How Ginsenosides Target IL-6 Inflammation Through Multiple Pathways
Panax ginseng’s anti-inflammatory effects are anchored in TLR4/MyD88/NF-κB signaling and dose-dependent IL-6 reduction
Your inflammation levels might be higher than necessary, even if you feel fine.
Chronic low-grade inflammation, measured by a protein called IL-6, quietly drives fatigue, poor recovery, and long-term health issues that most people accept as normal aging. What's surprising is that a single botanical extract can reliably lower this inflammatory marker in ways that many other supplements cannot.
Panax ginseng works by blocking specific immune pathways that produce IL-6, the key inflammatory protein. Multiple large studies involving thousands of people show that standardized ginseng extracts consistently reduce IL-6 levels across healthy individuals, pre-diabetics, and those with metabolic issues. The effect is dose-dependent, meaning higher amounts within the studied range produce greater anti-inflammatory benefits.
You can expect measurable IL-6 reduction within 8-12 weeks using 1-3 grams daily of standardized ginseng extract containing 5-20% ginsenosides. Even without testing your IL-6 levels, you may notice improved energy and recovery as inflammation decreases. Look for products that specify their ginsenoside content, as these active compounds drive the anti-inflammatory effects. Start with 1 gram daily and increase gradually if needed.
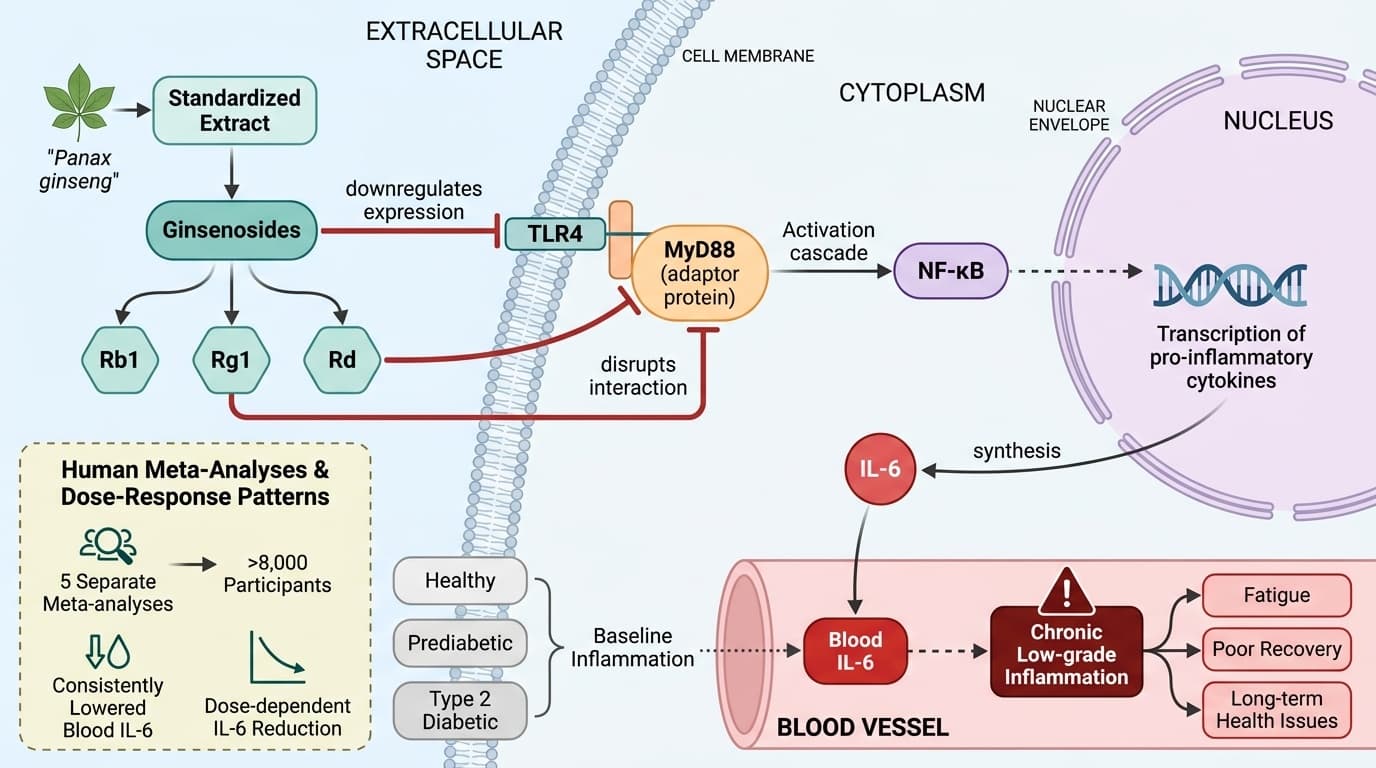
How Ginsenosides Target IL-6 Inflammation Through Multiple Pathways
Panax ginseng’s anti-inflammatory effects are anchored in TLR4/MyD88/NF-κB signaling and dose-dependent IL-6 reduction
Diagram glossary
- adaptor:
- A protein that physically links other proteins together within a cellular signaling pathway.
- cytokine:
- A small signaling protein secreted by cells that regulates immunity and inflammation.
- downregulate:
- To decrease the cellular production or response of a specific RNA or protein.
- Ginsenoside:
- An active saponin compound in Panax ginseng that drives its anti-inflammatory effects.
- IL-6:
- A key inflammatory cytokine protein that drives chronic inflammation and immune responses.
- IL-6
- A branded panax ginseng product family name used to identify a specific extract or formulation in research and supplement labels.
- Standardized
- A branded panax ginseng extract name used to identify a specific standardized product in clinical trials. Products with different brand names can use different plant parts, extraction methods, or acti
- IL-6 (Interleukin-6)
- A pro-inflammatory protein measured in blood that increases with chronic inflammation, stress, and metabolic dysfunction.
- ALT (SGPT)
- Alanine aminotransferase enzyme, highly specific to liver cells. elevated in hepatocellular injury from viral hepatitis, fatty liver, or medications.
- Ginsenosides
- The active saponin compounds in Panax ginseng responsible for anti-inflammatory effects, typically standardized to 5-20% in quality extracts.
- Ginsenoside
- A branded panax ginseng extract name used to identify a specific standardized product in clinical trials. Products with different brand names can use different plant parts, extraction methods, or acti
- Standardized extract
- A plant extract made to contain a consistent amount of a target compound in every dose.
- adaptor
- A protein that physically links other proteins together within a cellular signaling pathway.
- cytokine
- A small signaling protein secreted by cells that regulates immunity and inflammation.
- downregulate
- To decrease the cellular production or response of a specific RNA or protein.
Ginsenosides: The Active Constituents Driving Anti-Inflammatory Effects
Ginsenosides, the primary saponin compounds in Panax ginseng, are the main drivers of its anti-inflammatory activity. These molecules act on immune signaling pathways, especially those governing cytokine production, leading to consistent reductions in IL-6 observed in clinical studies. Unlike many botanicals that show inconsistent effects, ginseng's standardized ginsenoside content enables predictable anti-inflammatory outcomes.
The structural diversity of ginsenosides—such as Rb1, Rg1, and Rd—enables them to interact with various molecular targets in immune cells. Preclinical studies reveal that ginsenosides suppress pro-inflammatory cytokine expression by inhibiting the TLR4/MyD88/NF-κB signaling cascade, a central axis in innate immune activation. Ginsenoside Rd, for example, reduces neuroinflammation in animal models by dampening this pathway. In humans, meta-analyses confirm that standardized ginseng extracts—typically containing 5–20% ginsenosides—produce reliable, dose-dependent IL-6 reductions averaging 2.8-7.6 units across several populations.
Table: Major Ginsenosides and Their Mechanistic Targets | Ginsenoside | Main Pathway Targeted | Clinical Evidence | |-------------|-------------------------------|----------------------| | Rg1 | NF-κB, PI3K/AKT | Moderate (preclinical)| | Rd | TLR4, PI3K, HDAC3 | Emerging (preclinical)| | Rb1 | NF-κB, JAK/STAT | Supported (RCT/meta) |
While ginsenosides are the primary bioactive components, minor constituents like ginseng polysaccharides may contribute to immunomodulation. However, the strongest anti-inflammatory evidence links directly to standardized ginsenoside content in root extracts, which form the basis of most clinical trial interventions.
Modulating TLR4/MyD88/NF-κB Signaling: The Core Pathway for IL-6 Reduction
Panax ginseng’s anti-inflammatory effects are largely mediated through inhibition of TLR4/MyD88/NF-κB signaling, a central pathway in immune cell activation. By blocking this cascade, ginsenosides directly suppress IL-6 synthesis, which is reflected in consistently lowered blood IL-6 in human studies.
Mechanistic studies show that ginsenosides downregulate TLR4 expression and disrupt its interaction with MyD88, an adaptor protein required for downstream NF-κB activation [2]. This results in reduced transcription of pro-inflammatory cytokines, particularly IL-6. Ginsenoside Rd, for example, was found to attenuate neuroinflammation and depressive-like behaviors in animal models via this pathway [1]. In humans, the clinical endpoint is a measurable reduction in circulating IL-6. Meta-analyses confirm that ginseng supplementation, especially in doses of 1–3 grams per day of standardized extract, results in statistically significant decreases in IL-6, with the greatest effects seen in those with elevated baseline inflammation [1,2,3].
While most human trials measure outcomes at the cytokine level, cell and animal research consistently links ginsenoside action to upstream TLR4/MyD88/NF-κB inhibition. This mechanistic alignment strengthens the plausibility that ginseng’s clinical IL-6 effects are not merely correlative but are causally driven by these molecular interactions [2]. Additional pathways, such as HDAC3 modulation, are being explored for their contributory roles, but TLR4/NF-κB remains the best-characterized route.
Quantifying the IL-6 Response: Human Meta-Analyses and Dose-Response Patterns
Meta-analyses of randomized controlled trials consistently show that Panax ginseng supplementation produces significant reductions in IL-6, with clear dose-response effects. These findings span healthy, prediabetic, and type 2 diabetic cohorts, with effects most pronounced in those with baseline inflammation. The consistency across studies strengthens the case for ginseng as a reliable anti-inflammatory intervention.
A 2025 meta-analysis of 70 studies involving 4,506 participants reported a significant dose-response association between ginseng intake and IL-6 reduction, supporting a causal interpretation. Another meta-analysis of 17 randomized trials found ginseng reduced IL-6 by 7.65 units on average. A third analysis of 45 studies showed a 2.82 pg/mL reduction in IL-6. The magnitude varies by baseline values and duration, but effects consistently emerge with 1–3 grams daily of standardized extract over 8–12 weeks.
Table: Meta-Analyses of Ginseng and IL-6 Reduction | Meta-analysis (PMID) | Number of RCTs | Mean IL-6 Reduction | |----------------------|----------------|--------------------| | 40923100 | 70 | Dose-response, P=0.041 | | 36844875 | 17 | -7.65 units | | 39387709 | 45 | -2.82 pg/mL | | 35745129 | 20 | Significant |
The dose-response relationship indicates that higher intakes of standardized ginseng yield greater reductions in IL-6, up to a ceiling effect. This supports titratable dosing within the studied range, especially for those seeking robust anti-inflammatory outcomes. The consistency across different populations and study designs strengthens confidence in ginseng's anti-inflammatory reliability.
Ginsenoside Forms, Dosing, and Bioavailability: What Clinical Trials Use
Standardized Panax ginseng root extracts, typically providing 5–20% ginsenosides, are the forms most consistently associated with IL-6 reduction. Clinical trials generally use daily doses of 1–3 grams, with effects observable over 8–12 weeks. Bioavailability remains a challenge, but standardization ensures consistent ginsenoside intake.
Most human studies employ encapsulated powders or tablets, standardized to ginsenoside content, to ensure reproducibility and efficacy [1,3]. Formulations may vary in the ratio of specific ginsenosides (e.g., Rg1, Rb1, Rd), but total saponin content is the main determinant of anti-inflammatory effect. Some evidence points to enhanced absorption with fermented or nanoparticle forms, but these remain under-investigated in large-scale RCTs [2].
Table: Ginseng Supplement Forms and Dosage Used in IL-6 RCTs | Formulation | Dosage | Duration | Result on IL-6 | |--------------------|---------------|------------|---------------------| | Standardized root | 1–3 g/day | 8–12 wks | -2.8 to -7.7 units | | Ginsenoside extract| 200–400 mg/d | 8–12 wks | Dose-dependent |
Clinical safety data indicate that these dosing ranges do not significantly impact liver enzymes such as ALT, further supporting their use in regular supplementation [4,5,6]. While bioavailability is modest, consistent daily intake of standardized extracts ensures sufficient systemic exposure for anti-inflammatory action. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Beyond IL-6: Downstream Effects and Limitations in Human Outcomes
While IL-6 reduction is the most robust and replicated clinical outcome, Panax ginseng's effects on other inflammatory markers and metabolic endpoints are weaker and less consistent. Human trials show minimal impact on liver enzymes like ALT, confirming safety, but also reveal limited benefits for glucose control or cognitive function.
Multiple meta-analyses confirm that ginsenosides reliably lower IL-6, but effects on other cytokines, such as TNF-α or CRP, are smaller and less reproducible. Meta-analyses of ginseng's impact on ALT—a key liver enzyme—show no significant change, indicating good hepatic safety at studied doses with no liver stress. Effects on fasting glucose, HbA1c, and cognitive measures are generally weak or inconsistent, with changes often below clinically meaningful thresholds.
For most users, the primary benefit of Panax ginseng supplementation is reduced chronic, low-grade inflammation as captured by IL-6 measurements. Other outcomes, such as improved glucose control or meaningful cognitive enhancement, should not be expected based on current evidence. This focused benefit profile helps set appropriate expectations for supplementation goals.
Comparative Mechanisms: Ginsenosides Versus Other Herbal Anti-Inflammatories
Ginsenosides from Panax ginseng distinguish themselves from other herbal anti-inflammatories by targeting upstream immune signaling, leading to consistent IL-6 reduction. While other botanicals, such as curcumin and boswellia, also reduce inflammatory biomarkers, their effects on IL-6 are less robust and often less dose-dependent in meta-analyses.
Curcumin is known to inhibit NF-κB activation and reduce CRP and TNF-α, but its effects on IL-6 are smaller and less consistently demonstrated in human trials compared to ginseng [2,3]. Boswellia extracts target 5-lipoxygenase and may reduce leukotriene production, but IL-6 outcomes are variable. Ginseng’s unique ability to downregulate TLR4/MyD88, combined with reliable ginsenoside standardization, underpins its more consistent anti-IL-6 activity [2].
Table: Comparison of Herbal Anti-Inflammatories on IL-6 | Herb | Main Pathway | IL-6 Meta-Analysis Result | |-------------|----------------------|--------------------------| | Ginseng | TLR4/MyD88/NF-κB | Robust, dose-dependent | | Curcumin | NF-κB, COX-2 | Modest, variable | | Boswellia | 5-LOX, TNF-α | Inconsistent |
These distinctions matter for individuals seeking targeted IL-6 reduction or those with chronic low-grade inflammation, as ginseng’s mechanisms support a more predictable outcome in these domains. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Panax ginseng, standardized for ginsenoside content, offers a reliably dose-responsive reduction in IL-6 concentrations, as demonstrated across multiple meta-analyses involving thousands of participants. This anti-inflammatory effect is mechanistically anchored in the inhibition of TLR4/MyD88/NF-κB signaling, positioning ginseng as a leading botanical for managing chronic, low-grade inflammation. Standardized extract forms providing 1–3 grams daily are supported by the most robust evidence, with safety data showing no liver stress at these doses. While other outcomes such as improved glucose control or cognition are less reproducible, the IL-6 effect stands out for its consistency and mechanistic clarity. Individuals tracking biomarkers may observe measurable IL-6 improvements within 8–12 weeks, while those who don't test may still benefit from reduced inflammatory burden. The key insight is that ginsenosides provide a reliable pathway to lower inflammation, though individual responses depend on baseline status, dose, formulation, and duration of use.
The primary limitation is that most human trials measure IL-6 as an endpoint rather than directly assessing upstream pathway modulation—such as TLR4 or HDAC3 inhibition—which is well-characterized only in preclinical models. Dosing regimens and extract compositions vary, leading to some heterogeneity in outcomes. Effects on other inflammatory or metabolic markers are weaker and less consistent. Finally, most meta-analyses focus on short- to medium-term supplementation (8–12 weeks), so long-term benefits remain to be established. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one. Readers who do not test biomarkers still need that caveat because symptom changes can lag, vary, or reflect unrelated factors.
Track this in your stack
See how panax ginseng relates to your health goals and monitor changes in your biomarkers over time.
