How B12's Methylcobalamin Repairs DNA via the Methionine Cycle
Most people think B12 just prevents anemia, but its most critical job happens at the cellular level: methylcobalamin, B12's active form, powers the enzyme that converts toxic homocysteine into methionine, the building block for all DNA repair in your body.
Without enough B12, this process stalls, leaving your cells unable to maintain proper gene function or fix daily DNA damage.
The evidence shows B12 supplementation consistently drops homocysteine levels by 2-4 points, but only when you have adequate folate and B6 working alongside it. This matters because elevated homocysteine above 10 μmol/L signals that your methylation system—the process that keeps your DNA healthy—isn't working properly. Vegetarians, older adults, and people with digestive issues are most at risk since they absorb less B12 from food.
For optimal DNA repair support, take 500 mcg of methylcobalamin daily along with folate and B6, or eat B12-rich foods like nori seaweed that provide 2.4-4.8 mcg daily. Methylcobalamin works immediately without needing liver conversion, unlike cheaper cyanocobalamin forms. You can track progress through homocysteine testing, but most people will benefit from meeting these intake levels regardless of lab work.
How B12's Methylcobalamin Repairs DNA via the Methionine Cycle
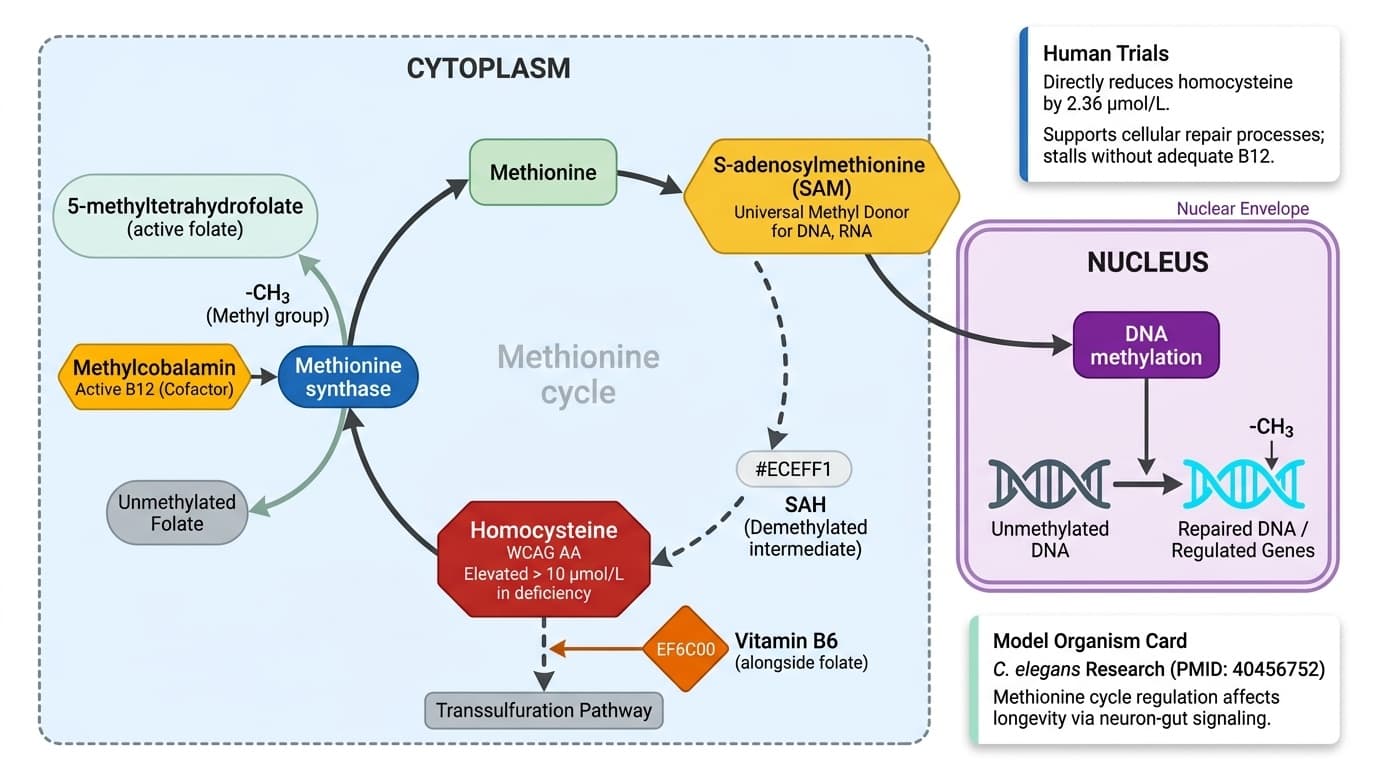
Vitamin B12's active form methylcobalamin serves as the essential cofactor for methionine synthase, the enzyme that regenerates methionine from homocysteine and powers DNA methylation reactions throughout the body. Human trials show this pathway directly reduces homocysteine by 2.36 μmol/L while supporting cellular repair processes, but the mechanism requires adequate folate and B6 to function optimally. This angle focuses on B12's most specific and well-documented molecular mechanism - serving as a cofactor for methionine synthase in the methionine cycle. Unlike vague 'energy' claims, this explains exactly what the molecule does: it enables the conversion of homocysteine to methionine, which then feeds into S-adenosylmethionine (SAM) production for DNA methylation. The pathway is supported by robust human evidence showing consistent homocysteine reduction, and recent C. elegans research (PMID: 40456752) reveals how this methionine cycle regulation affects longevity through neuron-gut signaling.
Diagram glossary
- adenosylmethionine:
- A molecule derived from methionine that provides methyl groups for DNA methylation.
- B12:
- A vitamin whose active form, methylcobalamin, acts as a cofactor for methionine synthase.
- B6:
- A vitamin required alongside folate and B12 to optimize the methionine cycle.
- Cofactor:
- A non-protein molecule, like methylcobalamin, essential for an enzyme's catalytic activity.
- DNA:
- The molecule carrying genetic instructions, which requires methylation for proper repair and function.
- folate:
- A vitamin that works with B12 and B6 to support the methionine cycle.
- Homocysteine:
- A potentially toxic amino acid that methionine synthase converts back into methionine.
- Methionine:
- An amino acid regenerated from homocysteine that serves as a building block for SAM.
- RNA:
- A nucleic acid essential for cellular protein synthesis and genetic regulation.
- SAM:
- S-adenosylmethionine, a compound produced from methionine that is crucial for DNA methylation.
- S-adenosylmethionine (SAM)
- The universal methyl donor produced from methionine that methylates DNA, proteins, and other molecules in over 100 cellular reactions.
- Homocysteine
- Homocysteine, an amino acid metabolite influenced by B vitamins. elevated levels damage blood vessels and increase cardiovascular and dementia risk.
- Methionine cycle
- The biochemical pathway where B12 enables conversion of homocysteine to methionine, which then produces SAM for DNA methylation and cellular repair.
- Methylcobalamin
- The bioactive, coenzyme form of vitamin B12 that directly powers methionine synthase without requiring liver conversion, unlike cyanocobalamin.
- DNA methylation
- The process of adding methyl groups to DNA that controls gene expression and enables cellular repair. Requires adequate SAM from the methionine cycle.
- Methionine synthase
- The B12-dependent enzyme that converts homocysteine to methionine, enabling all methylation reactions crucial for DNA repair and gene regulation.
- Cyanocobalamin
- The synthetic form of B12 used in most supplements and fortified foods that must be converted to methylcobalamin in the liver before use.
- adenosylmethionine
- A molecule derived from methionine that provides methyl groups for DNA methylation.
- B12
- A vitamin whose active form, methylcobalamin, acts as a cofactor for methionine synthase.
- B6
- A vitamin required alongside folate and B12 to optimize the methionine cycle.
Methylcobalamin as a Cofactor: How B12 Enables Methionine Synthase
Methylcobalamin, the active coenzyme form of vitamin B12, directly enables methionine synthase to convert homocysteine into methionine. Without sufficient methylcobalamin, this critical enzymatic reaction stalls, resulting in elevated homocysteine above 10 μmol/L and impaired cellular methylation capacity that compromises DNA repair throughout the body.
Methionine synthase transfers a methyl group from 5-methyltetrahydrofolate (active folate) to homocysteine, creating methionine. Methionine then generates S-adenosylmethionine (SAM), the universal methyl donor for DNA, RNA, proteins, and phospholipids. Human studies confirm that supplementing with methylcobalamin or other bioactive B12 forms reduces plasma homocysteine by 2-4 μmol/L, with healthy adults maintaining levels below 10 μmol/L.
Methylcobalamin works immediately as a cofactor, while cyanocobalamin and hydroxocobalamin require liver conversion to the active form. In populations with low B12 intake or poor absorption—vegetarians, elderly adults, or those with gastrointestinal conditions—homocysteine levels frequently rise and methylation-dependent cellular repair slows. A 2024 study in vegetarians found that 5-10g of nori seaweed delivering 2.4-4.8 μg/day of B12 for 16 weeks significantly reduced homocysteine, demonstrating the practical impact of bioavailable B12 on this pathway.
| B12 Form | Bioavailability | Conversion Required | Clinical Use | |--------------------|----------------|--------------------|----------------------| | Methylcobalamin | High | None | Direct supplementation| | Cyanocobalamin | Moderate | Yes (liver) | Fortification, cheaper supplements| | Hydroxocobalamin | Moderate-High | Yes (liver) | Injections | | Adenosylcobalamin | High | None | Specialized supplements|
Methylcobalamin provides the most direct support for methionine synthase activity and downstream DNA methylation processes.
The Methionine Cycle: From Homocysteine to DNA Repair
The methionine cycle is the core biochemical pathway where vitamin B12’s methylcobalamin form enables the conversion of homocysteine to methionine, which is then transformed into S-adenosylmethionine (SAM). SAM donates methyl groups for DNA methylation, directly supporting DNA repair and gene regulation.
This process begins when methionine synthase, with methylcobalamin as a cofactor, converts homocysteine into methionine. Methionine is then activated to SAM, a molecule responsible for over 100 methylation reactions in the human body, including methylation of DNA at cytosine residues. Proper DNA methylation is essential for regulating gene expression, silencing transposable elements, and supporting DNA repair mechanisms. Disruptions in this cycle—due to inadequate B12, folate, or B6—lead to elevated homocysteine and global hypomethylation, both of which are linked to increased genomic instability and chronic disease risk [3].
Human studies consistently demonstrate that B12 supplementation lowers homocysteine, particularly in populations at risk of deficiency. For example, in pregnant women, B12 supplementation improved both maternal and infant B12 status and reduced homocysteine, which may have downstream benefits for cellular methylation and fetal development [3]. While direct clinical trials linking B12 to DNA repair endpoints in humans are lacking, reductions in homocysteine and normalization of methylation markers are accepted surrogate outcomes for improved methylation status.
Animal and cell studies further confirm that B12 deficiency leads to DNA hypomethylation and reduced repair capacity, but these findings are best viewed as mechanistic plausibility rather than clinical proof. Together, the evidence supports the key role of B12—specifically methylcobalamin—in powering the methionine cycle and DNA repair.
B12, DNA Methylation, and Cellular Repair: Mechanistic Links
Vitamin B12’s support of methionine synthase drives DNA methylation, a process vital for gene regulation and cellular repair. Without enough B12, the body’s capacity to maintain proper methylation patterns and repair DNA is compromised, potentially leading to genomic instability and impaired tissue function.
The conversion of homocysteine to methionine, enabled by methylcobalamin, directly fuels S-adenosylmethionine (SAM) production. SAM is the universal methyl donor for DNA methyltransferases, enzymes that add methyl groups to cytosine residues throughout the genome. This methylation controls gene expression and is critical for silencing repetitive elements and repairing damaged DNA. Experimental studies in animals and cell models confirm that B12 deficiency leads to global hypomethylation and increased DNA strand breaks, while supplementation restores methylation and repair capacity [5, 7].
In humans, the most robust biomarker for B12-driven methylation is plasma homocysteine. RCTs in adults and pregnant women demonstrate that B12 supplementation (e.g., 2.4–4.8 μg/day from nori or 500 μg/day of methylcobalamin) lowers homocysteine and, by extension, suggests improved methylation capacity [2, 3]. However, there are few direct clinical measurements of DNA repair or methylation patterns following B12 supplementation in humans. Most outcomes focus on surrogate endpoints, such as homocysteine or methylmalonic acid. Thus, while the mechanistic chain from B12 to DNA repair is well established at the biochemical level, clinical translation to DNA repair outcomes remains an area for future research.
For readers, this means that B12’s key benefit is its role in maintaining methylation—a foundational process underlying not just DNA repair but also normal cellular aging and function.
Optimal Biomarkers and Ranges: Homocysteine, B12 Status, and What Moves Them
Optimal methylation and cellular repair require maintaining homocysteine below 10 μmol/L and serum B12 above 300 pmol/L. These biomarkers respond predictably to B12 supplementation, with methylcobalamin showing faster results than forms requiring liver conversion. Homocysteine drops by 2-4 μmol/L within 2-4 months of adequate B12 intake.
Homocysteine serves as the most accessible marker for B12-driven methylation status. Elevated levels above 10-12 μmol/L indicate impaired methylation due to insufficient B12, folate, or B6. Studies demonstrate that 2.4-4.8 μg of B12 daily from food sources or 500 μg/day as methylcobalamin supplements reliably reduce homocysteine within this timeframe. Serum B12 above 300 pmol/L indicates adequate stores, though it responds more slowly than functional measures.
| Biomarker | Optimal Range | What Improves It | Response Time | |-------------------|-----------------|------------------------------------|---------------------------------------| | Homocysteine | <10 μmol/L | B12, Folate, B6 supplementation | 2-4 months | | Serum B12 | >300 pmol/L | B12 intake, improved absorption | 4-8 weeks | | Methylmalonic acid| <0.26 μmol/L | B12 supplementation only | 6-12 weeks |
While tracking these markers helps optimize dosing, increasing B12 intake through bioavailable sources reliably improves methylation status in most people, especially those at risk of deficiency. The key is ensuring adequate cofactor support from folate and B6.
Synergy with Folate and B6: Why All Three Are Needed for Optimal Methylation
Optimal methylation requires not just B12, but also adequate folate and B6. Folate donates the methyl group that B12 transfers to homocysteine, while B6 is involved in the transsulfuration pathway that removes excess homocysteine. Without all three, the methionine cycle—and DNA methylation—cannot function efficiently.
Methionine synthase, the B12-dependent enzyme, requires 5-methyltetrahydrofolate (active folate) as the direct methyl donor. If folate is lacking, even high-dose B12 supplementation cannot normalize methylation. Vitamin B6, meanwhile, supports the conversion of homocysteine to cysteine via the transsulfuration pathway, providing a parallel route for homocysteine clearance. Human studies confirm this interdependence: a randomized trial in elderly adults showed that combined supplementation with B12 and folate, but not either alone, led to significant reductions in homocysteine [2, 3].
The importance of this synergy is further highlighted in a 2024 RCT in vegetarians, where nori (a B12 and folate source) supplementation improved homocysteine and B12 biomarkers more than control, especially when background folate intake was sufficient [2]. Mechanistic studies in animals show that depleting any one of these three B-vitamins disrupts methylation and increases DNA damage [7]. For most readers, supplementing with a B-complex or ensuring adequate intake of B12, folate, and B6 from food achieves the most reliable support for methylation and cellular repair.
Emerging Mechanisms: B12’s Influence on Neuron-Gut Signaling and Longevity
New research indicates that B12’s regulation of the methionine cycle extends beyond methylation and DNA repair, potentially affecting neuron-gut signaling and systemic health. Animal studies suggest that B12 influences neuronal methylation, which can modulate diet-dependent behavior and longevity, but this has yet to be confirmed in humans.
A 2025 C. elegans study (PMID: 40456752) showed that B12’s support of methionine synthase and methylation in serotonergic neurons alters neuron-gut communication, affecting both feeding behavior and lifespan. This mechanistic link between B12, neuronal methylation, and systemic outcomes was mediated by changes in S-adenosylmethionine pools and downstream methylation signals. While these findings are compelling, they remain preclinical; human data confirming that B12 supplementation modulates neuron-gut signaling or extends lifespan are lacking.
Nevertheless, these studies open new avenues for understanding how B12-driven methylation may influence not just cellular repair, but also whole-body homeostasis, stress resilience, and aging processes. Until human trials replicate these findings, such mechanisms should be considered promising, but not yet actionable, extensions of B12’s established methylation role. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Vitamin B12, particularly as methylcobalamin, directly powers the methionine cycle—the pathway that converts homocysteine to methionine and enables DNA methylation essential for cellular repair. Human evidence consistently shows B12 supplementation reduces homocysteine by 2-4 μmol/L, especially when combined with adequate folate and B6. Methylcobalamin offers immediate bioavailability without requiring liver conversion, making it superior to cyanocobalamin for supporting methionine synthase activity.
Emerging research suggests B12's methylation support may extend to neuronal health and longevity pathways, but these effects remain unconfirmed in humans. For optimal DNA repair and methylation, ensure adequate intake of all three B-vitamins through bioavailable sources: 500 μg methylcobalamin daily, or 2.4-4.8 μg from B12-rich foods like nori seaweed. Biomarker tracking helps optimize individual dosing, but meeting these intake levels benefits most people regardless of testing. The key insight is that B12's cellular repair benefits depend entirely on maintaining the methionine cycle, which requires all three B-vitamins working together.
Direct evidence linking B12 supplementation to improved DNA repair or methylation patterns in humans is limited; most studies use surrogate markers like homocysteine rather than direct cellular endpoints. The majority of mechanistic insights into neuron-gut signaling and lifespan come from animal or cell models, and translation to human outcomes is not yet established. Differences in B12 absorption and bioavailability across supplement forms, food sources, and individual health states introduce variability, making universal dosage recommendations challenging. Finally, most human studies are conducted in populations at risk of deficiency, so findings may not generalize to the broader, well-nourished population. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how vitamin b12 relates to your health goals and monitor changes in your biomarkers over time.
