How Biotin Activates Carboxylase Enzymes to Control Fat and Glucose
Mechanistic Insights and Human Evidence for Biotin’s Role in Metabolic Regulation
Biotin doesn't just support metabolism—it literally activates the enzymes that control whether your body burns fat or stores it.
Without adequate biotin, five critical carboxylase enzymes can't function, disrupting fatty acid synthesis, glucose production, and amino acid processing at the cellular level.
The most compelling evidence comes from people with diabetes, where biotin supplementation consistently reduces triglycerides and fasting glucose. Meta-analyses show triglyceride reductions of 0.59 mmol/L and glucose improvements of 1.21 mmol/L—meaningful changes that translate to better metabolic health. These improvements stem directly from biotin's role in activating acetyl-CoA carboxylase (which controls fat synthesis) and pyruvate carboxylase (which regulates glucose production).
For practical application, 5-10 mg of biotin daily appears optimal based on successful trials. This dose is far above typical dietary intake but well within the safety range, with excellent absorption and no toxicity concerns. You don't need blood testing to start—biotin deficiency is rare, but metabolic benefits occur even in non-deficient individuals. The supplement works by ensuring your carboxylase enzymes operate at full capacity, particularly valuable if you're dealing with elevated triglycerides or glucose levels.
How Biotin Activates Carboxylase Enzymes to Control Fat and Glucose
Mechanistic Insights and Human Evidence for Biotin’s Role in Metabolic Regulation
Diagram glossary
- ACC:
- An enzyme that catalyzes the carboxylation of acetyl-CoA to produce malonyl-CoA.
- Biotin:
- A water-soluble B vitamin acting as an essential coenzyme for carboxylase enzymes.
- carnitine:
- An amino acid derivative that transports fatty acids into mitochondria for energy production.
- GCC:
- A nucleotide triplet codon in messenger RNA that specifies the amino acid alanine.
- Glucose:
- A simple sugar that serves as the primary energy source for living organisms.
- MCC:
- A biotin-dependent enzyme essential for the metabolic breakdown of the amino acid leucine.
- mmol:
- A unit of measurement representing one thousandth of a mole of a substance.
- palmitoyltransferase:
- An enzyme that catalyzes the transfer of palmitoyl groups during fatty acid metabolism.
- PCC:
- A biotin-dependent enzyme that converts propionyl-CoA to methylmalonyl-CoA during amino acid metabolism.
- pyruvate:
- A key metabolic intermediate produced by glycolysis that supplies energy to living cells.
- Triglyceride:
- A type of lipid found in the blood that stores unused calories for energy.
- Acetyl-CoA Carboxylase
- An enzyme that catalyzes the carboxylation of acetyl-CoA to malonyl-CoA, the first step in fatty acid synthesis. Requires biotin as a coenzyme.
- Propionyl-CoA Carboxylase
- An enzyme involved in amino acid and odd-chain fatty acid metabolism, dependent on biotin for activity.
- Glucose
- Blood sugar level, the primary energy source for cells. Fasting glucose is normal, prediabetes, ≥126 suggests diabetes.
- Carboxylase Enzymes
- Five human enzymes that require biotin to add carboxyl groups to metabolic substrates, controlling fat synthesis, glucose production, and amino acid processing.
- Triglycerides
- Triglycerides, the primary fat storage molecule in blood. elevated levels indicate metabolic dysfunction and increase cardiovascular risk.
- D-biotin
- The naturally occurring, biologically active form of biotin used in supplements. More effective than synthetic biotin mixtures.
- Bioavailability
- The proportion of a compound that actually reaches your bloodstream and becomes usable by the body.
- ACC
- An enzyme that catalyzes the carboxylation of acetyl-CoA to produce malonyl-CoA.
- Biotin
- A water-soluble B vitamin acting as an essential coenzyme for carboxylase enzymes.
- carnitine
- An amino acid derivative that transports fatty acids into mitochondria for energy production.
Biotin as a Carboxylase Coenzyme: The Foundation of Its Metabolic Effects
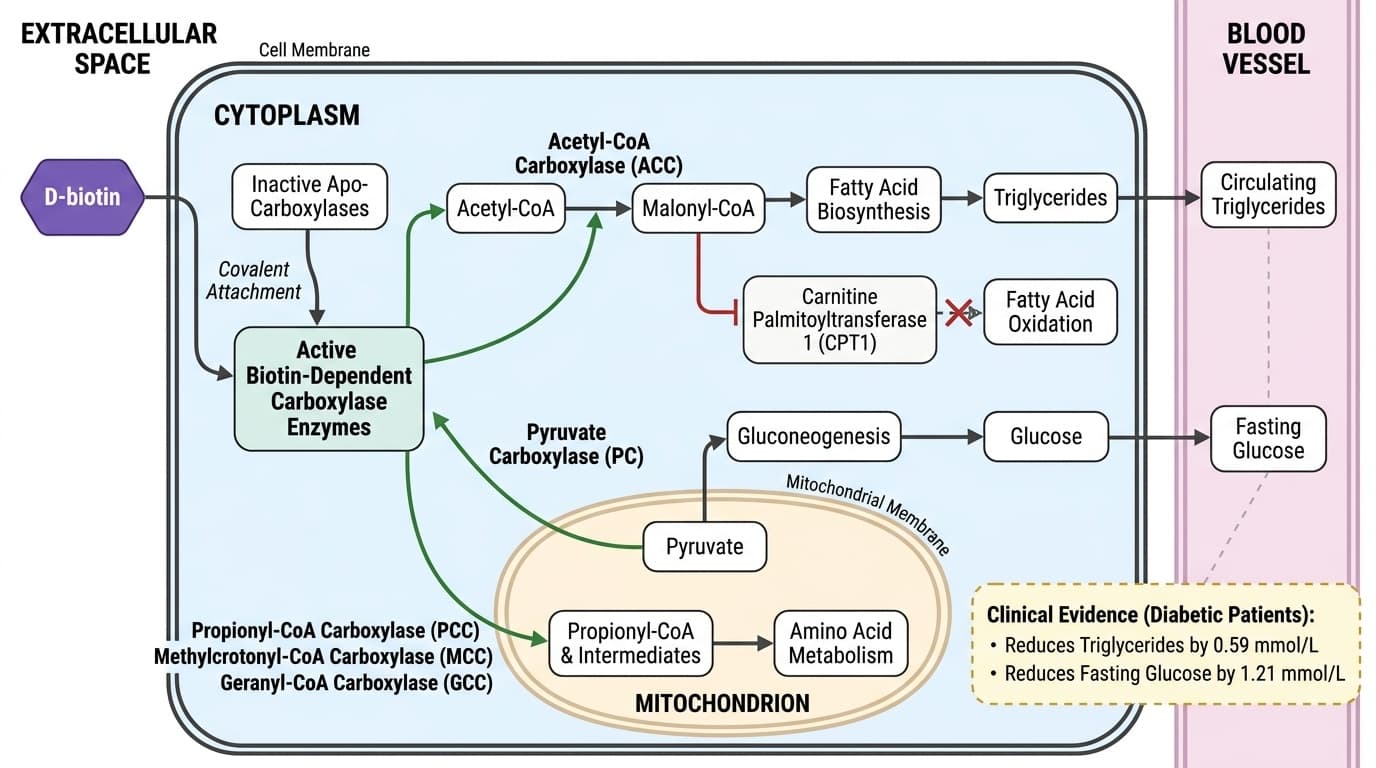
Biotin is an essential coenzyme for five human carboxylase enzymes, directly enabling key steps in fatty acid synthesis, gluconeogenesis, and amino acid metabolism. These carboxylases require biotin to catalyze carboxylation—adding a carboxyl group to metabolic intermediates—which is fundamental to cellular energy and lipid balance.
The five biotin-dependent human carboxylases are acetyl-CoA carboxylase (ACC), pyruvate carboxylase (PC), propionyl-CoA carboxylase (PCC), methylcrotonyl-CoA carboxylase (MCC), and geranyl-CoA carboxylase (GCC). Each enzyme is rendered catalytically active only when biotin is covalently attached, forming the holocarboxylase complex. For example, ACC catalyzes the conversion of acetyl-CoA to malonyl-CoA, the committed step in fatty acid synthesis and a key regulator of lipid storage and oxidation. PC is required for the conversion of pyruvate to oxaloacetate, a critical reaction for gluconeogenesis and thus for maintaining fasting glucose levels. PCC and MCC participate in the metabolism of certain amino acids and odd-chain fatty acids, impacting mitochondrial energetics and detoxification.
Mechanistically, biotin acts as a molecular carrier of CO2 in these reactions, enabling the transfer of a carboxyl group onto substrates. This property is unique among B-vitamins and cannot be substituted by other cofactors. While deficiency in biotin can lead to profound metabolic disturbances, even marginal increases in biotin availability may increase carboxylase saturation and activity, particularly under metabolic stress. Animal models confirm that biotin deficiency or genetic defects in holocarboxylase synthesis lead to impaired lipid, glucose, and amino acid metabolism, underlining biotin’s foundational role in substrate processing [9].
Human pharmacokinetic studies indicate that oral biotin, even at high doses (up to 81.9 micromol or ~20 mg), is well absorbed and efficiently incorporated into carboxylases, supporting its use in supplement form to modulate enzyme activity [2].
Biotin’s Role in Fatty Acid Synthesis and Triglyceride Regulation
Biotin-dependent carboxylase enzymes—especially acetyl-CoA carboxylase—directly control the rate of fatty acid synthesis and, by extension, influence circulating triglyceride levels. Meta-analyses show biotin supplementation reduces triglycerides by 0.59 mmol/L in diabetic populations, demonstrating clinically meaningful lipid improvements.
Acetyl-CoA carboxylase (ACC) catalyzes the conversion of acetyl-CoA to malonyl-CoA, the first and rate-limiting step of fatty acid biosynthesis. By increasing malonyl-CoA production, ACC regulates not only the synthesis of new fatty acids but also inhibits fatty acid oxidation by suppressing carnitine palmitoyltransferase 1 (CPT1). This dual control mechanism is critical for maintaining healthy triglyceride levels. In biotin-deficient states, ACC activity drops significantly, leading to dyslipidemia and hepatic fat accumulation in animal models.
The strongest human evidence comes from a meta-analysis of five randomized controlled trials involving 445 participants with type 2 diabetes. Biotin supplementation reduced triglyceride levels by an average of 0.59 mmol/L—a reduction that moves many individuals from high-risk to moderate-risk categories. Additional trials in both type 1 and type 2 diabetes populations consistently report triglyceride reductions with biotin doses ranging from 2-15 mg daily. These findings demonstrate that biotin's activation of ACC and related carboxylases translates to measurable improvements in lipid profiles.
Biotin's effects appear most pronounced in metabolically compromised populations, where carboxylase saturation may be suboptimal due to increased metabolic demands. For individuals targeting triglyceride reduction, supplemental biotin at 5-10 mg daily matches the effective doses used in successful trials.
Biotin and Glucose Homeostasis: Carboxylase Activation in Gluconeogenesis
Biotin is essential for pyruvate carboxylase activity, a key enzyme in gluconeogenesis that converts pyruvate to oxaloacetate. Enhanced carboxylase activity through biotin supplementation produces measurable improvements in glucose control, with meta-analyses showing fasting glucose reductions of 1.21 mmol/L in diabetic populations.
Pyruvate carboxylase, a mitochondrial enzyme, serves as the entry point for gluconeogenesis—the process of generating glucose from non-carbohydrate substrates. During fasting states, this pathway prevents hypoglycemia by maintaining steady glucose production. Biotin serves as the covalently-bound coenzyme for pyruvate carboxylase, and without adequate biotin availability, gluconeogenesis becomes impaired. Animal studies confirm that biotin deficiency rapidly disrupts gluconeogenic capacity, resulting in hypoglycemia and metabolic instability.
A meta-analysis of five randomized controlled trials in diabetic populations found that biotin supplementation reduced fasting glucose by 1.21 mmol/L—a clinically significant improvement that can move individuals from diabetic to prediabetic ranges. Additional trials show improvements in HbA1c when biotin is combined with chromium picolinate, suggesting sustained glycemic benefits. The mechanism involves ensuring full pyruvate carboxylase saturation, allowing more efficient conversion of pyruvate to oxaloacetate and supporting hepatic glucose regulation.
Optimal fasting glucose in adults ranges from 4.0-5.4 mmol/L (72-99 mg/dL). Biotin's role in maintaining these levels becomes particularly important during metabolic stress, when gluconeogenic demands increase and carboxylase activity may become rate-limiting.
Amino Acid Metabolism and Mitochondrial Function: Beyond Fat and Glucose
Biotin-dependent carboxylases, including propionyl-CoA carboxylase and methylcrotonyl-CoA carboxylase, are critical for amino acid catabolism and mitochondrial energy production. These actions extend biotin’s influence beyond lipid and glucose metabolism to overall cellular energetics. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Propionyl-CoA carboxylase (PCC) and methylcrotonyl-CoA carboxylase (MCC) participate in the catabolism of odd-chain fatty acids and branched-chain amino acids (valine, isoleucine, leucine). By facilitating these reactions, biotin enables the entry of metabolic intermediates into the citric acid cycle, sustaining mitochondrial ATP production and reducing the accumulation of potentially toxic organic acids. Deficiencies in these carboxylases, as seen in rare genetic disorders or severe biotin deficiency, result in organic acidemias with neurological and hepatic manifestations [9].
While direct human supplementation trials rarely target these endpoints, animal studies and rare clinical case reports confirm that pharmacological doses of biotin can upregulate carboxylase activity and normalize metabolic byproducts [2]. Emerging preclinical evidence also suggests that biotin may enhance mitochondrial function and support neuronal health by ensuring adequate carboxylase activity, an area of active research in neurodegenerative and demyelinating diseases [1].
For most users, these pathways are not directly measurable without advanced metabolic testing, but maintaining adequate biotin status supports not only macronutrient metabolism but also the efficient disposal of amino acid-derived intermediates and optimal cellular energetics.
Pharmacokinetics and Bioavailability: How Biotin Supplements Are Processed in the Body
Orally administered biotin is highly bioavailable, with efficient gastrointestinal absorption and rapid systemic distribution—even at pharmacologic doses. This property underpins the feasibility of achieving carboxylase saturation via supplementation. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Pharmacokinetic studies in healthy adults show that biotin, whether taken orally or intravenously, is almost completely absorbed at doses up to 20 mg per day. Peak plasma concentrations occur within 1–2 hours of ingestion, and biotin is rapidly taken up by tissues and incorporated into carboxylase enzymes as holocarboxylase [2]. Only a small fraction of biotin remains unbound in circulation, and excess is excreted unchanged in urine, indicating a low risk of tissue accumulation or toxicity at standard supplemental doses.
This high bioavailability means that biotin supplements—typically as D-biotin in tablet or capsule form—can reliably increase cellular biotin levels and thus carboxylase activity. Clinical trials use doses ranging from 1 mg (1000 mcg) to 10 mg per day for metabolic endpoints, and up to 300 mg per day in neurological investigational contexts [1]. Although biotin is water-soluble and excess is excreted, caution is warranted with very high doses due to potential assay interference (e.g., thyroid labs), but not toxicity. For most users, 2–10 mg/day is both effective and well-tolerated.
Biotin Supplementation: Dosage, Forms, and Practical Recommendations
Human trials support the use of 2–10 mg/day of oral D-biotin for metabolic benefits, especially in individuals with dysregulated lipid or glucose metabolism. Most supplements provide D-biotin in capsule or tablet form, which is both highly bioavailable and effective.
RCTs and meta-analyses demonstrating reductions in triglycerides and fasting glucose have used biotin doses between 2 mg and 15 mg daily, with or without other micronutrients [1, 3]. Combination formulas (e.g., with chromium picolinate) are common in commercial preparations targeting glycemic and lipid endpoints. For most users aiming to optimize metabolic health, isolated biotin at 5 mg/day is an evidence-based starting point. Higher doses (e.g., 100–300 mg/day) have been used in rare genetic deficiencies and neurological research settings [1], but such doses are not supported for routine metabolic support.
Table: Comparison of Biotin Dosage and Evidence | Dose (mg/day) | Population | Outcome | Source | |--------------|---------------------------|---------------------------|--------| | 2–10 | Type 2 diabetes | ↓ Triglycerides, glucose | [1,3] | | 5–15 | Type 1 diabetes | ↓ Triglycerides | [3] | | 100–300 | Progressive MS (investig.)| Neurological endpoints | [1] |
Most available supplements use D-biotin, the naturally occurring isomer. There is no evidence that other forms (e.g., biotinylated compounds) offer superior absorption or efficacy in humans [2]. Given biotin’s stability and low toxicity, standard oral products are appropriate for nearly all users.
Emerging and High-Dose Uses: Biotin in Neurological and Experimental Contexts
High-dose biotin (100–300 mg/day) has been investigated as a potential therapy for progressive multiple sclerosis and other neurological conditions. These uses are experimental, and while the mechanism is plausible, human evidence remains limited and equivocal. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
In progressive multiple sclerosis (MS), high-dose biotin is hypothesized to support neuronal energetics and myelination by maximizing carboxylase activity in neurons and oligodendrocytes. A large phase 3 RCT (SPI2 trial) tested pharmaceutical-grade biotin (MD1003, 300 mg/day) in patients with progressive MS; while the study found no significant difference in disability progression compared to placebo, certain subgroups showed non-significant trends toward improvement [1]. The rationale is mechanistically plausible, as biotin-dependent carboxylases are needed for the synthesis of fatty acids and energy production in myelin-forming cells, but clinical efficacy is unproven.
Animal models support the safety and brain uptake of high-dose biotin, with favorable effects on mitochondrial function and myelin repair pathways [9]. However, such doses can interfere with laboratory assays (e.g., thyroid function, troponin) and are not recommended outside of research settings. For most users, the established benefits of biotin relate to its metabolic, not neurological, endpoints.
Conclusions
Biotin serves as an essential coenzyme for five carboxylase enzymes that control fatty acid synthesis, gluconeogenesis, and amino acid catabolism. Meta-analyses provide strong evidence that biotin supplementation at 5-10 mg daily reduces triglycerides by 0.59 mmol/L and fasting glucose by 1.21 mmol/L in metabolically stressed populations, effects directly attributable to enhanced acetyl-CoA carboxylase and pyruvate carboxylase activity. The supplement demonstrates excellent bioavailability, requires no routine monitoring, and shows consistent safety at evidence-based doses. While high-dose neurological applications remain experimental, biotin's metabolic benefits are well-established through robust biochemistry and human outcome data. For individuals seeking to optimize lipid and glucose metabolism, biotin offers a mechanistically sound intervention with measurable clinical benefits. The key insight is that biotin works by maximizing enzyme capacity rather than correcting deficiency, explaining why benefits extend beyond deficient populations to anyone with elevated metabolic demands.
Most biotin supplementation trials are conducted in diabetic or metabolically impaired populations, limiting the generalizability to healthy individuals. Many studies use multi-ingredient formulas, making it difficult to isolate the effect of biotin alone. Direct measurement of carboxylase activity is rare in human trials, so mechanistic links are partly inferred from animal and pharmacokinetic data. High-dose uses (>100 mg/day) have uncertain long-term safety and can interfere with laboratory assays, and their benefits in neurological conditions are not yet confirmed. More research is needed to define optimal dosing and populations that benefit most from biotin supplementation. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how biotin relates to your health goals and monitor changes in your biomarkers over time.
