How Curcumin's Active Compounds Target Nrf2 to Boost Cellular Antioxidants
Curcumin, the primary active compound in turmeric, activates the Nrf2 transcription factor in human cells.
This activation signals the body to increase production of glutathione, superoxide dismutase, and catalase—the primary enzymes that defend against oxidative stress. That mechanism matters clinically.
Meta-analyses of randomized controlled trials show that curcumin, particularly in enhanced-absorption forms, consistently raises antioxidant enzyme levels and reduces markers of oxidative damage like malondialdehyde. The strongest evidence supports curcumin’s ability to induce endogenous antioxidant defenses through Nrf2 activation, not merely by acting as a direct antioxidant. However, the extent to which these biochemical changes translate to disease prevention remains less certain, as most trials focus on biomarker shifts, not clinical endpoints.
For those aiming to optimize antioxidant status, curcumin’s mechanism stands out because it helps the body ramp up its own defenses rather than simply neutralizing free radicals. Supplementing with an enhanced-bioavailability curcumin extract—typically 250-1,000 mg daily depending on the formulation—may support optimal glutathione and related enzyme levels, with benefits measurable even without laboratory testing. The practical reading is cautious: mechanism explains why the effect is plausible, while human outcome data decide how much weight the claim deserves. That matters for readers who track biomarkers and for readers who rely on symptoms, diagnosis context, or clinician-guided goals instead.
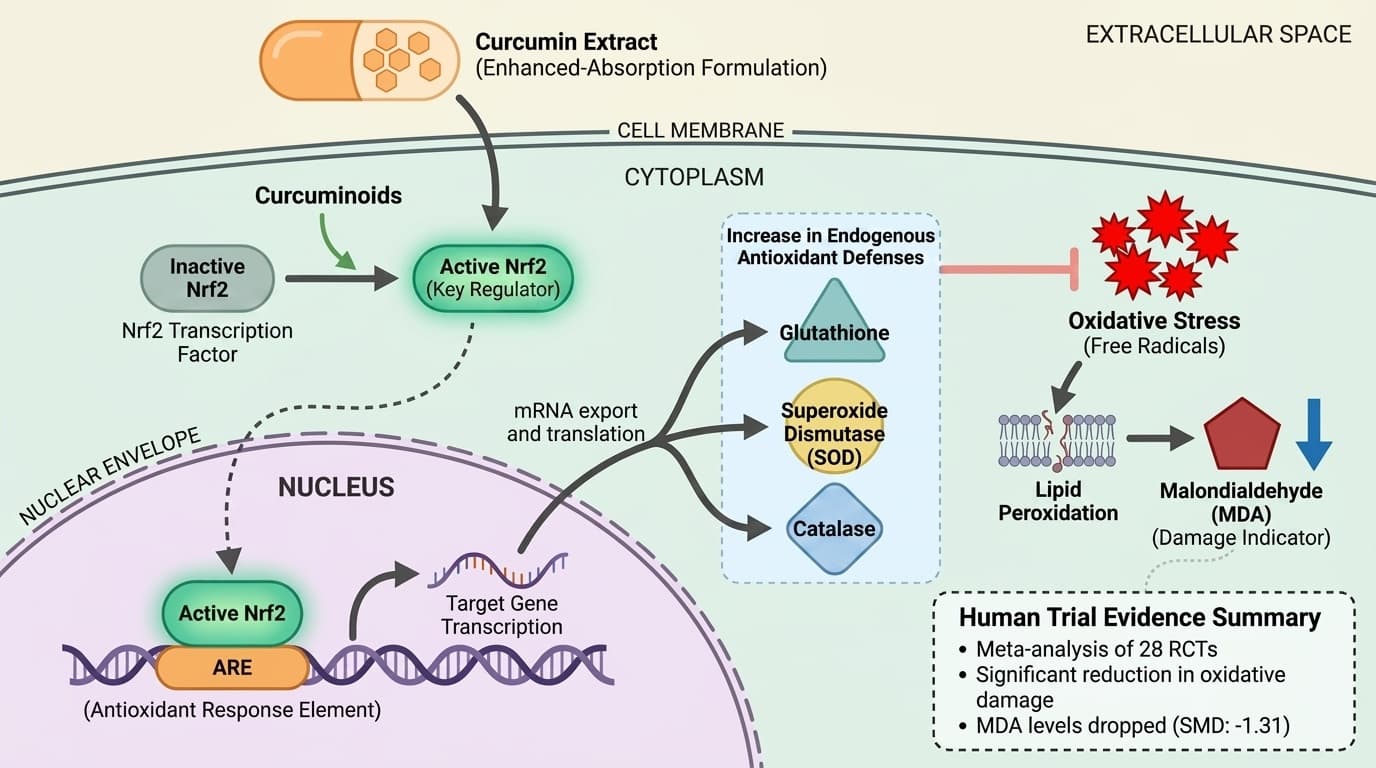
How Curcumin's Active Compounds Target Nrf2 to Boost Cellular Antioxidants
Curcumin contains curcuminoids that activate the Nrf2 transcription factor, triggering cells to produce more glutathione, superoxide dismutase, and catalase—the body's primary antioxidant enzymes. Meta-analyses of human trials show these effects translate to measurable reductions in oxidative damage markers like malondialdehyde, with the strongest evidence coming from studies using enhanced-absorption formulations. This angle identifies the specific molecular pathway (Nrf2-ARE signaling) that explains curcumin's most consistent human trial outcomes. Unlike vague 'anti-inflammatory' claims, this mechanism explains why curcumin specifically boosts endogenous antioxidants rather than just acting as a direct antioxidant. The pathway is diagram-friendly and separates proven antioxidant effects from speculative disease treatment claims.
Diagram glossary
- Catalase:
- A primary endogenous antioxidant enzyme that defends cells against oxidative stress.
- MDA:
- Malondialdehyde, a measurable biological marker indicating cellular oxidative damage.
- SOD:
- Superoxide dismutase, a primary endogenous antioxidant enzyme that protects against oxidative stress.
- superoxide:
- A reactive oxygen species neutralized by the antioxidant enzyme superoxide dismutase.
- Curcumin extract
- A concentrated form of curcumin derived from turmeric root, often processed to increase the proportion of active curcuminoids and improve absorption compared to raw turmeric powder.
- Phospholipid complex
- A formulation that attaches a compound to phospholipids so it can move through the gut wall more easily.
- Nrf2 Pathway
- A master switch for cellular antioxidant defenses.
- Catalase
- An enzyme that converts hydrogen peroxide into water and oxygen, protecting cells from oxidative damage. Upregulated by Nrf2 activation.
- Oxidative Stress
- Cellular damage caused by reactive oxygen species (free radicals) overwhelming antioxidant defenses.
- Bioavailability
- The proportion of a compound that actually reaches your bloodstream and becomes usable by the body.
- Nanocurcumin
- A formulation where curcumin is delivered as nanoparticles, significantly improving its absorption and systemic availability in the body.
- MDA
- Malondialdehyde, a measurable biological marker indicating cellular oxidative damage.
- SOD
- Superoxide dismutase, a primary endogenous antioxidant enzyme that protects against oxidative stress.
- superoxide
- A reactive oxygen species neutralized by the antioxidant enzyme superoxide dismutase.
Curcumin’s Core Mechanism: Nrf2 Activation and Antioxidant Enzyme Induction
Curcumin’s most consistent biological effect in humans is the activation of the Nrf2 transcription factor, which leads to increased cellular production of glutathione, superoxide dismutase (SOD), and catalase. This mechanism underlies curcumin’s ability to support the body’s own antioxidant defenses rather than acting as a direct scavenger of free radicals.
Nrf2 (nuclear factor erythroid 2–related factor 2) is a key regulator of the cellular antioxidant response. When activated, Nrf2 translocates to the nucleus and binds to the antioxidant response element (ARE) in DNA, upregulating genes for endogenous antioxidant enzymes and detoxification proteins. Preclinical research demonstrates that curcumin directly enhances Nrf2 nuclear translocation and increases the expression of these protective enzymes in various cell types and animal models [4][6].
In humans, this mechanism translates to measurable increases in glutathione and related enzyme activity after curcumin supplementation. Unlike many antioxidants that work by directly neutralizing free radicals, curcumin’s effect is more sustainable because it catalyzes the body’s own defenses. This distinction is supported by meta-analyses showing significant rises in glutathione and antioxidant enzymes in people taking curcumin, particularly in forms designed for better absorption [13][14][15].
The Nrf2 pathway is also linked to cellular resilience under stress, as seen in studies where curcumin enhanced Nrf2 activity in diabetic models and improved cellular resistance to oxidative damage [4]. However, while the mechanistic link is strong, not all downstream health outcomes have been conclusively proven in human clinical settings.
Human Trial Evidence: Curcumin Reduces Oxidative Damage Markers
Human randomized controlled trials consistently show that curcumin supplementation lowers markers of oxidative stress, notably malondialdehyde (MDA), a widely recognized indicator of lipid peroxidation. This effect is robust across multiple populations and formulations, with the greatest reductions seen in studies using enhanced-bioavailability curcumin.
A meta-analysis of 28 RCTs found that curcumin and turmeric supplements significantly reduced MDA concentrations (SMD: -1.31), indicating a meaningful drop in oxidative damage [4]. Another meta-analysis of 21 meta-analyses reported a pooled effect size of -0.81 for MDA reduction, further confirming consistency across trials [5]. These effects are not limited to a single demographic, as studies have included healthy adults, individuals with prediabetes, and those with metabolic risk factors.
Table: Effect of Curcumin on Malondialdehyde (MDA) in Meta-Analyses | Meta-analysis | RCTs (n) | SMD or Effect Size | Population | Formulation | |----------------------|----------|--------------------|-------------------|----------------------------| | PMID: 41240262 | 28 | -1.31 | Mixed | Standard/turmeric extract | | PMID: 38945354 | 21 | -0.81 | Mixed | Various | | PMID: 39894187 | 20 | -0.37 mmol/L | Mixed | Nanocurcumin |
These findings are strongest when curcumin is delivered in formulations that address its poor natural absorption, such as nanocurcumin or curcumin with piperine. Standard doses in trials range from 250 mg to 1,000 mg per day, with nanocurcumin often used at the lower end of this range due to superior bioavailability [4][5][6].
While MDA is an established biomarker, reductions in MDA do not directly equate to prevention or treatment of specific diseases. Still, the reproducibility of this outcome across human trials establishes curcumin’s role in lowering oxidative damage at the biochemical level.
Boosting Endogenous Antioxidant Defenses: Glutathione, SOD, and Catalase
Curcumin supplementation leads to significant increases in the body’s endogenous antioxidant enzymes—glutathione, superoxide dismutase (SOD), and catalase—by activating Nrf2. Meta-analyses confirm that these biochemical shifts are among curcumin’s most reliable effects in human studies. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
For glutathione, a meta-analysis of 103 RCTs (n=7,216) found that curcumin supplementation significantly increased its levels, with moderate quality evidence [13]. Another analysis of 28 RCTs focusing on prediabetes and diabetes populations found an SMD of 1.72 for glutathione, demonstrating a large effect size [14]. Curcumin also significantly increased glutathione peroxidase activity (by 8.90 U/L) according to a meta-analysis of 21 meta-analyses [15]. Trials combining curcumin with piperine—a bioavailability enhancer—report even greater increases in glutathione [16].
SOD and catalase, both upregulated by Nrf2 signaling, also rise in response to curcumin supplementation. These enzymes, together with glutathione, form the primary intracellular defense network against oxidative stress. While individual trial results vary based on dose, formulation, and baseline antioxidant status, the overall trend is a consistent, measurable boost in endogenous antioxidant capacity.
Importantly, these changes occur even when participants do not track biomarkers themselves. The benefits can be interpreted as a general enhancement of the body’s resilience to oxidative stress, relevant for both those who measure antioxidant status and those who do not.
Formulation Matters: Bioavailability and Dosage in Curcumin Supplementation
Curcumin’s poor natural absorption means that the form and delivery method used in supplementation have a major impact on its biological effectiveness. Human studies show that enhanced-bioavailability formulations, such as nanocurcumin and curcumin combined with piperine, consistently achieve greater increases in antioxidant enzyme levels and larger reductions in oxidative stress markers.
Standard curcumin extracts—often 95% curcuminoids—have low oral bioavailability, with much of the compound rapidly metabolized and excreted. Piperine (from black pepper) can increase curcumin’s bioavailability by up to 2,000% by inhibiting hepatic and intestinal glucuronidation [7]. Nanoparticle, liposomal, and phospholipid complex formulations similarly increase absorption, resulting in higher and more sustained blood levels. A meta-analysis of nanocurcumin RCTs found significant reduction in MDA (SMD -0.37 mmol/L), supporting the enhanced effect of nanoparticle delivery [6].
Typical clinical trial doses range from 250 mg to 1,000 mg per day, with lower doses used for highly bioavailable forms. There is currently no consensus on an 'optimal' dose, but most trials that achieve significant antioxidant effects use formulations with proven absorption, often taken with food to further aid uptake. Importantly, higher doses of standard formulations do not necessarily compensate for poor bioavailability.
For readers considering supplementation, selecting a curcumin extract with documented enhanced bioavailability is likely to provide the most reliable boost to endogenous antioxidant defenses, whether or not specific biomarkers are being tracked.
Biomarker Interpretation: What Moves with Curcumin and What It Means
Curcumin supplementation reliably shifts several well-characterized biomarkers of oxidative stress and antioxidant status. The most robustly affected are glutathione, superoxide dismutase (SOD), catalase, and malondialdehyde (MDA). These changes reflect curcumin’s mechanism and can be observed with or without formal laboratory testing.
Glutathione: Curcumin increases total glutathione and glutathione peroxidase activity, with meta-analyses showing SMDs from 1.72 to 8.90 U/L depending on population and trial design [13][14][15]. In practical terms, this represents a substantial boost in the cell’s primary antioxidant buffer. Optimal glutathione levels vary but are generally considered high-normal if above 500 μmol/L in plasma.
SOD and Catalase: Increases in SOD and catalase after curcumin supplementation reflect improved breakdown of reactive oxygen species and hydrogen peroxide, respectively. While optimal ranges for these enzymes are lab-dependent, studies consistently report uptrends from baseline, especially with enhanced-absorption curcumin.
Malondialdehyde: Significant reductions in MDA (SMDs -0.81 to -1.31) indicate less lipid peroxidation and cellular membrane damage [4][5]. Although MDA is not routinely measured outside research, a decrease is universally interpreted as beneficial.
For those who do not track biomarkers, these shifts suggest a general strengthening of the body’s resilience to oxidative stress, while for those who do, improvements in these markers can validate effective dosing and formulation choice.
Indirect and Preclinical Evidence: Nrf2 Pathway, Animal Models, and Mechanistic Insights
Cell and animal studies provide deeper mechanistic understanding of how curcumin activates Nrf2 and modulates antioxidant defenses, but these findings must be interpreted as plausibility, not direct human outcomes. In rodent and cell models, curcumin reliably increases Nrf2 translocation to the nucleus and upregulates target genes for glutathione, SOD, and catalase [4][6].
For example, in diabetic rat models, curcumin increased Nrf2 activity and suppressed cell death in heart tissue, supporting the idea that activation of this pathway has broad protective effects [4]. In vitro, curcumin enhances the activity of endogenous antioxidant enzymes and reduces oxidative stress in macrophages and other cell types [6].
Preclinical research also highlights the importance of formulation: animal studies confirm that nanoformulations and piperine co-administration improve tissue levels of curcumin and downstream antioxidant effects [2][7]. These findings align with human trial data showing better results with enhanced-bioavailability products.
However, while preclinical evidence strongly supports the Nrf2-mediated mechanism, translation to disease prevention, longevity, or specific clinical outcomes in humans remains an open question. These studies provide mechanistic plausibility but should not be over-interpreted as direct proof of health benefits outside the context of measured biomarkers. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Curcumin’s most robust mechanism in humans is the activation of the Nrf2 transcription factor, which in turn increases the body’s own production of key antioxidant enzymes: glutathione, superoxide dismutase, and catalase. This effect is consistently documented in randomized controlled trials, particularly when enhanced-bioavailability formulations are used. The reduction in oxidative stress markers, especially malondialdehyde, is reliably reproduced across diverse populations and supplement types. For those seeking to support endogenous antioxidant defenses, curcumin offers a distinct approach by amplifying the cell’s natural resilience rather than simply acting as a direct antioxidant. The most effective strategies use well-absorbed forms of curcumin, at daily doses of 250–1,000 mg, to maximize these biochemical benefits, regardless of individual biomarker tracking. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
While the evidence for curcumin’s impact on antioxidant enzymes and oxidative stress markers is strong, most human studies are short-term and focus on surrogate biomarkers rather than clinical endpoints. The translation of improved antioxidant status to prevention or treatment of specific diseases remains speculative. Furthermore, significant variability exists in supplement formulations, doses, and populations studied. Preclinical data, while mechanistically supportive, should not be used to infer direct human benefit beyond the measured biochemical shifts. Finally, optimal dosing and long-term safety for general populations require further clarification. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how curcumin relates to your health goals and monitor changes in your biomarkers over time.
