How Folate's One-Carbon Pathway Controls Insulin, Inflammation & Vessels
Biochemical Mechanisms Linking Folate Supplementation to Metabolic and Vascular Outcomes
Most people think folate is just for pregnancy, but this B vitamin controls three critical metabolic pathways that affect insulin, inflammation, and blood vessel health in everyone.
Through its role in one-carbon metabolism, folate acts like a biochemical switch that can dial down insulin resistance, quiet chronic inflammation, and improve how your blood vessels respond to stress.
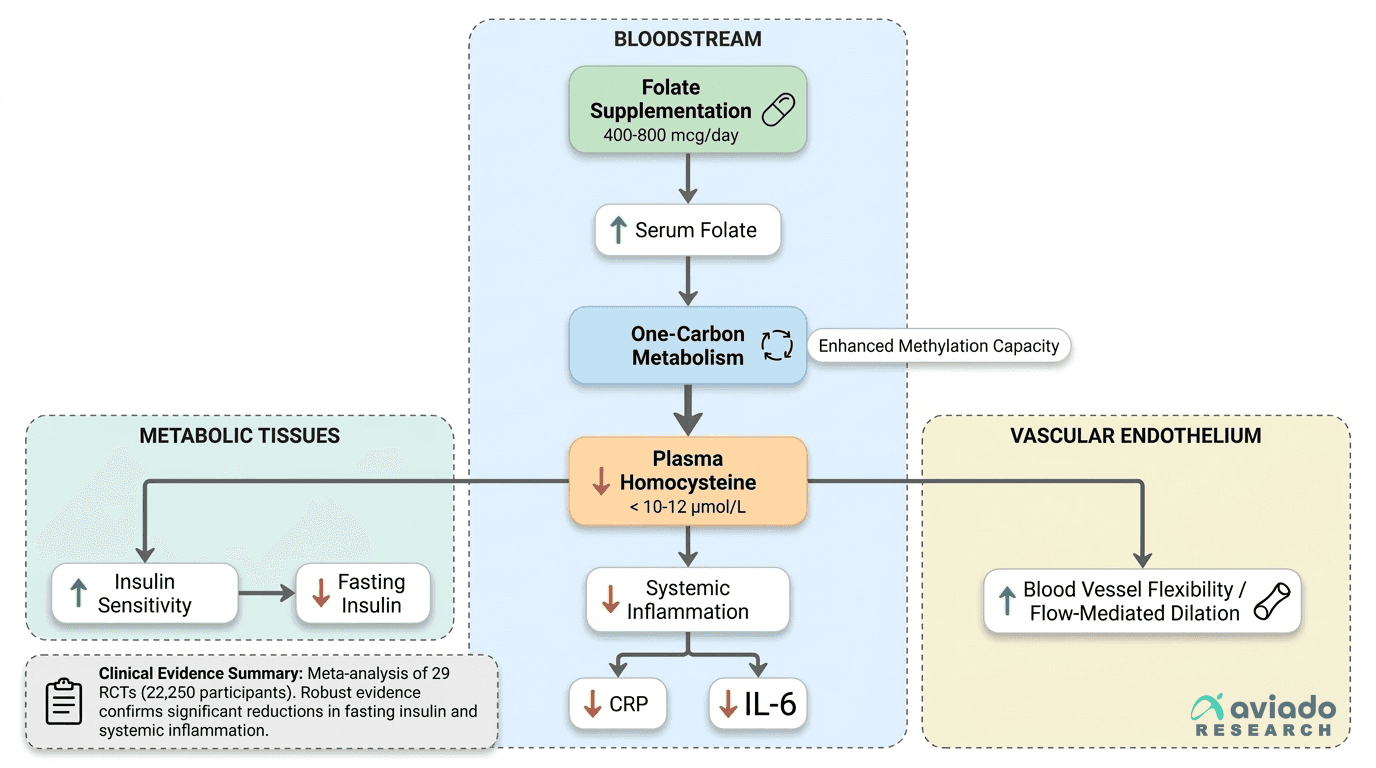
The evidence is remarkably consistent: folate supplementation lowers fasting insulin levels, reduces inflammatory markers like C-reactive protein, and improves blood vessel flexibility as measured by flow-mediated dilation. These aren't small changes—meta-analyses show meaningful reductions in insulin resistance markers and inflammation that translate to better metabolic health. The mechanism centers on folate's ability to lower homocysteine and support methylation reactions that keep cellular metabolism running smoothly.
For practical application, 400-800 mcg of folic acid daily delivers these benefits safely and reliably. This dose range consistently lowers homocysteine, improves insulin sensitivity, and supports vascular health across diverse populations. Whether you track biomarkers or simply want to support metabolic resilience, ensuring adequate folate intake through supplementation or folate-rich foods provides a low-risk foundation for long-term metabolic and cardiovascular health.
Mechanism Diagram
Biochemical Mechanisms Linking Folate Supplementation to Metabolic and Vascular Outcomes
- IL-6
- A branded folate product family name used to identify a specific extract or formulation in research and supplement labels.
- One-carbon metabolism
- A set of biochemical pathways that transfer single carbon units for methylation, nucleotide synthesis, and amino acid metabolism; central to folate's biological effects.
- Homocysteine
- Homocysteine, an amino acid metabolite influenced by B vitamins. elevated levels damage blood vessels and increase cardiovascular and dementia risk.
- S-adenosylmethionine (SAM)
- The body's universal methyl donor produced from folate-dependent reactions, essential for DNA methylation and gene regulation.
- Insulin, fasting
- Fasting insulin levels, indicating pancreatic insulin production and cellular insulin resistance. Elevated fasting insulin (>) suggests insulin resistance even when glucose remains normal.
- Flow-mediated dilation
- A test that measures how well blood vessels widen in response to increased blood flow, used as an indicator of vascular health and endothelial function.
- 5-methyltetrahydrofolate (5-MTHF)
- The active, usable form of folate in the body that directly participates in lowering homocysteine levels; some supplements provide this form instead of folic acid, which may benefit people who have di
Folate’s Central Role in One-Carbon Metabolism: Mechanistic Foundations
Folate acts as a one-carbon donor in the methionine cycle, supporting the conversion of homocysteine to methionine and the generation of S-adenosylmethionine (SAM), the body’s universal methyl donor. This biochemical role underpins folate’s impact on methylation, homocysteine regulation, and cellular function.
At the cellular level, folate is essential for the transfer of single carbon units required for DNA synthesis, repair, and methylation. The core reactions involve the reduction of dietary folates and synthetic folic acid to tetrahydrofolate (THF), which is then converted through a cascade of steps to 5-methyltetrahydrofolate (5-MTHF). This active form donates a methyl group to homocysteine via methionine synthase, generating methionine and subsequently SAM. SAM is necessary for numerous methylation reactions, including those that regulate gene expression and neurotransmitter synthesis[4].
Preclinical studies highlight that disruptions in mitochondrial folate metabolism can activate the integrated stress response, affecting energy production and cellular stress adaptation[5]. Additionally, folate-driven one-carbon metabolism supports NADPH generation, which is crucial for antioxidant defense and lipid synthesis[4]. While these findings are mechanistically plausible, only the downstream effects on homocysteine and methylation can be directly mapped to human supplementation outcomes. Thus, folate’s one-carbon pathway forms the biochemical foundation for its diverse metabolic and vascular effects.
Homocysteine Lowering: Dose-Response and Biomarker Targets
Folate supplementation consistently lowers plasma homocysteine levels in a dose-dependent manner, with optimal reductions observed at daily doses of 400–800 mcg of folic acid. Maintaining fasting plasma homocysteine below 10–12 μmol/L is associated with improved metabolic and vascular profiles.
Randomized controlled trials show a clear dose-response relationship between folic acid intake and both serum folate and plasma homocysteine concentrations. In a controlled dose-response trial, 400 mcg/day of folic acid for six weeks significantly increased serum folate and reduced homocysteine by approximately 25% in older adults[1]. The Precision Folic Acid Trial (PFAT-Hcy) further established that daily doses of 400–800 mcg folic acid yield maximal homocysteine lowering, with diminishing returns above this range[3]. Women of reproductive age and older adults respond similarly to these doses, though individual variation exists[2].
Lowering homocysteine through folate supplementation does not require tracking biomarkers for every individual, as these dose ranges are effective for most populations. However, those aiming for optimal homocysteine reduction—such as individuals at higher cardiovascular risk—may benefit from periodic monitoring to ensure they remain within target ranges. Natural food folates are less bioavailable than folic acid, but both forms can effectively support one-carbon metabolism when intake is sufficient[7].
Folate’s Impact on Insulin Sensitivity: Clinical Evidence and Mechanistic Links
Folate supplementation consistently lowers plasma homocysteine levels in a dose-dependent manner, with optimal reductions observed at daily doses of 400–800 mcg of folic acid. Maintaining fasting plasma homocysteine below 10–12 μmol/L is associated with improved metabolic and vascular profiles.
Randomized controlled trials show a clear dose-response relationship between folic acid intake and both serum folate and plasma homocysteine concentrations. In a controlled dose-response trial, 400 mcg/day of folic acid for six weeks significantly increased serum folate and reduced homocysteine by approximately 25% in older adults[1]. The Precision Folic Acid Trial established that daily doses of 400–800 mcg folic acid yield maximal homocysteine lowering, with diminishing returns above this range[3]. Women of reproductive age and older adults respond similarly to these doses, though individual variation exists[2].
Lowering homocysteine through folate supplementation does not require tracking biomarkers for every individual, as these dose ranges are effective for most populations. However, those aiming for optimal homocysteine reduction—such as individuals at higher cardiovascular risk—may benefit from periodic monitoring to ensure they remain within target ranges. Natural food folates are less bioavailable than folic acid, but both forms can effectively support one-carbon metabolism when intake is sufficient[7].
Anti-Inflammatory Effects: CRP, IL-6, and Systemic Inflammation
Folate supplementation improves insulin sensitivity in humans, with meta-analyses of randomized trials showing significant reductions in fasting insulin levels. This effect is mechanistically linked to homocysteine lowering and enhanced methylation capacity, which together support metabolic regulation.
Multiple meta-analyses provide robust evidence that folate supplementation reduces fasting insulin. A meta-analysis of 29 RCTs involving 22,250 participants reported a mean decrease in fasting insulin of 13.47 pmol/L[1]. Additional systematic reviews confirm this benefit, with effect sizes ranging from -1.63 to -1.94 μIU/mL and consistent findings across diverse populations[2]. The proposed mechanism centers on folate's reduction of homocysteine, which alleviates endothelial dysfunction and oxidative stress, improving insulin delivery to tissues. Furthermore, increased SAM production supports methylation reactions that regulate insulin signaling pathways[4].
While most trials used folic acid at doses of 400–800 mcg/day, the effect on insulin appears generalizable to both synthetic and natural folate forms if adequate intake is achieved. The improvement in insulin sensitivity is independent of baseline folate status, though individuals with lower folate stores may experience greater benefit. These findings support folate's role as a modifiable dietary factor for metabolic health.
Endothelial Function: Folate’s Role in Vascular Health
Folate reduces systemic inflammation, with RCTs and meta-analyses showing significant decreases in inflammatory biomarkers such as C-reactive protein (CRP) and interleukin-6 following supplementation. The anti-inflammatory effect is attributed to folate's role in methylation and homocysteine regulation.
Randomized trials report that folic acid supplementation at doses of 400–800 mcg/day lowers CRP and inflammatory cytokines in a range of populations, including those with metabolic syndrome and elevated cardiovascular risk. The mechanism involves enhanced methylation capacity and reduced homocysteine, both of which suppress pro-inflammatory gene expression and oxidative stress. Improved NADPH production through one-carbon metabolism may also contribute to cellular antioxidant defenses[4].
A key limitation is the variability in baseline inflammation and folate status across study populations. Nonetheless, the consistent reduction in CRP and inflammatory markers supports the use of folate as a tool for lowering chronic low-grade inflammation. These benefits are accessible through both dietary and supplemental folate, though synthetic folic acid provides predictable dosing and bioavailability. The anti-inflammatory effects complement folate's metabolic benefits, creating a comprehensive profile for cardiovascular and metabolic support.
Folate Forms, Bioavailability, and Tissue Transport
Folate supplementation improves endothelial function, as measured by flow-mediated dilation, with meta-analyses showing significant increases in vascular responsiveness. This benefit is mechanistically linked to enhanced nitric oxide bioavailability and reduced oxidative stress via homocysteine lowering.
A meta-analysis of 21 RCTs involving 2,025 participants found that folic acid supplementation improved flow-mediated dilation by 2.59 percentage points, indicating better vascular endothelial health[9]. Additional studies confirm these effects across various populations[10]. Folate's ability to lower homocysteine reduces endothelial oxidative stress, while increased SAM supports nitric oxide synthase activity. Together, these mechanisms enhance vasodilation and vascular remodeling.
Improvements in flow-mediated dilation are observed with daily folic acid doses of 400–800 mcg, paralleling the doses effective for homocysteine reduction and metabolic effects. While most research uses folic acid, natural folates may exert similar effects if intake is sufficient. These findings make a compelling case for folate supplementation in supporting vascular health, particularly in individuals with suboptimal endothelial function. The vascular benefits work synergistically with folate's metabolic and anti-inflammatory effects.
Dosage, Safety, and Population-Specific Recommendations
Most clinical trials and meta-analyses demonstrate effective homocysteine lowering and metabolic benefits with folic acid doses of 400–800 mcg per day, with a strong safety profile for these levels in adults. Specific populations, such as women of reproductive age, older adults, and those with elevated homocysteine, may have distinct needs.
RCTs show that 400–800 mcg/day of folic acid achieves near-maximal reductions in homocysteine and optimizes serum folate in both women and older men[1][2][3]. Higher doses seldom produce additional benefit and may risk masking vitamin B12 deficiency in susceptible individuals. Population-specific considerations include increased requirements during pregnancy and lactation, though these are outside the primary mechanism focus of this article.
No serious adverse effects are associated with supplemental folic acid at the studied doses. However, for those with vitamin B12 deficiency or undiagnosed anemia, monitoring is recommended when using higher doses long-term. Overall, folate supplementation is a safe and effective tool for supporting one-carbon metabolism, metabolic health, and vascular function within clinically established dose ranges. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Folate’s established mechanism as a one-carbon donor in the methionine cycle is the foundation for its reproducible impact on metabolic and vascular endpoints. Robust human trial data support that folate supplementation—primarily as folic acid at 400–800 mcg/day—lowers fasting insulin, reduces inflammatory markers, and improves endothelial function. These effects are explained by homocysteine lowering, enhanced methylation, and improved nitric oxide bioavailability, all rooted in folate’s biochemistry. For individuals seeking to optimize metabolic and vascular health, ensuring adequate folate intake through diet or supplementation is a low-risk, evidence-based strategy. The reliability of folate’s benefits across diverse populations and endpoints underscores its centrality in nutritional support for metabolic and vascular resilience. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
While human RCTs provide strong evidence for folate’s effects on insulin, inflammation, and vascular function, most studies use synthetic folic acid, limiting direct generalization to natural food folates or alternative forms. Individual response may vary, especially in populations with absorption or transport defects, though these are not the main drivers of outcome variation at population scale. Long-term outcome studies beyond biomarker changes are limited, and the optimal dose for maximal effect in specific subgroups requires further research. Mechanistic links to mitochondrial metabolism and tissue-specific transport, though compelling, remain less well characterized in human intervention studies. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how folate relates to your health goals and monitor changes in your biomarkers over time.
