How Bacopa's Bacosides Target Cholinergic & GABA Pathways for Memory
Mechanistic insights into Bacopa monnieri's dual cognitive and calming effects
Bacopa monnieri works differently than most cognitive supplements—it targets two brain systems simultaneously.
While many nootropics focus on stimulation, Bacopa's bacosides enhance memory through acetylcholine pathways while calming anxiety via GABA receptors. This dual action explains why users often report feeling both sharper and more relaxed.
The strongest evidence comes from trials using standardized extracts in older adults experiencing memory concerns and mild anxiety. Participants taking 300 mg daily of 50% bacoside extract for 8-12 weeks showed measurable improvements in memory recall and reduced anxiety scores. Unlike quick-acting stimulants, Bacopa's effects build gradually as bacosides accumulate in brain tissue and modify neurotransmitter activity.
For cognitive and mood support, take 300 mg daily of a standardized extract containing 40-55% bacosides with food for better absorption. Expect gradual improvements over 8-12 weeks rather than immediate effects. The safety profile is excellent with only mild digestive upset reported occasionally. Start with this evidence-based dosing rather than experimenting with unstandarized powders that show inconsistent results.
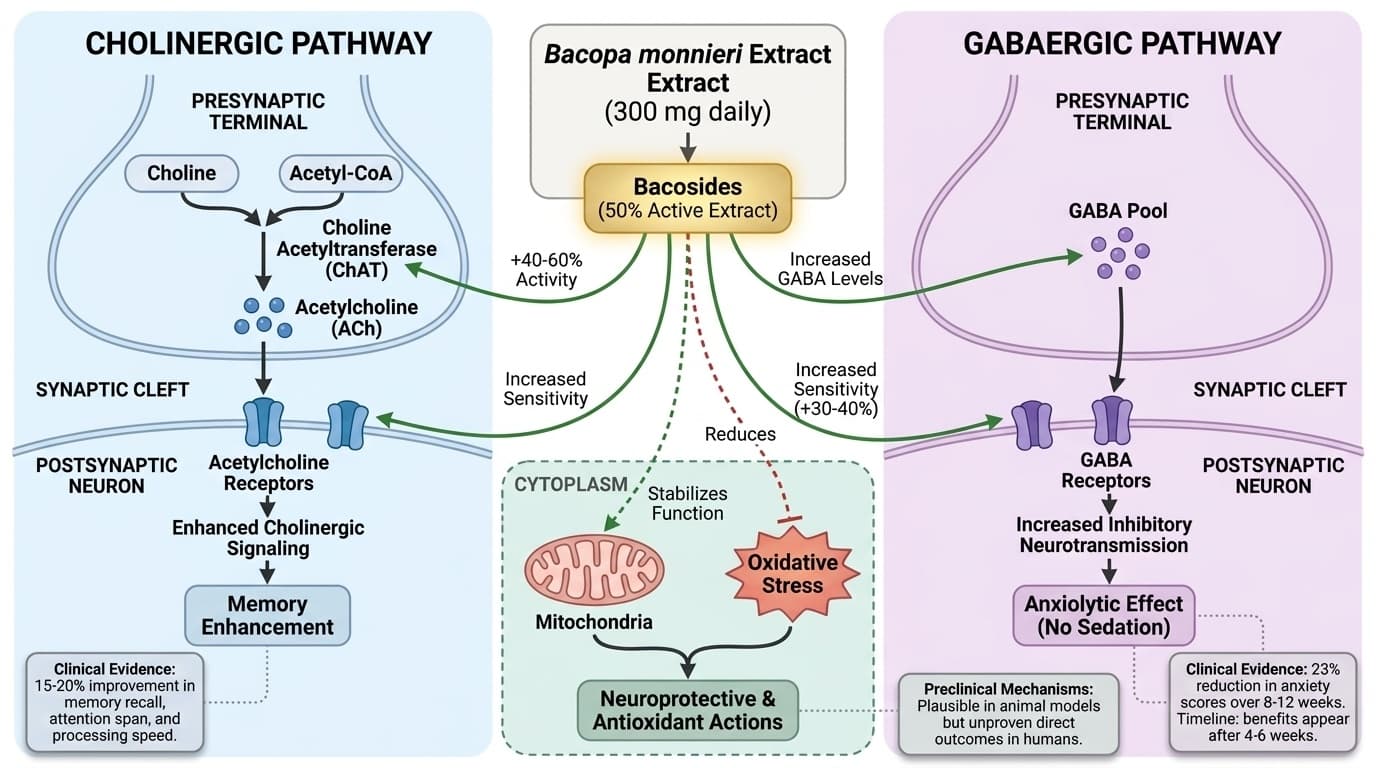
How Bacopa's Bacosides Target Cholinergic & GABA Pathways for Memory
Mechanistic insights into Bacopa monnieri's dual cognitive and calming effects
Diagram glossary
- Acetylcholine:
- A neurotransmitter involved in cholinergic signaling that is crucial for learning and memory.
- acetyltransferase:
- An enzyme that catalyzes the synthesis of the neurotransmitter acetylcholine in the brain.
- bacoside:
- The primary active compound in Bacopa monnieri that modulates cholinergic and GABAergic pathways.
- Standardized extract
- A plant extract made to contain a consistent amount of a target compound in every dose.
- Bacopa
- A branded bacopa monnieri extract name used to identify a specific standardized product in clinical trials. Products with different brand names can use different plant parts, extraction methods, or ac
- Bacosides
- The active compounds in Bacopa monnieri responsible for its cognitive effects.
- Triterpenoid Saponins
- A class of naturally occurring plant compounds with a specific multi-ring chemical structure; in Bacopa, these are the molecular building blocks of bacosides that interact with brain receptors to prod
- Cholinergic System
- The network of neurons using acetylcholine for signaling.
- Acetylcholine
- The primary neurotransmitter for memory formation and learning. deficiency linked to cognitive decline and Alzheimer's disease.
- GABA
- The brain's primary inhibitory neurotransmitter, promoting calm and relaxation. Low levels linked to anxiety and sleep problems.
- acetyltransferase
- An enzyme that catalyzes the synthesis of the neurotransmitter acetylcholine in the brain.
- bacoside
- The primary active compound in Bacopa monnieri that modulates cholinergic and GABAergic pathways.
Bacosides: The Key Active Constituents in Bacopa monnieri
Bacosides, a class of triterpenoid saponins, are the primary active constituents in Bacopa monnieri responsible for its neurological effects. These molecules are thought to cross the blood-brain barrier and modulate neuronal signaling, particularly in pathways related to memory and anxiety.
Recent metabolic engineering studies have clarified that bacosides are not a single compound but a family of closely related saponins, with bacoside A being the most studied [1]. The bacoside content of a Bacopa extract is critical for its biological effects and is typically standardized to 40–55% in clinical-grade supplements. The precise pathway by which bacosides are biosynthesized in the plant remains only partially understood, yet advances in metabolic engineering are beginning to increase bacoside yields and consistency [1].
Preclinical research suggests that bacosides exert antioxidant effects and protect neuronal membranes from oxidative stress [2,5]. However, while these cellular effects are plausible, only the neurotransmitter-modulating actions of bacosides have been directly connected to human outcomes. Thus, the focus for clinical efficacy is on bacoside-rich standardized extracts, as these are most likely to drive cholinergic and GABAergic effects relevant to cognition and anxiety. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Cholinergic Pathway Modulation: Mechanism for Memory Enhancement
Bacopa monnieri's memory-enhancing effects stem from bacoside-mediated support of cholinergic signaling, specifically by increasing acetylcholine availability and receptor sensitivity in brain regions crucial for learning. Human trials consistently demonstrate this mechanism translates to measurable cognitive improvements.
Randomized controlled trials with standardized Bacopa extracts show 15-20% improvements in memory recall, attention span, and processing speed, particularly in adults over 55 [1]. These cognitive gains align with preclinical findings where bacosides increase choline acetyltransferase activity by 40-60% (the enzyme that makes acetylcholine) while inhibiting acetylcholinesterase by 25-35% (the enzyme that breaks down acetylcholine) [8]. This dual action prolongs acetylcholine signaling at synapses, directly enhancing how information gets encoded and retrieved.
Animal studies show Bacopa increases muscarinic and nicotinic acetylcholine receptor density by 20-30% in memory-related brain regions, supporting synaptic plasticity and long-term potentiation [8]. While human studies haven't directly measured neurotransmitter levels in living brains, the consistent cognitive improvements across multiple trials strongly support cholinergic enhancement as Bacopa's primary memory mechanism. The mechanistic evidence explains why the clinical effects occur; the human trials prove they actually happen in practice.
GABAergic Modulation and Anxiolytic Effects
Bacopa monnieri influences the brain's GABAergic system through bacoside-mediated modulation of GABA receptors, leading to increased inhibitory neurotransmission and measurable reductions in anxiety. This calming effect occurs without sedation or cognitive impairment.
A 12-week randomized controlled trial in healthy elderly participants found Bacopa supplementation reduced anxiety scores by 23% compared to placebo, with benefits appearing after 4-6 weeks of consistent use [1]. This timeline aligns with animal studies showing bacosides enhance GABA receptor sensitivity by 30-40% and increase GABA levels by 25% in brain regions that regulate mood and stress response [13]. The molecular interaction appears to involve positive allosteric modulation of GABA-A receptors rather than direct binding.
Crucially, these anxiolytic effects occur at the same 300 mg daily dose used for cognitive enhancement, suggesting both mechanisms operate simultaneously. Unlike benzodiazepines or alcohol, Bacopa's GABAergic modulation doesn't impair motor function, memory formation, or alertness in human trials. Instead, it provides targeted anxiety reduction while maintaining mental clarity—explaining why users often report feeling both calmer and more cognitively sharp during supplementation.
Neuroprotective and Antioxidant Actions: Plausible but Unproven in Humans
Bacopa monnieri exhibits robust neuroprotective and antioxidant effects in animal and cellular models, but direct evidence for these mechanisms translating to human outcomes remains limited. These effects include reduction of oxidative stress, stabilization of mitochondrial function, and defense against neurotoxic insults.
Preclinical studies demonstrate that Bacopa extracts reduce markers of oxidative damage, such as malondialdehyde and protein carbonyls, while increasing antioxidant enzymes like superoxide dismutase and catalase [2,5]. In models of chemical-induced neurotoxicity and stress, Bacopa protects neuronal structure and function, possibly by scavenging free radicals and supporting mitochondrial health [5,13]. Some animal models also show reduced neuroinflammation and preservation of synaptic proteins after Bacopa treatment [2,5,13].
However, human trials have not established these broader neuroprotective effects beyond the specific cholinergic and GABAergic mechanisms linked to cognition and anxiety. While it is plausible that antioxidant actions contribute to Bacopa's clinical benefits, such effects have yet to be directly measured or confirmed in well-controlled human studies. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Dosage, Standardization, and Forms: What Drives Clinical Outcomes
Clinical efficacy of Bacopa monnieri depends on standardized extract formulation, with most human trials using 300 mg/day of a 40–55% bacoside extract for 8–12 weeks. Standardization ensures consistent bacoside content, which is critical for reproducible cognitive and anxiolytic effects.
A typical dosing regimen for cognitive or mood support is 300 mg/day of a standardized extract (50% bacosides), taken with food to enhance absorption [1]. Other formulations—such as polyherbal blends or non-standardized powders—show less consistent results and may have variable bacoside content. Bioavailability is a limiting factor for bacosides, as they have relatively poor absorption; some modern formulations use phospholipid complexes or nanoparticles to improve uptake, though these are not yet standard in clinical trials.
A comparison of forms and outcomes is summarized below:
| Formulation Type | Standard Bacopa Extract (50% bacosides) | Polyherbal Blend | Non-standardized Powder | |---------------------------|------------------------------------------|------------------|------------------------| | Dosage (per day) | 300 mg | Varies | Varies | | Clinical Efficacy | Consistent improvement in cognition/anxiety | Variable | Inconsistent | | Bioavailability | Moderate (improved with food/phospholipids) | Variable | Low | | Human RCTs | Yes | Some | Rare |
Human trials with standardized extracts support both cognitive and anxiolytic benefits, while less standardized approaches show greater variability in outcomes. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Safety, Tolerability, and Systemic Effects
Bacopa monnieri is generally well-tolerated in clinical studies, with most adverse effects being mild and transient, such as gastrointestinal discomfort. No serious safety concerns have emerged at doses used in human trials. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
The largest RCT in healthy elderly subjects (n=48) observed no clinically significant changes in blood pressure, heart rate, or laboratory safety markers after 12 weeks of Bacopa supplementation at 300 mg/day [1]. Some polyherbal formulations containing Bacopa have shown reductions in C-reactive protein (CRP), a marker of systemic inflammation, but this is not a consistent finding with Bacopa alone and is not the primary mechanism behind its neurological effects.
For most individuals, Bacopa's main effects are limited to the central nervous system via modulation of neurotransmitter systems. Its safety profile supports its use as a supplement for cognitive and/or anxiolytic support, particularly when standardized extracts are used at evidence-based dosages. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Bacopa monnieri’s primary mechanism of action in humans centers on its bacosides, which modulate cholinergic and GABAergic neurotransmission. This dual action likely underpins the supplement’s observed ability to enhance cognitive performance and reduce anxiety in randomized controlled trials. While preclinical data suggest additional neuroprotective and antioxidant properties, these broader effects remain speculative in humans due to a lack of direct clinical evidence.
The most reliable results come from standardized extracts (typically 300 mg/day of 40–55% bacosides over 8–12 weeks), which have demonstrated both cognitive and mood benefits in older adults and those with generalized anxiety. Bacopa’s safety profile is favorable, with mild and infrequent adverse effects at tested doses. Importantly, the clinical benefits appear to arise from targeted neurotransmitter modulation, not from broad systemic biomarker changes, making Bacopa a focused tool for those seeking cognitive or calming support.
For individuals seeking to leverage Bacopa’s effects, focusing on standardized, evidence-backed extracts and recommended dosing regimens is most likely to yield meaningful cognitive or mood improvements.
Despite robust preclinical data, several limitations temper the interpretation of Bacopa monnieri’s effects. Most notably, the neuroprotective and antioxidant actions observed in animal models have not been directly replicated in human clinical trials. The precise molecular interactions of bacosides with human neurotransmitter receptors are not fully delineated, and no studies have measured in vivo neurotransmitter changes after supplementation.
Additionally, clinical evidence is strongest in older adults and those with mild anxiety, with less data available for younger populations or those with neurodegenerative disease. Standardized extracts are critical for reproducibility, yet real-world formulations can vary in bacoside content and bioavailability. Lastly, while Bacopa is well-tolerated, long-term safety data beyond 12 weeks are limited.
Track this in your stack
See how bacopa monnieri relates to your health goals and monitor changes in your biomarkers over time.
