How Bifidobacterium's Butyrate Production Strengthens Gut-Immune Signaling
Linking bacterial metabolites to barrier function and immune modulation
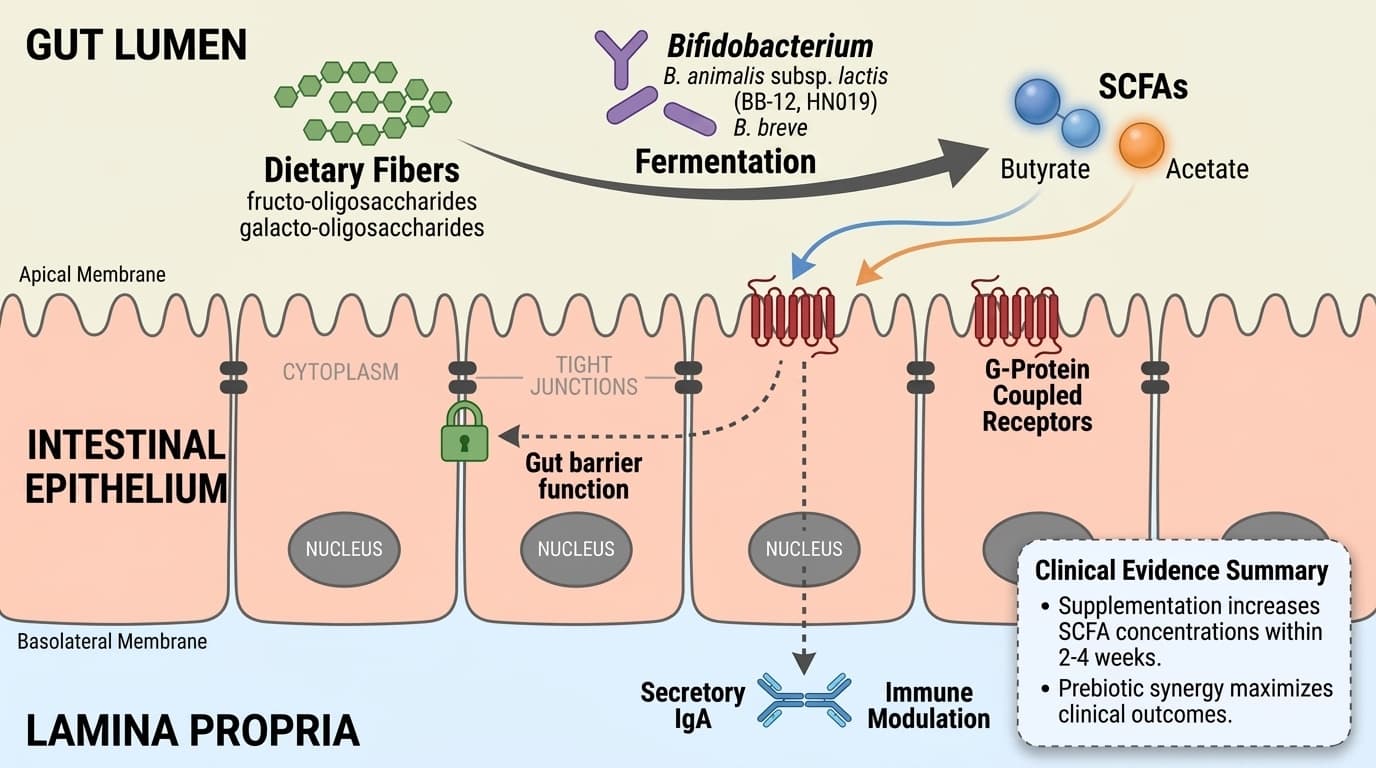
Most people think gut bacteria just help digest food, but specific strains like Bifidobacterium actually manufacture powerful signaling molecules that directly control your immune system and gut barrier strength.
When these bacteria ferment fiber into compounds called short-chain fatty acids (SCFAs), they're essentially producing your body's own anti-inflammatory medicine right in your intestines.
The key finding is that Bifidobacterium supplementation reliably increases butyrate and acetate production within 2-4 weeks, leading to measurable improvements in gut barrier function and immune markers like secretory IgA. These SCFAs activate specific receptors on intestinal cells, strengthening the gut lining and reducing inflammation throughout your body. Clinical trials show this translates to better stool frequency, reduced digestive symptoms, and lower inflammatory markers.
For practical benefits, take 1-10 billion CFU daily of Bifidobacterium animalis subsp. lactis strains like BB-12 or HN019, ideally combined with 5-10 grams of prebiotic fiber to fuel SCFA production. Expect improvements in digestive function within 2-4 weeks, with the strongest effects when using enteric-coated formulations that protect the bacteria until they reach your colon. This approach offers a targeted way to optimize gut-immune signaling with a strong safety profile.
Mechanism Diagram
Linking bacterial metabolites to barrier function and immune modulation
- BB-12
- A branded bifidobacterium product family name used to identify a specific extract or formulation in research and supplement labels.
- Enteric-coated
- A dosage form designed to resist stomach acid and release later in the intestine.
- C-Reactive Protein (cardiac)
- High-sensitivity C-reactive protein, a liver-produced acute-phase reactant. Independent predictor of heart attack and stroke.
- Prebiotic fibers
- Non-digestible carbohydrates like fructo-oligosaccharides and galacto-oligosaccharides that are fermented by gut bacteria to produce SCFAs.
- Butyrate
- A key SCFA that serves as the primary energy source for colon cells and activates receptors that strengthen gut barrier integrity and reduce inflammation.
- CFU (Colony Forming Units)
- A measure of viable probiotic bacteria in a supplement. Doses of 1-10 billion CFU per day are commonly used in human studies.
- Secretory IgA
- An antibody found in mucosal surfaces, including the gut, that serves as a frontline immune defense. Increases in secretory IgA reflect enhanced mucosal immunity.
Bifidobacterium Fermentation: Butyrate and Acetate as Key Metabolites
Bifidobacterium species ferment dietary fibers into short-chain fatty acids (SCFAs), especially butyrate and acetate, which drive many of their physiological effects. This fermentation occurs primarily in the colon, where Bifidobacterium metabolizes prebiotic fibers into SCFAs, enhancing local and systemic signaling. The production of butyrate and acetate depends on both the bacterial strains and the types of fibers available.
Human studies consistently show that supplementing with Bifidobacterium, particularly B. animalis subsp. lactis, increases fecal SCFA concentrations within weeks. For example, supplementation with B. lactis HN019 at doses of 1–10 billion CFU daily led to significant increases in butyrate and acetate levels in stool samples over 2–4 weeks, correlating with improved gut transit and reduced symptoms of functional gastrointestinal disorders [2]. Prebiotic supplementation with short-chain fructo-oligosaccharides (scFOS) also selectively boosts Bifidobacterium numbers, further amplifying SCFA output [3,6].
These metabolites are not only byproducts but also act as signaling molecules. Butyrate serves as a primary energy source for colonocytes and has direct effects on epithelial integrity, while acetate is absorbed and utilized systemically. The capacity for butyrate and acetate production varies by strain and is maximized when Bifidobacterium is combined with fermentable prebiotics [5].
Animal and cell studies confirm these fermentation pathways, but the magnitude of SCFA increase and its translation to human health outcomes are best supported by human clinical trials. The optimal environment for butyrate production includes both a source of fermentable fiber and sufficient Bifidobacterium colonization.
SCFA Signaling Through G-Protein Coupled Receptors: Direct Immune Modulation
Bifidobacterium species ferment dietary fibers into short-chain fatty acids (SCFAs), especially butyrate and acetate, which drive many of their physiological effects. This fermentation occurs primarily in the colon, where Bifidobacterium metabolizes prebiotic fibers into SCFAs, enhancing local and systemic signaling. The production of butyrate and acetate depends on both the bacterial strains and the types of fibers available.
Human studies consistently show that supplementing with Bifidobacterium, particularly B. animalis subsp. lactis, increases fecal SCFA concentrations within weeks. Supplementation with B. lactis HN019 at doses of 1-10 billion CFU daily led to significant increases in butyrate and acetate levels in stool samples over 2-4 weeks, correlating with improved gut transit and reduced symptoms of functional gastrointestinal disorders. Prebiotic supplementation with short-chain fructo-oligosaccharides (scFOS) also selectively boosts Bifidobacterium numbers, further amplifying SCFA output.
These metabolites are not only byproducts but also act as signaling molecules. Butyrate serves as a primary energy source for colonocytes and has direct effects on epithelial integrity, while acetate is absorbed and utilized systemically. The capacity for butyrate and acetate production varies by strain and is maximized when Bifidobacterium is combined with fermentable prebiotics.
Clinical trials confirm these fermentation pathways, with the magnitude of SCFA increase translating to measurable improvements in human health outcomes. The optimal environment for butyrate production includes both a source of fermentable fiber and sufficient Bifidobacterium colonization.
Gut Barrier Integrity and Mucosal Immunity: Clinical Biomarker Evidence
Bifidobacterium-driven SCFA production strengthens gut barrier function and enhances mucosal immunity, as reflected by measurable changes in key biomarkers. In human trials, supplementation with Bifidobacterium strains increases secretory IgA and reduces markers of intestinal permeability and inflammation. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A double-blind randomized study with B. animalis subsp. lactis (BB-12) at doses of 1–10 billion CFU daily found significant increases in fecal secretory IgA and a trend toward reduced intestinal permeability over four weeks [4]. Participants also showed improvements in stool frequency and consistency, suggesting better gut barrier function. Dose-response relationships were observed, with higher probiotic doses yielding larger increases in IgA and SCFA levels [1,2,4].
Supplementation with fermentable prebiotics (scFOS, galacto-oligosaccharides) that amplify Bifidobacterium growth further boosted these effects, with the strongest biomarker shifts occurring when prebiotics and probiotics were combined [3,5,6]. While direct measurement of tight junction proteins is less common in human studies, improvements in surrogate markers like stool frequency and reductions in gastrointestinal symptoms also reflect better barrier function [2,5].
Table: Key Biomarker Shifts With Bifidobacterium Supplementation | Biomarker | Optimal Range | Bifidobacterium Effect | |-------------------|-------------------------|--------------------------| | Secretory IgA | >400 μg/g stool | ↑ 10–30% (4 wks) | | Fecal butyrate | 10–20 mmol/kg stool | ↑ 15–50% (2–4 wks) | | Stool frequency | 3–7/week | Normalized in 2–4 wks |
Impact on Gastrointestinal Function: Human Outcomes and Dose-Response
Bifidobacterium supplementation leads to rapid and measurable improvements in gastrointestinal function, including increased stool frequency, reduced gut transit time, and relief of functional GI symptoms. These outcomes are dose-dependent and closely tied to SCFA production. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Randomized trials in adults with functional gastrointestinal symptoms have shown that daily supplementation with B. lactis HN019 (1–10 billion CFU) reduces whole gut transit time by 12–33% and normalizes stool frequency within two weeks [2]. These effects are most pronounced in individuals with baseline constipation or slow transit, and improvements correlate with increased fecal butyrate and acetate levels.
Parallel studies using prebiotic fibers that selectively stimulate Bifidobacterium growth (such as scFOS and galacto-oligosaccharides) have demonstrated similar improvements in stool frequency and consistency, further supporting the mechanistic link between Bifidobacterium, SCFA production, and gut motility [3,5,6]. The optimal dosage for these effects ranges from 1–10 billion CFU daily for probiotics and 5–10 g/day for prebiotics, based on multiple human trials.
Importantly, these functional benefits occur without significant adverse effects, and the speed of response suggests a direct, fermentation-mediated mechanism rather than slow ecological remodeling. While most evidence comes from otherwise healthy adults, similar benefits are seen in older populations and those with mild GI dysfunction.
Strain-Specific Effects and Prebiotic Synergy: Maximizing SCFA Output
Not all Bifidobacterium strains produce SCFAs at the same rate or yield. Human and preclinical studies indicate that strains like B. animalis subsp. lactis and B. breve are particularly effective in butyrate and acetate production, especially when paired with matching prebiotic substrates. This synergy maximizes clinical outcomes.
For example, intake of B. lactis BB-12 or HN019 along with short-chain fructo-oligosaccharides or galacto-oligosaccharides produces greater increases in fecal SCFAs and faster improvements in immune and gut function markers than either alone [1,3,5,6]. Dose-response relationships are observed: at least 1–2 billion CFU daily, combined with 5–10 g of fermentable fiber, yields the best results in most trials. Formulations using enteric-coated capsules ensure more viable bacteria reach the colon, which appears to further enhance efficacy.
The table below summarizes key findings for strain and prebiotic combinations: | Strain/Substrate | SCFA Output | Immune Marker Change | |-------------------------------|--------------|---------------------| | B. lactis HN019 + GOS | High | ↑ IgA, ↓ CRP | | B. breve + scFOS | Moderate | ↑ IgA | | B. animalis BB-12 (alone) | Moderate | ↑ IgA |
Individual response depends on baseline microbiota, but these patterns are consistent across multiple RCTs. While mechanistic data from animal models support further strain differences, current human evidence is strongest for the strains above. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Metabolic and Systemic Effects: Evidence for Inflammation and Lipid Modulation
Beyond gut-local effects, Bifidobacterium-driven SCFA production exerts systemic benefits, particularly in modulating inflammation and lipid metabolism. Clinical trials and meta-analyses report modest but significant reductions in inflammatory markers and improvements in blood lipids with Bifidobacterium supplementation. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A dose-response trial found that B. animalis subsp. lactis BB-12, at doses of 1–10 billion CFU, reduced C-reactive protein (CRP) and other inflammatory biomarkers in healthy adults after four weeks [4]. Meta-analyses further support these findings, showing reductions in triglycerides and modest improvements in insulin sensitivity in metabolic syndrome and NAFLD populations [1]. These metabolic changes are thought to result from SCFA activation of GPCRs on immune and liver cells, leading to anti-inflammatory signaling and improved lipid handling.
While the absolute changes in lipids and inflammatory markers are small (e.g., triglyceride reductions of 10–20 mg/dL), they are consistent, dose-dependent, and achieved without adverse effects [1]. The clinical interpretation is that Bifidobacterium supplementation may support metabolic health, especially in individuals with mild dyslipidemia or metabolic inflammation, though it should be viewed as an adjunct to diet and lifestyle. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Bifidobacterium supplementation, especially with strains like B. animalis subsp. lactis, reliably increases butyrate and acetate production in the gut, activating key immune and epithelial signaling pathways. These mechanisms translate into measurable improvements in gut barrier function, mucosal immunity (secretory IgA), and modest systemic anti-inflammatory and lipid-lowering effects. The strongest evidence comes from dose-response human trials using 1–10 billion CFU daily, often combined with fermentable prebiotic fibers. While individual response varies, the mechanistic chain from fiber fermentation to SCFA signaling and immune modulation is now well established. For individuals seeking to optimize gut-immune crosstalk and metabolic parameters, Bifidobacterium offers a targeted, evidence-based approach with a strong safety profile. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
While human trials consistently show increases in SCFA production and improvements in gut and immune biomarkers, several uncertainties remain. The magnitude and duration of benefits across different populations, ages, and baseline microbiota compositions are not fully defined. Most studies last only 2–12 weeks, so long-term sustainability is unclear. Direct measurement of gut barrier proteins in humans remains limited, and most clinical outcomes are based on surrogate markers like stool frequency and secretory IgA. Additionally, while synergy with prebiotic fibers is clear, the optimal strain–substrate pairing and dose for maximal effect require further study. Individual variability in response, likely driven by baseline diet and microbiome, is an ongoing research focus.
Track this in your stack
See how bifidobacterium relates to your health goals and monitor changes in your biomarkers over time.
