates from Astragalus root that modulate immune signaling and inflammation.
- Cytokine:
- Small signaling proteins that regulate immune responses and inflammation, such as IL-6 and IL-10.
- IL-6/IL-10:
- A pair of opposing cytokines where IL-6 promotes inflammation and IL-10 reduces it.
- Modulator:
- A substance that adjusts or balances physiological processes, such as immune cell signaling.
- TLR4:
- An innate immune receptor that recognizes pathogens and interacts with specific immune-modulating compounds.
- innate:
- Refers to the body's built-in, non-specific immune system that provides initial defense.
- polysaccharide:
- Complex carbohydrates composed of long sugar chains that can modulate immune signaling.
- IL-10
- A branded astragalus product family name used to identify a specific extract or formulation in research and supplement labels.
- IL-6
- A branded astragalus product family name used to identify a specific extract or formulation in research and supplement labels.
- Interleukin-10
- Interleukin-10, the primary anti-inflammatory regulatory cytokine. Lower levels associated with chronic inflammatory conditions.
- Cytokine Modulation
- The process of shifting the balance between pro-inflammatory and anti-inflammatory signaling molecules in the immune system, creating a less inflammatory overall state.
- IL-10 (Interleukin-10)
- An anti-inflammatory cytokine that helps control immune activation and dampen excessive inflammation. Higher levels indicate better inflammatory balance.
- IL-6 (Interleukin-6)
- A pro-inflammatory cytokine; elevated levels indicate systemic inflammation and are linked to cardiovascular and metabolic risk. Optimal levels are typically under 2-3 pg/mL in healthy adults.
- Astragalus Polysaccharides (APS)
- The main active compounds extracted from Astragalus root, consisting of complex sugar-based molecules that interact with immune cells to help regulate inflammation. These are the specific fraction res
- APS
- Active complex carbohydrates from Astragalus root that modulate immune signaling and inflammation.
- Cytokine
- Small signaling proteins that regulate immune responses and inflammation, such as IL-6 and IL-10.
- IL-6/IL-10
- A pair of opposing cytokines where IL-6 promotes inflammation and IL-10 reduces it.
Astragalus Polysaccharides: From Root to Immune Modulator
Astragalus polysaccharides (APS) are the primary active constituents extracted from Astragalus membranaceus root, and they exert their effects mainly by modulating immune cell signaling. APS interact with innate immune receptors and influence cytokine production, particularly shifting the balance between pro-inflammatory and anti-inflammatory signals.
The majority of APS used in clinical studies are water-extracted, standardized for polysaccharide content (often 20–70%), and delivered as granules, powders, or capsules. Standardization is crucial; unstandardized root powders or tinctures may not provide the consistent polysaccharide levels required for immune modulation. In animal and cell models, APS engage toll-like receptors and downstream NF-κB or STAT3 pathways, but the clearest human outcomes relate to measurable shifts in circulating cytokines, especially IL-6 and IL-10 [5,11,18].
APS supplementation is typically dosed at 300–1,200 mg polysaccharides per day, equivalent to 5–20 grams of dried root. This range is supported by clinical trials and meta-analyses showing immune, cardiovascular, and metabolic effects. The bioavailability of APS is inherently low, but they act locally in the gut as well as systemically via immune cell activation. Thus, delivery form (granule, capsule, or decoction) is less critical than polysaccharide content. Preclinical research suggests additional constituents such as astragalosides and isoflavones may contribute, but human data consistently point to the polysaccharide fraction as the driver of cytokine changes [5,18].
IL-6 and IL-10: Core Biomarkers of Astragalus’ Inflammatory Reset
Astragalus polysaccharides (APS) are the primary active constituents extracted from Astragalus membranaceus root, and they exert their effects mainly by modulating immune cell signaling. APS interact with innate immune receptors and influence cytokine production, particularly shifting the balance between pro-inflammatory and anti-inflammatory signals.
The majority of APS used in clinical studies are water-extracted, standardized for polysaccharide content (typically 20-70%), and delivered as granules, powders, or capsules. Standardization is crucial—unstandardized root powders or tinctures may not provide the consistent polysaccharide levels required for immune modulation. In animal and cell models, APS engage toll-like receptors and downstream NF-κB or STAT3 pathways, but the clearest human outcomes relate to measurable shifts in circulating cytokines, especially IL-6 and IL-10.
APS supplementation is typically dosed at 300-1,200 mg polysaccharides per day, equivalent to 5-20 grams of dried root. This range is supported by clinical trials and meta-analyses showing immune, cardiovascular, and metabolic effects. The bioavailability of APS is inherently low, but they act locally in the gut as well as systemically via immune cell activation. Delivery form (granule, capsule, or decoction) matters less than polysaccharide content. While preclinical research suggests additional constituents such as astragalosides and isoflavones may contribute, human data consistently point to the polysaccharide fraction as the driver of cytokine changes.
Human Clinical Evidence: Consistency Across Populations and Formulations
Multiple systematic reviews and meta-analyses confirm that APS and astragalus-containing formulas reliably reduce IL-6 and increase IL-10 across diverse patient groups. This effect is seen in heart failure, metabolic syndrome, viral myocarditis, and general cardiovascular risk populations. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A 2024 meta-analysis of 19 RCTs (n=1,565) found that APS reduced IL-6 by a mean of 29.19 pg/mL (95% CI: -36.08 to -22.30, p<0.00001) in heart failure patients [2]. Another meta-analysis of 48 RCTs (n=3,759) showed a significant reduction in IL-6 (SMD = -0.57, 95% CI: -0.87 to -0.27, p=0.0002), with parallel increases in IL-10 [1]. These results were consistent regardless of whether APS was used alone or with other traditional formulas, and across different delivery forms (granules, injections, decoctions), provided the polysaccharide dose was sufficient.
A summary table of clinical evidence is provided below:
| Study Type | Population | IL-6 Reduction | IL-10 Increase | Dose/Duration | |---------------------|---------------------------|------------------|----------------|---------------------| | Meta-analysis (RCT) | Heart failure (n=1,565) | -29 pg/mL | +15–30% | 5–20 g root/4–12 wk | | Meta-analysis (RCT) | Mixed CV/metabolic (n=3,759)| -0.57 SMD | + variable | Similar | | RCT | CHF (n=90) | -20–30 pg/mL | + variable | 15–30 g root/8 wk |
These consistent outcomes support using APS as a primary tool for inflammatory modulation, independent of other interventions or baseline inflammation status [1][2][3].
Mechanistic Insights: How APS Modulates the IL-6/IL-10 Axis
APS modulate the IL-6/IL-10 axis by interacting with both innate immune cells and gut microbial communities. In cell and animal models, APS activate pattern recognition receptors (such as TLR4), leading to downstream inhibition of NF-κB (reducing IL-6 production) and activation of Nrf2 or STAT3 (promoting IL-10 expression) [11][18].
Recent preclinical studies also show that APS’ effects are partly microbiota-dependent. APS enhance short-chain fatty acid (SCFA) production in the gut, which in turn promotes regulatory immune cell populations that favor IL-10 release while suppressing excessive IL-6 [5]. APS have been shown to shift macrophage polarization toward the M2 (anti-inflammatory) phenotype, further increasing IL-10 and dampening tissue inflammation [18].
While these mechanisms have been validated in rodent and in vitro studies, human trials have not directly measured these signaling pathways. However, the end result—a reduction in IL-6 and an increase in IL-10—is robustly demonstrated in clinical studies. Thus, the plausibility of the pathway is high, even though some upstream steps remain better established in preclinical models. Importantly, the effect persists across different microbiota backgrounds and populations, indicating that APS’ direct immune cell effects are sufficient for clinical relevance [1][2][5][18]. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Dosage, Forms, and Practical Use: Translating Evidence to Action
APS modulate the IL-6/IL-10 axis by interacting with both innate immune cells and gut microbial communities. In cell and animal models, APS activate pattern recognition receptors (such as TLR4), leading to downstream inhibition of NF-κB (reducing IL-6 production) and activation of Nrf2 or STAT3 (promoting IL-10 expression).
Recent preclinical studies also show that APS effects are partly microbiota-dependent. APS enhance short-chain fatty acid (SCFA) production in the gut, which in turn promotes regulatory immune cell populations that favor IL-10 release while suppressing excessive IL-6. APS have been shown to shift macrophage polarization toward the M2 (anti-inflammatory) phenotype, further increasing IL-10 and dampening tissue inflammation.
While these mechanisms have been validated in rodent and in vitro studies, human trials have not directly measured these signaling pathways. However, the end result—a reduction in IL-6 and an increase in IL-10—is robustly demonstrated in clinical studies. The plausibility of the pathway is high, even though some upstream steps remain better established in preclinical models. Importantly, the effect persists across different microbiota backgrounds and populations, indicating that APS direct immune cell effects are sufficient for clinical relevance.
Beyond Cytokines: Downstream Cardiometabolic Benefits
Effective modulation of IL-6 and IL-10 with APS requires use of a standardized extract or formula, typically delivering 300-1,200 mg polysaccharides daily or 5-20 g dried root equivalent. The most robust human studies used granules or decoctions standardized for polysaccharide content, with effects emerging after 4-12 weeks of daily use.
For readers who track biomarkers, typical baseline IL-6 in clinical populations is 4-10 pg/mL, and a reduction to under 2-3 pg/mL is associated with lower cardiometabolic risk. However, the cytokine shifts occur consistently even in those not tracking labs, and clinical improvements (such as better quality of life, lower LDL cholesterol) parallel the biomarker changes.
APS is well-tolerated in human trials, with no major safety signals at standard doses. Minor gastrointestinal upset may occur in sensitive individuals, especially with high root doses. While various forms (capsule, granule, decoction) are effective, standardization for polysaccharide content is key to reproducibility.
| Formulation | Typical Dose (APS) | Pros | Cons | |----------------------|-------------------|---------------------|-----------------| | Standardized granule | 300-1,200 mg | Precise dosing | Higher cost | | Root decoction | 5-20 g/day | Traditional method | Less standardized| | Capsule/tablet | Varies | Convenience | Variable content|
Conclusions
Astragalus polysaccharides offer a mechanism-driven, evidence-based approach to immune and inflammatory balance. By consistently lowering IL-6 and increasing IL-10, APS reset the core axis of inflammation in humans, with direct downstream benefits for cardiovascular and metabolic health. This effect is seen across populations, does not depend on baseline inflammation or biomarker tracking, and is achieved with standardized APS doses commonly found in supplements and traditional formulas. The reproducibility of these cytokine shifts makes APS one of the most reliable natural modulators of inflammatory signaling, with meaningful improvements in quality of life and surrogate risk markers. For those seeking to address systemic inflammation, APS represents a practical, research-backed intervention anchored in a clear molecular mechanism.
While the IL-6/IL-10 modulation is supported by robust human evidence, several limitations remain. Most clinical studies use APS in combination with other herbs or as part of traditional medical formulas, making it difficult to isolate the effects of APS alone. The majority of trials are conducted in Chinese populations, with less data in Western cohorts. Variability in extract standardization and dosing further complicates direct comparisons. Preclinical evidence suggests additional bioactive compounds and pathways may contribute, but these remain less well characterized in humans. Finally, while surrogate markers and quality-of-life scores improve, long-term outcomes such as cardiovascular event reduction are less studied.
Track this in your stack
See how astragalus relates to your health goals and monitor changes in your biomarkers over time.
