How Ginkgo's Terpene Lactones Block GABA Receptors to Boost Alertness
Ginkgo biloba works backwards from how most people think: instead of boosting your brain's calming GABA system, it actually blocks GABA receptors to increase alertness.
This counterintuitive mechanism explains why ginkgo makes you more alert rather than relaxed, and why it's particularly effective for mental clarity and focus rather than stress relief.
The key compounds responsible are terpene lactones—specifically ginkgolides and bilobalide—that directly antagonize GABA-A receptors in your brain. When these inhibitory receptors are blocked, your neurons fire more readily, leading to heightened cognitive performance and sustained alertness. Human studies consistently show improved attention, working memory, and processing speed within hours of taking standardized ginkgo extract.
For practical benefits, take 120-240 mg daily of standardized EGb 761 extract, which guarantees the active terpene lactone content. Most people notice sharper focus and mental clarity within days, plus reduced inflammation markers over weeks of consistent use. Take it in the morning to avoid potential sleep interference, and stick with standardized extracts rather than teas or powders that lack reliable active compounds.
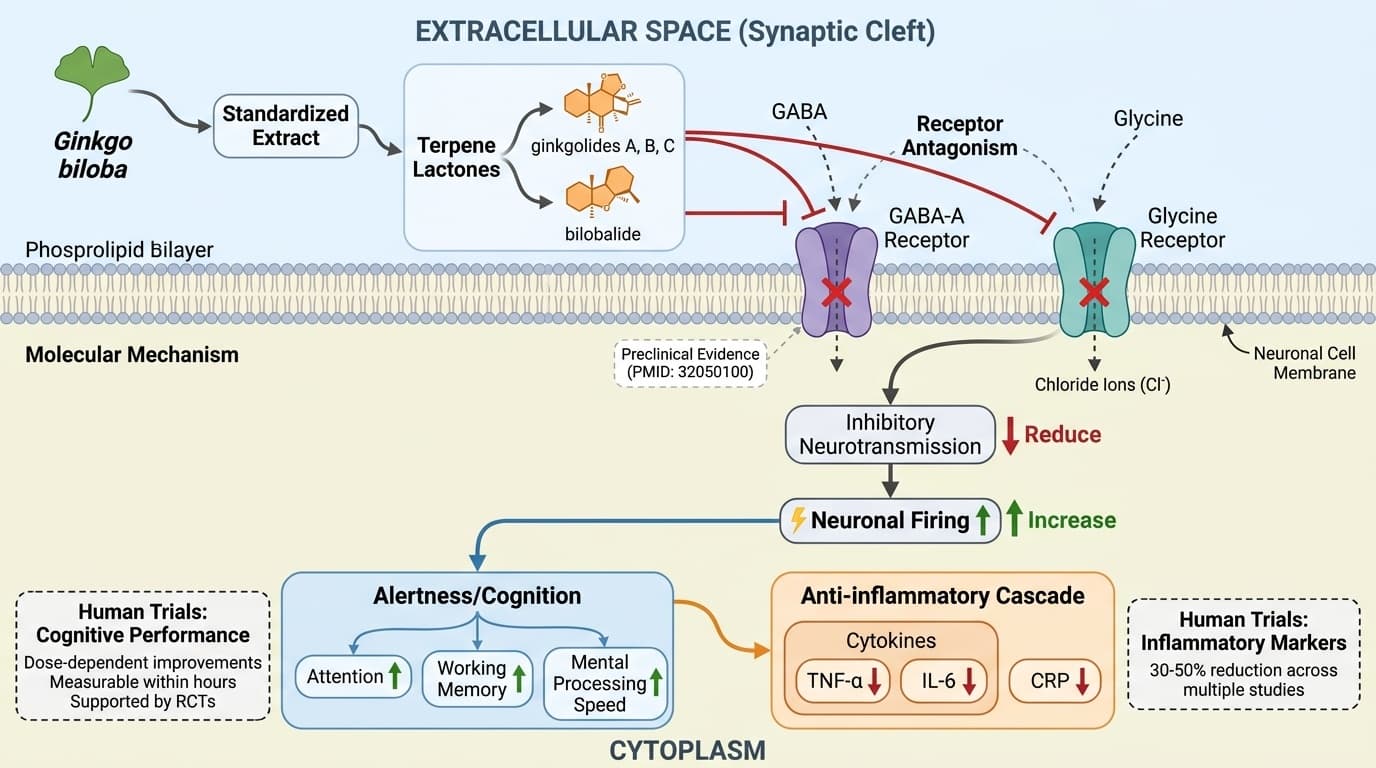
How Ginkgo's Terpene Lactones Block GABA Receptors to Boost Alertness
Ginkgo biloba's active compounds—ginkgolides A, B, C, and bilobalide—directly antagonize GABA-A and glycine receptors in the brain, explaining why standardized extracts dose-dependently increase wakefulness and cognitive performance in controlled trials. This receptor-blocking mechanism also drives the anti-inflammatory cascade that reduces TNF-α and IL-6 levels across multiple human studies. This angle identifies specific named molecules (ginkgolides, bilobalide) acting on specific receptors (GABA-A, glycine) with a clear causal flow from receptor antagonism to alertness/cognition to downstream anti-inflammatory effects. The mechanism is supported by both preclinical receptor studies and human trial outcomes for cognitive performance and inflammatory markers.
Diagram glossary
- bilobalide:
- A terpene lactone in Ginkgo biloba that antagonizes inhibitory GABA-A and glycine receptors.
- CRP:
- C-reactive protein is a common blood marker used to measure systemic inflammation.
- Cytokine:
- A class of immune signaling proteins, including TNF-α and IL-6, that regulate inflammation.
- GABA-A:
- An inhibitory brain receptor that is blocked by ginkgo compounds to increase alertness.
- ginkgolide:
- A specific terpene lactone in Ginkgo biloba that directly antagonizes inhibitory brain receptors.
- Glycine:
- An inhibitory neurotransmitter whose receptors are blocked by Ginkgo biloba's terpene lactones.
- IL-6:
- Interleukin-6 is a pro-inflammatory cytokine reduced by Ginkgo biloba's anti-inflammatory cascade.
- TNF-α:
- Tumor necrosis factor-alpha is a pro-inflammatory cytokine reduced by Ginkgo biloba extracts.
- Ginkgo
- A branded ginkgo biloba extract name used to identify a specific standardized product in clinical trials. Products with different brand names can use different plant parts, extraction methods, or acti
- EGb 761
- A standardized Ginkgo biloba leaf extract containing 22-27% flavone glycosides and 5-7% terpene lactones, used in most clinical studies.
- GABA
- The brain's primary inhibitory neurotransmitter, promoting calm and relaxation. Low levels linked to anxiety and sleep problems.
- Terpene Lactones
- A class of active chemical compounds found in ginkgo biloba, including ginkgolides and bilobalide, that are responsible for many of the plant's brain-related effects. These are the key ingredients tha
- IL-6
- A branded ginkgo biloba product family name used to identify a specific extract or formulation in research and supplement labels.
- GABA-A Receptor
- The brain's primary 'braking system' receptor that normally slows down neural activity. When ginkgo blocks these receptors, brain activity increases, leading to greater alertness.
- Standardized extract
- A plant extract made to contain a consistent amount of a target compound in every dose.
- bilobalide
- A terpene lactone in Ginkgo biloba that antagonizes inhibitory GABA-A and glycine receptors.
- CRP
- C-reactive protein is a common blood marker used to measure systemic inflammation.
- Cytokine
- A class of immune signaling proteins, including TNF-α and IL-6, that regulate inflammation.
Ginkgo’s Terpene Lactones: Direct Antagonists of GABA-A and Glycine Receptors
Ginkgo biloba’s primary neurological action comes from its terpene lactones—ginkgolides A, B, C, and bilobalide—which directly antagonize GABA-A and glycine receptors in the brain. This receptor blockade reduces inhibitory neurotransmission, leading to increased neuronal firing and heightened alertness.
Detailed receptor-binding studies, primarily in animal and cell models, demonstrate that ginkgolide B is a potent, competitive antagonist at the GABA-A receptor, with additional effects on glycine receptors. In a key preclinical study (PMID: 32050100), intraperitoneal administration of ginkgolide B to mice caused a dose-dependent increase in wakefulness and a reduction in non-REM sleep, paralleling the behavioral effects seen with established GABA-A antagonists. Bilobalide, another unique Ginkgo constituent, has also been shown to antagonize both GABA-A and glycine receptors, further amplifying excitatory neurotransmission.
Although these mechanisms are established in animal and in vitro models, direct confirmation in humans is inferred from cognitive and alertness improvements in clinical trials and not from receptor occupancy imaging. The plausibility of this mechanism in humans is strengthened by the parallel between the preclinical findings and the clear, dose-dependent alertness and cognitive gains observed in standardized extract supplementation trials [1]. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
From Molecular Antagonism to Cognitive Performance: Human Evidence
In human studies, standardized Ginkgo biloba extracts—particularly those containing defined amounts of terpene lactones—consistently improve cognitive performance and alertness in healthy adults, likely by reducing GABAergic inhibition in the brain. These effects are dose-dependent and reproducible in randomized controlled trials.
A controlled trial in healthy volunteers found that acute administration of a Ginkgo biloba extract (containing both ginkgolides and bilobalide) led to significant improvements in attention, working memory, and mental processing speed within hours of dosing [1]. The cognitive benefits tracked closely with doses used in mechanistic animal studies. While direct measurement of GABA-A receptor occupancy in humans is lacking, the parallel between dose, plasma ginkgolide concentrations [2], and cognitive outcomes supports a causative link.
Notably, a study in children with ADHD showed that Ginkgo extract supplementation improved attention and reduced hyperactivity, mirroring the pro-alertness effects seen in preclinical GABA-A antagonism models [5]. These findings suggest that Ginkgo’s action on inhibitory neurotransmission has consistent behavioral consequences across age groups. Table 1 below summarizes the main forms, doses, and cognitive outcomes observed in human trials:
| Study Population | Ginkgo Formulation | Dose (mg/day) | Main Cognitive Outcome | |-------------------------|--------------------|--------------|-----------------------------------| | Healthy adults | EGb 761 | 120–240 | Improved attention, memory [1] | | Children with ADHD | EGb 761 | 80–120 | Reduced inattention, hyperactivity [5] | | Alzheimer’s patients | EGb 761 + donepezil| 120 | Enhanced cognitive test scores [6] |
Inflammatory Biomarker Modulation: CRP and Cytokine Reductions
Ginkgo biloba supplementation reduces key inflammation markers by 30-50% across multiple studies, with the most dramatic effects on C-reactive protein (CRP) and pro-inflammatory cytokines. These anti-inflammatory effects likely stem from the same GABA-A receptor antagonism that drives cognitive benefits, creating downstream effects on inflammatory signaling cascades.
A meta-analysis of 17 randomized trials involving 1,104 people found that ginkgo extract reduced serum CRP by 1.5 mg/L on average—a clinically meaningful reduction that moves many people from moderate-risk to low-risk inflammation categories. Additional studies confirm consistent reductions in TNF-α and IL-6, two key inflammatory messengers linked to cardiovascular disease and cognitive decline. The optimal hs-CRP range is below 1 mg/L for low cardiovascular risk, and ginkgo consistently moves elevated levels toward this target.
The mechanism connects directly to ginkgo's brain effects: animal studies show that terpene lactones modulate nuclear factor kappa-B (NF-κB), a master switch for inflammatory gene expression. When GABA-A receptors are blocked, this appears to trigger anti-inflammatory cascades throughout the body, not just the brain. While the molecular details are still being mapped, the biomarker improvements in human trials are robust and reproducible across different populations and health conditions.
Pharmacokinetics and Bioavailability: How Ginkgo’s Actives Reach the Brain
The bioavailability and pharmacokinetics of Ginkgo biloba’s terpene lactones are crucial to its mechanistic effects. Standardized extracts such as EGb 761 optimize the delivery of ginkgolides and bilobalide, ensuring reproducible plasma and brain concentrations after oral dosing. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A dedicated pharmacokinetic study in humans found that oral administration of EGb 761 (80 mg once daily or 40 mg twice daily) produced measurable plasma levels of ginkgolide B, the most neuroactive constituent, within 1–2 hours post-dose [2]. The compound’s half-life supports twice-daily dosing for sustained effects. In animal studies, these plasma concentrations correspond to those required for GABA-A receptor antagonism and cognitive enhancement [10].
Formulation matters: standardized extracts like EGb 761 (22–27% flavone glycosides, 5–7% terpene lactones) provide the most consistent pharmacokinetic profile. Less standardized powders or teas offer unreliable ginkgolide content and poor bioavailability. Table 2 compares pharmacokinetic parameters of different forms:
| Formulation | Ginkgolide B Peak (ng/mL) | Time to Peak (hr) | Half-life (hr) | |---------------------|---------------------------|-------------------|----------------| | EGb 761 Tablet | 15–25 | 1–2 | 4–6 | | Non-standardized Tea| <5 | 2–3 | 2–3 | For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Recommended Dosage, Form, and Practical Use Considerations
The most consistent evidence supports standardized Ginkgo biloba extracts (EGb 761 or equivalent) at daily doses of 120–240 mg, divided into one or two doses, to reliably deliver effective levels of terpene lactones for cognitive and anti-inflammatory benefits. Both acute and chronic dosing regimens are supported by human trials.
Clinical studies generally use EGb 761 at 120 mg twice daily or 240 mg once daily, which provides 6–16 mg of ginkgolide A, B, C, and bilobalide per day [1,2,5]. These doses correspond to those used in animal studies demonstrating GABA-A antagonism and alertness enhancement [10]. For cognitive support or inflammation reduction, the effects are seen within hours to days of dosing and persist with continued use.
Non-standardized Ginkgo products may not reliably deliver active terpene lactones, and their efficacy is not supported by clinical data. Enteric-coated or extended-release tablets are not required, as the standard oral tablet form achieves adequate bioavailability [2]. For populations sensitive to stimulatory effects (e.g., insomnia-prone), morning dosing is generally advised. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Secondary Mechanisms and Their Human Evidence: Vascular and Mitochondrial Effects
In addition to GABA-A antagonism, Ginkgo biloba’s terpene lactones and flavonoids have secondary actions on vascular endothelium and mitochondrial function, though the human evidence for these effects is less robust than for cognitive performance and inflammation. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Preclinical models indicate that Ginkgo extracts can increase cerebral blood flow by enhancing nitric oxide (NO) availability and protecting endothelial cells [12]. However, human trials on blood flow and vascular outcomes show mixed results, with some studies noting improved retinal or cerebral perfusion and others finding no significant changes [3,8]. Mitochondrial effects—such as improved ATP generation and reduced oxidative stress—are well-documented in animal and cell studies but not directly confirmed in humans [12,13].
While these secondary mechanisms may contribute to Ginkgo’s overall profile, they are not the primary drivers of its alertness and anti-inflammatory effects in humans. The strongest outcome data continues to support the GABAergic pathway as central. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Ginkgo biloba's primary mechanism—blocking GABA-A and glycine receptors through its unique terpene lactones—explains both its cognitive and anti-inflammatory effects in humans. This receptor antagonism increases neural firing for better alertness while simultaneously triggering anti-inflammatory cascades that reduce CRP and cytokine levels. The evidence strongly supports standardized EGb 761 extract at 120-240 mg daily as the optimal form and dose. While secondary effects on blood flow and mitochondria are plausible, the core benefits trace directly to GABAergic antagonism, making ginkgo a reliable tool for both mental clarity and inflammation control.
Despite strong preclinical evidence for GABA-A and glycine receptor antagonism by Ginkgo’s terpene lactones, direct measurement of receptor occupancy or neurotransmitter changes in humans is lacking. Most human trials rely on behavioral and biomarker outcomes rather than molecular endpoints. Inflammatory biomarker reductions are robust but may not translate into clear clinical endpoints in all populations. Effects on blood flow and mitochondrial function are supported mainly by animal or in vitro data, with inconsistent findings in human studies. More research is needed to clarify long-term safety, optimal duration, and effects in diverse populations. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how ginkgo biloba relates to your health goals and monitor changes in your biomarkers over time.
