How DHA Directly Integrates Into Cell Membranes to Control Inflammation
Membrane incorporation as the gateway to DHA’s anti-inflammatory effect
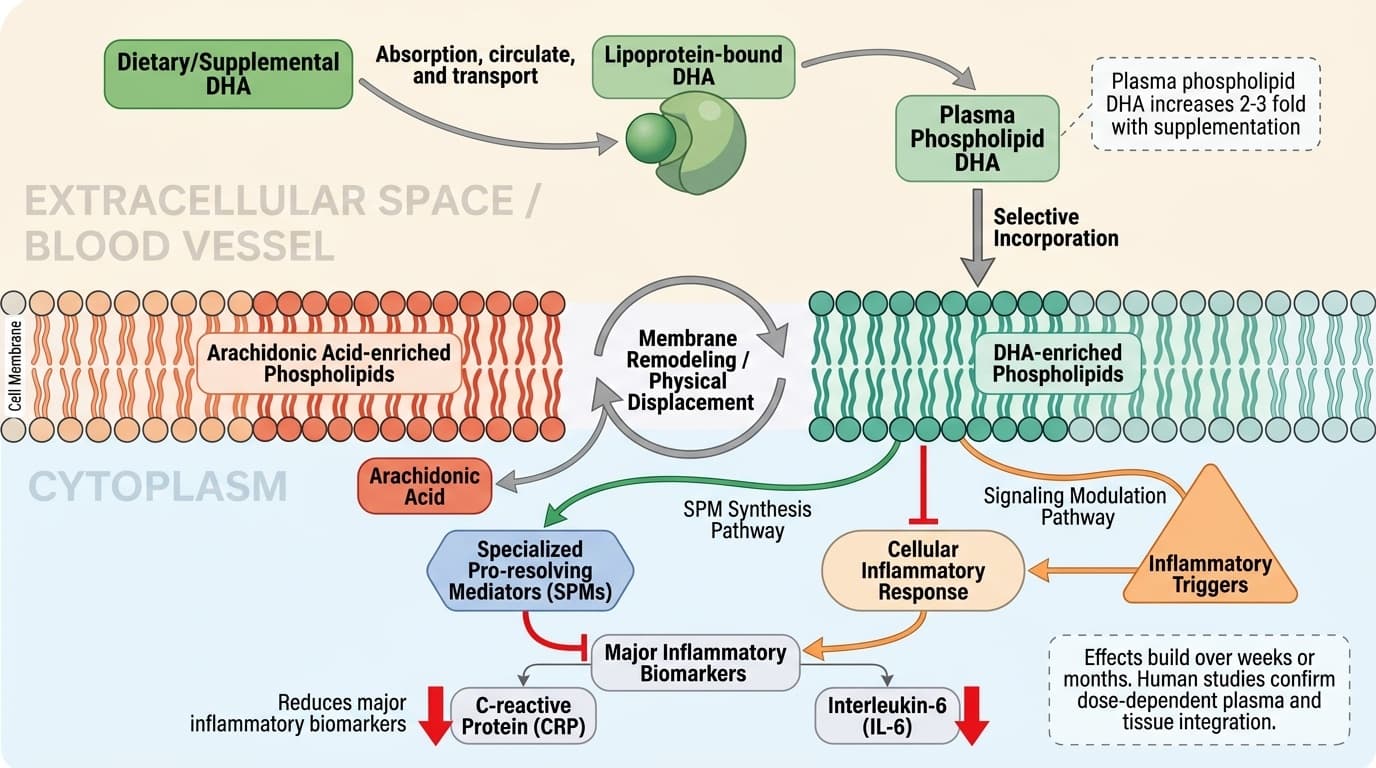
DHA works differently than most supplements—it doesn't just circulate in your blood temporarily.
Instead, it physically replaces inflammatory fats in your cell membranes, creating a structural foundation that actively reduces inflammation for weeks or months. This membrane integration is why DHA's effects build over time rather than appearing immediately.
When you take DHA consistently, it displaces arachidonic acid and other pro-inflammatory fats from cell membranes throughout your body. This swap changes how your cells respond to inflammatory triggers, reducing key markers like C-reactive protein and IL-6. The process takes several weeks because your body gradually rebuilds membranes with the new fatty acids you're providing through supplementation.
To achieve this membrane remodeling, you need 1-3 grams of DHA daily in triglyceride or phospholipid form—these are absorbed much better than ethyl ester versions. The anti-inflammatory benefits appear within 8-12 weeks of consistent use and depend on maintaining steady intake rather than sporadic high doses. This explains why DHA supplementation works as a long-term inflammation management strategy rather than a quick fix.
How DHA Directly Integrates Into Cell Membranes to Control Inflammation
Membrane incorporation as the gateway to DHA’s anti-inflammatory effect
Diagram glossary
- CRP:
- C-reactive protein is an inflammatory marker whose levels decrease following DHA supplementation.
- DHA:
- Docosahexaenoic acid is a fatty acid that integrates into cell membranes to reduce inflammation.
- IL-6:
- An inflammatory marker whose levels are reduced when DHA integrates into cell membranes.
- SPM:
- Specialized pro-resolving mediators are anti-inflammatory molecules synthesized from DHA within cell membranes.
- IL-6
- A branded dha product family name used to identify a specific extract or formulation in research and supplement labels.
- Phospholipid
- The main structural component of cell membranes, into which DHA is incorporated to exert its anti-inflammatory effects.
- Ethyl ester
- A chemical form used in some omega-3 supplements where fatty acids are attached to an ethanol backbone.
- Bioavailability
- The proportion of a compound that actually reaches your bloodstream and becomes usable by the body.
- DHA (Docosahexaenoic Acid)
- An omega-3 fatty acid that integrates directly into cell membranes, where it replaces pro-inflammatory fats and enables production of inflammation-resolving compounds.
- Plasma Phospholipid DHA
- A blood test measuring DHA content in membrane-like structures, used as a biomarker for tissue DHA levels and anti-inflammatory potential.
- Arachidonic Acid
- A pro-inflammatory fatty acid that DHA displaces from cell membranes, reducing the production of inflammatory compounds.
- CRP
- C-reactive protein is an inflammatory marker whose levels decrease following DHA supplementation.
- DHA
- Docosahexaenoic acid is a fatty acid that integrates into cell membranes to reduce inflammation.
- SPM
- Specialized pro-resolving mediators are anti-inflammatory molecules synthesized from DHA within cell membranes.
DHA Must Incorporate Into Cell Membranes to Exert Its Effects
DHA’s primary mechanism is its physical integration into cell membrane phospholipids, where it replaces arachidonic acid and other pro-inflammatory fatty acids. This membrane remodeling is essential for DHA to influence cellular signaling and generate anti-inflammatory mediators. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
The process begins when dietary or supplemental DHA is absorbed, circulates bound to lipoproteins, and is then selectively incorporated into the phospholipid bilayer of cell membranes. Human studies confirm that plasma and tissue phospholipid DHA increase in a dose-dependent manner with oral supplementation. For example, a 12-week trial in healthy adults showed that DHA supplementation at doses of 1.5–6 grams/day led to significant, linear increases in plasma and erythrocyte phospholipid DHA content, typically doubling or tripling baseline levels [1]. This incorporation is not instantaneous; it occurs gradually over weeks, reflecting the turnover of membrane phospholipids and the body’s regulation of fatty acid pools.
Membrane DHA displaces other fatty acids, notably arachidonic acid—a precursor to pro-inflammatory eicosanoids. As DHA accumulates in membranes, it alters the substrate availability for enzymes like cyclooxygenases and lipoxygenases, shifting the balance from pro-inflammatory to pro-resolving mediator production. This mechanism is supported by both human and preclinical evidence, including RCTs that directly measure changes in membrane composition following supplementation [2,3]. The physical presence of DHA in membranes is thus a prerequisite for its downstream effects on inflammation and cell signaling.
Membrane DHA Content Predictably Lowers Inflammatory Biomarkers
DHA's primary mechanism is its physical integration into cell membrane phospholipids, where it replaces arachidonic acid and other pro-inflammatory fatty acids. This membrane remodeling is essential for DHA to influence cellular signaling and generate anti-inflammatory mediators.
The process begins when dietary or supplemental DHA is absorbed, circulates bound to lipoproteins, and is then selectively incorporated into the phospholipid bilayer of cell membranes. Human studies confirm that plasma and tissue phospholipid DHA increase in a dose-dependent manner with oral supplementation. A 12-week trial in healthy adults showed that DHA supplementation at doses of 1.5–6 grams daily led to significant, linear increases in plasma and erythrocyte phospholipid DHA content, typically doubling or tripling baseline levels [1]. This incorporation occurs gradually over weeks, reflecting the turnover of membrane phospholipids and the body's regulation of fatty acid pools.
Membrane DHA displaces arachidonic acid—a precursor to pro-inflammatory eicosanoids. As DHA accumulates in membranes, it alters the substrate availability for enzymes like cyclooxygenases and lipoxygenases, shifting the balance from pro-inflammatory to pro-resolving mediator production. This mechanism is supported by both human and preclinical evidence, including RCTs that directly measure changes in membrane composition following supplementation [2,3]. The physical presence of DHA in membranes is thus a prerequisite for its downstream effects on inflammation and cell signaling.
DHA Enables Synthesis of Specialized Pro-resolving Mediators (SPMs)
Increasing membrane DHA consistently reduces major inflammatory biomarkers such as C-reactive protein (CRP) and interleukin-6 (IL-6) in humans. The anti-inflammatory effect is proportional to how much DHA is integrated into cell membranes, not just circulating in plasma.
A meta-analysis of 46 RCTs found that omega-3 supplementation—including DHA—reduced CRP by 0.40 mg/L, with higher doses achieving greater reductions [4]. Another network meta-analysis of 20 RCTs found no significant difference between EPA and DHA for CRP lowering, suggesting both exert their effects via similar membrane integration pathways [5]. Human RCTs using 1–3 grams daily of DHA have reliably shown reductions in IL-6 and CRP after 8–12 weeks, corresponding with measurable increases in plasma phospholipid DHA [3,6].
Importantly, the reduction in inflammatory markers is not instant but follows the time course required for DHA to accumulate in membranes. Individuals who do not track biomarkers still benefit from consistent daily supplementation, as the underlying mechanism—membrane remodeling—predictably lowers inflammation over time. This relationship is also observed in special populations, such as pregnant women, where higher DHA dosing (1000 mg daily versus 200 mg daily) led to greater reductions in inflammatory cytokines and improved pregnancy outcomes [7].
DHA Supplementation and Dose: How Much Is Needed for Membrane Integration?
Effective membrane integration of DHA in humans requires sustained daily intake, typically 1–3 grams per day of a highly bioavailable form such as triglyceride or phospholipid-bound DHA. Studies show that this range reliably increases plasma and erythrocyte phospholipid DHA content in a dose-dependent, linear manner.
A randomized controlled trial in healthy volunteers demonstrated that 1.5–6 grams/day of DHA for 12 weeks led to a proportional rise in membrane DHA, with the greatest gains seen at higher doses [1]. Meta-analyses confirm that anti-inflammatory effects—including CRP and IL-6 reduction—are most pronounced in this dosing range [4,6]. Importantly, the form of DHA matters: triglyceride and phospholipid-bound DHA show greater bioavailability and membrane incorporation compared to ethyl ester forms [3]. This aligns with evidence from preterm infant studies, where dosing at 40–120 mg/kg/day as a lipid emulsion led to clear, dose-dependent rises in blood phospholipid DHA [2].
For individuals not tracking blood levels, adherence to a daily 1–3 gram DHA intake over several weeks is a practical approach that reliably raises membrane DHA, as confirmed by multiple RCTs. Consistency—rather than sporadic high doses—is crucial for achieving and maintaining the necessary membrane integration for anti-inflammatory effects.
Comparing DHA Forms: Triglyceride, Phospholipid, and Ethyl Ester
The form in which DHA is consumed—triglyceride, phospholipid, or ethyl ester—directly affects its bioavailability and efficiency of membrane incorporation. Triglyceride and phospholipid forms are generally more bioavailable, leading to greater increases in membrane DHA at equivalent doses. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A comparison of DHA supplementation forms in human studies shows that triglyceride and phospholipid-bound DHA are absorbed more efficiently, resulting in higher plasma and erythrocyte phospholipid DHA levels than ethyl ester forms at the same dose [3]. This difference is attributed to the body’s natural digestive processes, which better handle triglyceride and phospholipid molecules. For example, phosphatidylserine-bound DHA has demonstrated higher brain and liver membrane integration in preclinical models, and human RCTs indicate similar trends for plasma phospholipid DHA [1,3].
A summary comparison of DHA forms:
| Formulation | Bioavailability | Membrane Integration | Notes | |--------------------|----------------|---------------------|---------------------------------| | Triglyceride | High | High | Most common in fish oil | | Phospholipid | High | High | Found in krill oil, some algae | | Ethyl Ester | Moderate–Low | Lower | Common in concentrated oils |
Choosing a highly bioavailable form enhances the likelihood of achieving the target membrane DHA concentration for anti-inflammatory benefits. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Plasma and Tissue DHA as Biomarkers: What Levels Reflect Optimal Integration?
Plasma phospholipid DHA serves as a reliable biomarker of membrane DHA content and is directly linked to anti-inflammatory outcomes. Optimal ranges are generally considered to be above 4–5% of total plasma phospholipid fatty acids for cardiovascular and inflammatory health benefits.
Human studies consistently show that DHA supplementation of 1–3 grams/day increases plasma phospholipid DHA from baseline averages of 2–3% up to 6% or higher within 8–12 weeks [1,3]. These levels correlate with significant reductions in CRP and IL-6, as well as improved clinical endpoints such as reduced risk of early preterm birth and lower systemic inflammation [2,4,7]. For those not tracking biomarkers, reaching and maintaining these levels is best achieved with consistent, daily DHA intake. For those who do track, aiming for a plasma phospholipid DHA of 5–8% is associated with optimal membrane integration and downstream anti-inflammatory effects in the published literature.
While red blood cell (RBC) DHA content is another common biomarker, plasma phospholipid DHA responds more rapidly to supplementation and is directly reflective of recent intake and membrane remodeling. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
The anti-inflammatory benefits of DHA are rooted in its direct incorporation into cell membrane phospholipids, where it displaces arachidonic acid and enables the synthesis of specialized pro-resolving mediators. This mechanism is well established in humans, with consistent evidence showing that daily intake of 1–3 grams of highly bioavailable DHA reliably raises membrane DHA content and reduces systemic inflammatory markers such as CRP and IL-6. The form of DHA—triglyceride or phospholipid—significantly impacts bioavailability and membrane integration. For most individuals, steady, daily supplementation over several weeks is necessary to achieve and maintain the membrane changes needed for anti-inflammatory effects. Understanding this mechanism clarifies why consistent intake, rather than sporadic dosing, is key to harnessing DHA's health benefits.
While the mechanism of DHA integration into cell membranes and its correlation with reduced inflammatory markers is strongly supported by human trials, some uncertainties remain. Not all downstream clinical outcomes—such as disease-specific effects—are fully mapped, and optimal dosing for all populations has not been established. Most evidence comes from supplementation studies of 1–3 grams/day; less is known about lower doses or intake from diet alone. Additionally, individual variability in absorption and membrane incorporation exists, although the overall relationship remains robust. More research is needed to define exact biomarker targets and to clarify differences among DHA supplement forms in diverse populations.
Track this in your stack
See how dha relates to your health goals and monitor changes in your biomarkers over time.
