How B-Complex Vitamins Lower Homocysteine Through Methylation Pathways
Mechanistic Evidence and Practical Insights for Cardiovascular Biomarker Optimization
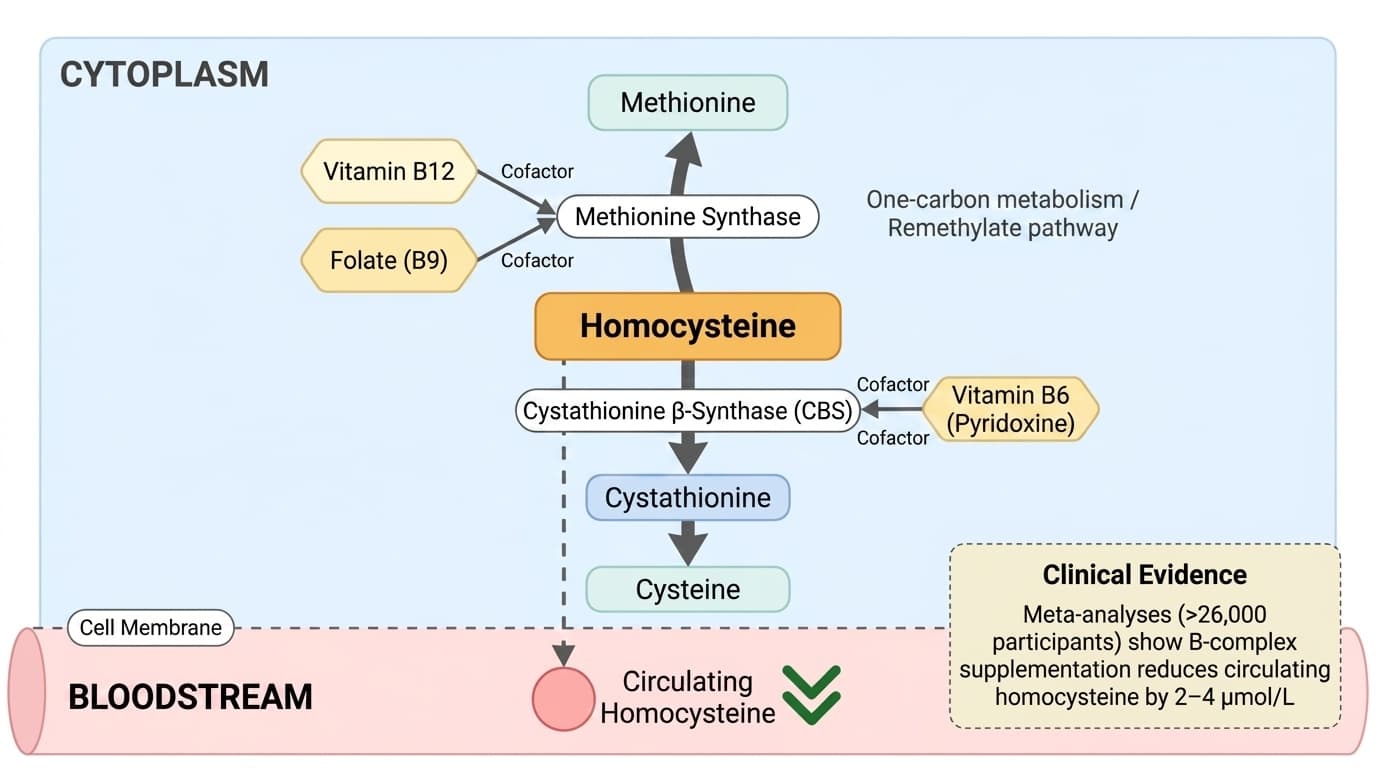
B-complex vitamins—especially B6, B9 (folate), and B12—act as essential cofactors in the one-carbon metabolism pathway, enabling the conversion of homocysteine to methionine or cysteine.
This mechanistic role consistently leads to lower homocysteine levels in the blood, a biomarker linked to cardiovascular health.
Large-scale meta-analyses including over 26,000 participants show that supplementation with B-complex vitamins (often at doses of B6 ≥2 mg, folic acid 400–800 mcg, and B12 ≥6 mcg daily) reduces circulating homocysteine by 2–4 μmol/L. While this effect is robust and reproducible, the direct translation to clinical cardiovascular events remains less certain. The evidence does not show consistent improvement in cognitive outcomes, indicating the effect is mechanistically specific to the methylation cycle and homocysteine biomarker.
For individuals seeking to optimize homocysteine—whether or not they track their numbers—B-complex supplementation offers a reliable, evidence-supported way to modulate this biomarker. The effect is not dependent on baseline homocysteine or responder status, and the forms used in trials are typically standard synthetic B-complex blends. Choosing a balanced B-complex supplement that includes B6, B9, and B12 in at least the studied doses is supported by both mechanism and high-quality human evidence. The practical reading is cautious: mechanism explains why the effect is plausible, while human outcome data decide how much weight the claim deserves.
How B-Complex Vitamins Lower Homocysteine Through Methylation Pathways
Mechanistic Evidence and Practical Insights for Cardiovascular Biomarker Optimization
Diagram glossary
- B12:
- A vitamin acting as an essential cofactor to remethylate homocysteine into methionine.
- B6:
- A vitamin serving as a cofactor in the conversion of homocysteine to cysteine.
- CBS:
- An enzyme that uses vitamin B6 to convert homocysteine into cystathionine.
- cofactor:
- A non-protein chemical compound required for an enzyme's biological activity.
- Cystathionine:
- An intermediate amino acid formed during the conversion of homocysteine to cysteine.
- folate:
- Vitamin B9, an essential cofactor required for the remethylation of homocysteine to methionine.
- Homocysteine:
- An amino acid biomarker whose elevated blood levels are linked to cardiovascular disease.
- Methionine:
- An essential amino acid produced through the remethylation of homocysteine.
- pyridoxine:
- Another name for vitamin B6, an essential cofactor in amino acid metabolism.
- remethylate:
- The biochemical process of adding a methyl group back to a molecule like homocysteine.
- One-carbon metabolism
- A biochemical pathway that transfers single carbon units between molecules and is central to DNA synthesis and methylation; B vitamins are key cofactors in this cycle.
- Pyridoxine hydrochloride
- The most common supplemental form of vitamin B6, where pyridoxine (the active vitamin) is bound to a hydrochloride salt to improve stability and solubility in pill or capsule form.
- Homocysteine
- Homocysteine, an amino acid metabolite influenced by B vitamins. elevated levels damage blood vessels and increase cardiovascular and dementia risk.
- Cystathionine β-synthase
- An enzyme that converts homocysteine to cystathionine, requiring vitamin B6; another main route for homocysteine metabolism.
- Methylation
- A chemical process involving the transfer of a methyl group, crucial for gene expression and metabolism, and dependent on B vitamins.
- Liposomal
- A delivery form that wraps a compound in tiny fat-like spheres to improve absorption or stability.
- Bioavailability
- The proportion of a compound that actually reaches your bloodstream and becomes usable by the body.
- B12
- A vitamin acting as an essential cofactor to remethylate homocysteine into methionine.
- B6
- A vitamin serving as a cofactor in the conversion of homocysteine to cysteine.
- CBS
- An enzyme that uses vitamin B6 to convert homocysteine into cystathionine.
B-Complex Vitamins as Cofactors in One-Carbon Metabolism
B-complex vitamins, specifically B6, B9 (folate), and B12, serve as indispensable cofactors in one-carbon metabolism, which orchestrates the transfer of single carbon units for methylation reactions. Their coordinated action enables the efficient conversion of homocysteine to methionine or cysteine, directly impacting circulating homocysteine concentrations.
Vitamin B12 (cobalamin) and folate are primarily responsible for remethylating homocysteine to methionine via the methionine synthase enzyme, while vitamin B6 (pyridoxine) is essential for the transsulfuration pathway, converting homocysteine to cystathionine through cystathionine β-synthase. This division of labor among B vitamins ensures that both remethylation and transsulfuration pathways remain functional, preventing excessive accumulation of homocysteine and maintaining a balance in methyl group supply for DNA and protein methylation.
Human trials and meta-analyses repeatedly confirm that supplementation with B-complex vitamins, especially with adequate doses of B6, B9, and B12, reliably lowers homocysteine levels [9][10]. This mechanistic specificity distinguishes B-complex supplementation from general multivitamin use or dietary interventions that do not target these methylation pathways directly. The consistent reduction in homocysteine seen in supplementation trials underscores the central biochemical role of B vitamins in one-carbon metabolism. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Homocysteine: Biomarker Significance and Target Ranges
Homocysteine is a sulfur-containing amino acid whose plasma concentration is a widely accepted biomarker for cardiovascular risk. Optimal fasting plasma homocysteine is generally considered to be below 10 µmol/L, with levels above 15 µmol/L classified as hyperhomocysteinemia and associated with increased risk for vascular events.
The relevance of homocysteine as a modifiable biomarker is supported by large-scale human studies. Meta-analyses of randomized controlled trials (RCTs) show that B-complex vitamin supplementation, particularly combinations of folic acid, B12, and B6, can reduce serum homocysteine by approximately 2–4 μmol/L in both general and high-risk populations [9][10]. This effect is observed regardless of baseline homocysteine, age, or sex, indicating broad applicability.
While reductions in homocysteine are consistent, the translation to clinical endpoints such as cardiovascular events remains less clear. Nevertheless, lowering homocysteine is still viewed as a valid target for individuals seeking to optimize this biomarker, given its mechanistic link to methylation and vascular integrity. B-complex supplementation represents a direct and reliable method to achieve this biomarker modulation. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Enzyme Targets: Methionine Synthase and Cystathionine β-Synthase
The primary enzymatic targets for B-complex action in homocysteine metabolism are methionine synthase (MS) and cystathionine β-synthase (CBS). Methionine synthase requires both vitamin B12 and folate as cofactors to remethylate homocysteine, converting it back into methionine—a vital amino acid for methylation reactions.
Dysfunction or insufficiency in any of these enzymes, or their respective B-vitamin cofactors, leads to impaired homocysteine clearance and elevated plasma homocysteine. This is directly evidenced in genetic disorders affecting these enzymes, but subclinical insufficiency of B6, B9, or B12 can also raise homocysteine in otherwise healthy adults. Human RCTs with B-complex supplementation demonstrate that providing all three key B vitamins leads to the largest reductions in homocysteine, supporting the need for full pathway support [9][10].
A recent animal study also supports the nephroprotective effect of B-complex (specifically B1, B6, B12) in the context of drug-induced oxidative stress, suggesting that adequate B-vitamin status supports both methylation and antioxidant defenses, though these findings require confirmation in humans [7]. Overall, the mechanistic evidence for these enzymes as targets is robust and directly explains the observed homocysteine-lowering effect. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Clinical Evidence: B-Complex Supplementation and Homocysteine Reduction
Human clinical trials and meta-analyses provide strong support for B-complex supplementation as an efficacious intervention to lower homocysteine. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions. That separation keeps the claim useful without overstating what the cited studies can prove.
These effects are consistent across diverse populations and not limited by age, sex, or baseline homocysteine level. Most trials use folic acid (400–800 mcg), B12 (6–500 mcg), and B6 (2–50 mg) as the core intervention, either alone or in combination with other B vitamins. The effect appears dose-dependent up to typical supplemental ranges, but higher doses have not shown proportionally greater benefit in homocysteine lowering. Table 1 summarizes the typical forms and doses used in key studies:
| Study/Meta-Analysis | Folic Acid Dose | B12 Dose | B6 Dose | Mean Homocysteine Reduction | |-----------------------|----------------|-----------|-----------|-----------------------------| | PMID: 27453118 | 400–800 mcg | 6–500 mcg | 2–50 mg | -2.53 μmol/L | | PMID: 41615824 | 400–1200 mcg | 6–500 mcg | 2–50 mg | -2.36 μmol/L |
The magnitude of effect is sufficient to move many individuals from above to below the 10–12 μmol/L threshold, supporting both clinical and practical utility. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Dosage, Form, and Bioavailability Considerations
B-complex supplements used in clinical trials typically contain folic acid (the synthetic, stable form of B9), cyanocobalamin or methylcobalamin (B12), and pyridoxine hydrochloride (B6). The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Bioavailability is generally high for folic acid and B6, but B12 absorption can be limited by intrinsic factor and gastrointestinal health. For individuals with absorption issues or atrophic gastritis, sublingual, chewable, or injectable forms of B12 may be more effective, although most RCTs have used standard oral supplements. There is no evidence that liposomal or nanoparticle formulations enhance homocysteine reduction over standard forms in humans, but these may be considered in cases of documented malabsorption.
The choice between folic acid and methylfolate, or cyanocobalamin and methylcobalamin, is often driven by personal preference or specific health considerations, but current human evidence supports efficacy with standard forms. Consistency in daily intake appears more important than specific formulation for the homocysteine-lowering effect. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Comparing Direct and Indirect Evidence: Human Trials Versus Mechanistic Models
Direct human evidence, including meta-analyses of RCTs, demonstrates that B-complex vitamins reliably lower homocysteine, with reductions of 2–4 μmol/L observed across populations [9][10]. This effect size is robust and reproducible with standard supplement forms and doses, and the mechanism is well-mapped biochemically.
Indirect evidence from animal and mechanistic models provides additional insight into the pathways involved, such as the role of B-complex in antioxidant defense and cellular protection under stress conditions [7]. However, these findings remain speculative for human clinical outcomes. Notably, while animal studies highlight broader metabolic and protective roles for B vitamins, only the methylation/homocysteine pathway shows consistent, large-scale effects in human trials. Other hypothesized outcomes, such as cognitive enhancement or disease prevention, have not been replicated in large RCTs or meta-analyses.
The convergence of biochemical mechanism, clinical trial data, and biomarker outcomes makes the homocysteine pathway the most actionable and evidence-based target for B-complex supplementation. Table 2 summarizes the evidence strength for various B-complex mechanisms:
| Mechanism | Human RCTs/Meta-analyses | Animal/Cell Models | Evidence Strength (Humans) | |----------------------------|--------------------------|--------------------|----------------------------| | Homocysteine lowering | Yes | Yes | High | | Cognitive enhancement | No | Yes | Low | | Antioxidant defense | No | Yes | Low | For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
The primary, evidence-backed mechanism for B-complex supplementation is the lowering of homocysteine through direct support of methylation pathways. Large, well-controlled human trials confirm that providing adequate amounts of folic acid, B12, and B6 can consistently lower homocysteine by 2–4 μmol/L, which is enough to move many people into optimal biomarker ranges. This effect is independent of baseline status or responder type and is achieved with standard supplement forms and doses. While the broader clinical implications (such as direct cardiovascular or cognitive benefit) remain uncertain, the homocysteine-lowering effect itself is robust, reproducible, and mechanistically specific. For individuals interested in methylation health or optimizing this biomarker, B-complex supplementation is a well-supported intervention. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
Although the homocysteine-lowering effect of B-complex vitamins is well established in human trials, evidence linking this reduction to improved clinical outcomes (such as cardiovascular events or cognitive decline) remains inconsistent. Most studies use synthetic forms and typical doses, so results may not generalize to every formulation or delivery method. Animal and mechanistic studies suggest broader roles for B vitamins, but these hypotheses require further validation in humans. Finally, the optimal homocysteine range for all individuals is still debated, and not everyone may benefit equally from supplementation. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how vitamin b complex relates to your health goals and monitor changes in your biomarkers over time.
