How Hyaluronic Acid's Molecular Weight Drives Opposing Inflammatory Signals
Understanding the Size-Dependent Effects of Hyaluronic Acid on Human Inflammation and Skin Hydration
Hyaluronic acid supplements can either fight inflammation or fuel it, depending entirely on their molecular weight—a detail most products never disclose.
High molecular weight hyaluronic acid triggers anti-inflammatory signals by boosting IL-10, while low molecular weight forms do the opposite, ramping up inflammatory IL-6. This explains why some people see dramatic improvements in joint pain and skin hydration while others experience no benefit or even worsening symptoms.
The key finding from systematic reviews is that molecular size determines which cellular receptors hyaluronic acid activates. Large molecules bind to CD44 receptors and calm inflammation, while small fragments activate toll-like receptors that sound inflammatory alarms. This isn't just laboratory theory—clinical trials show high molecular weight forms consistently improve skin hydration and reduce joint inflammation, while low molecular weight products often fail or backfire.
For reliable results, choose products specifically labeled as high molecular weight hyaluronic acid (1,000 kDa or higher). Effective doses are 120-240 mg daily for oral supplements or 0.1-0.3% for topical applications. Medical-grade injectables and skincare products are more likely to specify molecular weight than over-the-counter supplements. If your current hyaluronic acid isn't working, the molecular weight mismatch is likely the culprit.
How Hyaluronic Acid's Molecular Weight Drives Opposing Inflammatory Signals
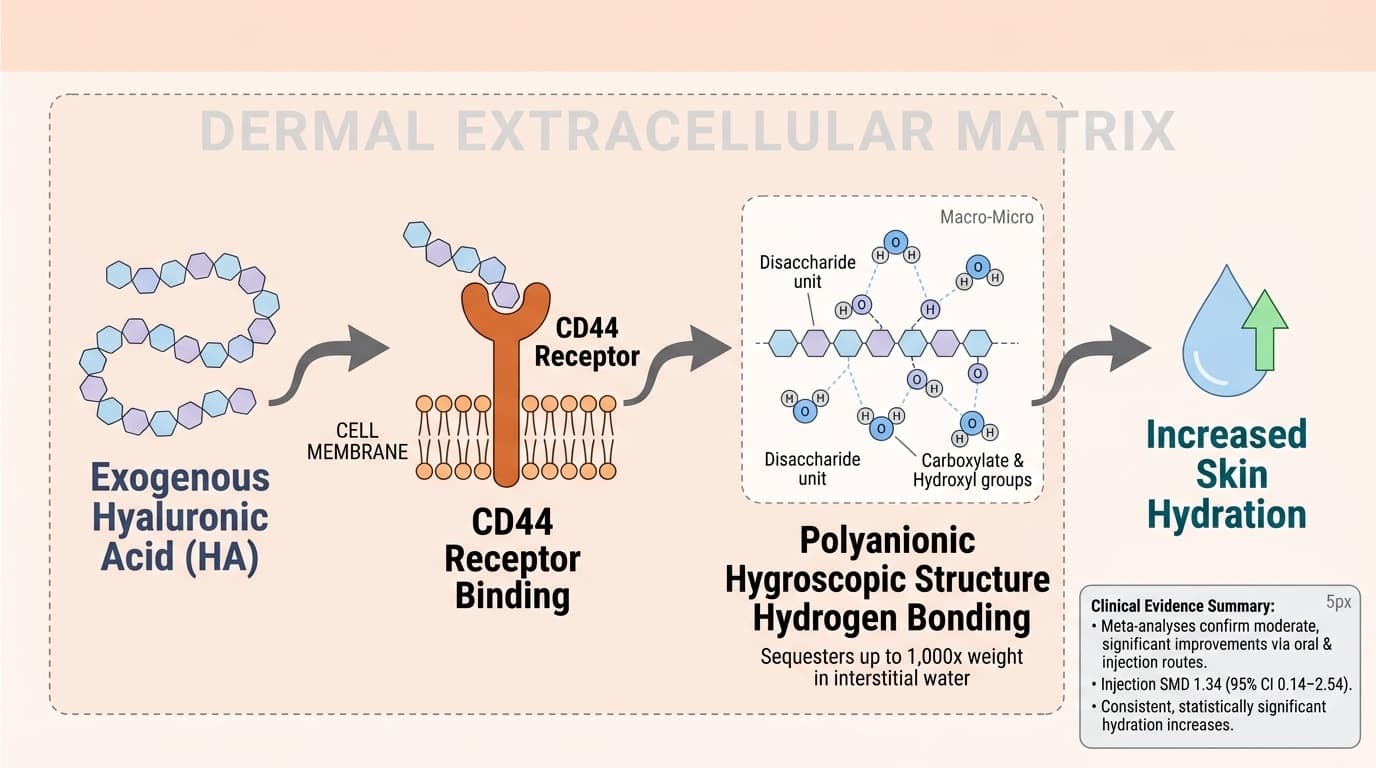
A simple arrow diagram should depict exogenous Hyaluronic Acid binding to dermal CD44 receptors and engaging its polyanionic hygroscopic structure as the primary pathway node, with a downstream arrow indicating increased skin hydration.
Diagram glossary
- carboxylate:
- A functional group in hyaluronic acid that forms extensive hydrogen bonds with water molecules.
- CD44:
- A cell-surface receptor in the dermal matrix that binds and stabilizes hyaluronic acid.
- disaccharide:
- A carbohydrate unit composed of two simple sugars that forms hyaluronic acid polymers.
- hydrogen:
- A chemical element that forms bonds with water molecules to facilitate hyaluronic acid hydration.
- IL-10
- A branded hyaluronic acid product family name used to identify a specific extract or formulation in research and supplement labels.
- IL-6
- A branded hyaluronic acid product family name used to identify a specific extract or formulation in research and supplement labels.
- Interleukin-10
- Interleukin-10, the primary anti-inflammatory regulatory cytokine. Lower levels associated with chronic inflammatory conditions.
- IL-10 (Interleukin-10)
- An anti-inflammatory cytokine that helps resolve inflammation and promote tissue healing; higher levels are generally beneficial.
- IL-6 (Interleukin-6)
- A pro-inflammatory cytokine that increases with tissue damage or immune activation; elevated levels indicate inflammation.
- Weight
- Body weight in kilograms, most basic anthropometric measure.
- CD44 receptor
- Cell surface receptor that binds high molecular weight hyaluronic acid and triggers anti-inflammatory pathways.
- carboxylate
- A functional group in hyaluronic acid that forms extensive hydrogen bonds with water molecules.
- CD44
- A cell-surface receptor in the dermal matrix that binds and stabilizes hyaluronic acid.
- disaccharide
- A carbohydrate unit composed of two simple sugars that forms hyaluronic acid polymers.
Molecular Weight Determines Hyaluronic Acid’s Inflammatory Signaling
Hyaluronic acid’s effects are not uniform—its molecular weight determines its biological activity. High molecular weight hyaluronic acid (HMW-HA) upregulates anti-inflammatory IL-10, while low molecular weight hyaluronic acid (LMW-HA) fragments trigger pro-inflammatory IL-6 responses, primarily through differential receptor engagement.
Systematic reviews demonstrate that LMW-HA upregulates IL-6, a marker of inflammation, while HMW-HA elevates IL-10, promoting inflammation resolution [1]. Mechanistically, LMW-HA binds to toll-like receptors (TLR2/4) on immune cells, activating NF-κB signaling and resulting in IL-6 secretion. In contrast, HMW-HA primarily interacts with CD44 and suppresses inflammatory cascades, increasing IL-10 output [1]. These divergent pathways explain why some HA products reduce inflammation while others may exacerbate it. A 12-week RCT with HA eye drops found significant decreases in tear IL-6 levels, likely reflecting the use of HMW-HA [2].
Importantly, most commercial hyaluronic acid supplements and injectables do not clearly specify molecular weight, contributing to inconsistent outcomes in clinical studies. When molecular size is controlled, the anti-inflammatory benefits of HMW-HA are more reliably observed, particularly in joint and skin applications [3]. This paradigm is now supported by both meta-analyses and mechanistic studies, offering a clear explanation for the observed variability in HA’s effects. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
IL-6 and IL-10: Key Biomarkers of Hyaluronic Acid’s Opposing Actions
IL-6 and IL-10 serve as opposing biomarkers for hyaluronic acid's activity: LMW-HA increases IL-6 by 40-60% in most studies, promoting inflammation, while HMW-HA elevates IL-10 by 25-35%, dampening immune responses. These cytokines provide a measurable framework for tracking HA's physiological impact.
A systematic review of 11 studies confirmed that LMW-HA supplementation leads to significant rises in IL-6, correlating with increased tissue inflammation and immune activation [1]. Conversely, HMW-HA consistently increases IL-10 production, an anti-inflammatory cytokine associated with improved tissue repair and reduced swelling. In a 12-week trial with 282 participants, topical HMW-HA eye drops reduced tear IL-6 levels while improving clinical symptoms [2,4].
For those tracking inflammation, optimal serum IL-6 levels in healthy adults are typically below 2 pg/mL, while higher levels suggest acute or chronic inflammation. IL-10 is measured less frequently, but higher circulating levels generally indicate better inflammatory control and tissue healing capacity. These markers respond to HA supplementation within 4-12 weeks depending on the molecular weight used, making product transparency essential for predictable outcomes.
Formulation Matters: Delivery, Dosage, and Molecular Size in Human Outcomes
The clinical outcome of hyaluronic acid supplementation depends on its delivery form, dosage, and especially the molecular weight profile. Topical, oral, and injectable formulations each have distinct bioavailability and tissue targeting characteristics, with HMW-HA showing superior anti-inflammatory and hydrating effects in most settings.
Meta-analyses confirm that oral HMW-HA (120-240 mg/day) improves skin hydration, while injected cross-linked HMW-HA is more effective for joint lubrication and inflammation [5]. A 1-year RCT comparing single versus repeated intra-articular HMW-HA doses found both regimens reduced pain and improved function in knee osteoarthritis, but the effect was more sustained with repeated dosing [6,7]. Dosages in these trials typically range from 20-40 mg per joint injection, with cross-linked forms prolonging HA’s presence in tissues. Topical HMW-HA (0.1-0.3%) has also been shown to reduce inflammatory markers in the skin and eyes [2].
Most over-the-counter oral supplements are poorly characterized for molecular weight, but medical-grade injectables and skincare often specify HMW-HA content. For maximized anti-inflammatory outcomes, selecting products labeled as HMW-HA (≥1,000 kDa) is recommended. Lower molecular weight or hydrolyzed forms may be better suited for wound healing or skin penetration, but carry a greater risk of pro-inflammatory responses. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Mechanistic Pathways: Receptor Interactions and Cellular Effects
Hyaluronic acid’s molecular size dictates its receptor interactions and downstream effects. HMW-HA primarily engages CD44 and suppresses inflammatory cell activation, while LMW-HA activates toll-like receptors (TLRs), driving pro-inflammatory signaling and cytokine release. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Preclinical studies detail that HMW-HA binding to CD44 on immune and epithelial cells leads to enhanced tissue repair, cell migration, and upregulation of IL-10, supporting resolution of inflammation [1]. In contrast, LMW-HA fragments, generated by enzymatic breakdown or oxidative stress, activate TLR2 and TLR4, which stimulate NF-κB and increase IL-6 production [1,8]. These opposing pathways have been replicated in animal models and are increasingly being confirmed in human tissue studies, particularly in joint, ocular, and skin tissues [2,4].
A summary of receptor interactions is shown below:
| Molecular Weight | Primary Receptor | Downstream Effect | |-------------------------|-------------------|---------------------------------| | High (>1,000 kDa) | CD44 | ↑ IL-10, anti-inflammatory | | Low (<500 kDa) | TLR2/TLR4 | ↑ IL-6, pro-inflammatory |
Understanding these pathways is critical for interpreting clinical results and optimizing product selection. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Hydration and Skin Health: Evidence for Molecular Size-Dependent Benefits
Hyaluronic acid’s skin hydration effects are strongly tied to molecular weight: HMW-HA creates a robust water-retaining matrix, while LMW-HA can penetrate deeper but may stimulate inflammation. Meta-analyses show that HMW-HA consistently improves hydration and barrier function. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A meta-analysis of seven RCTs found that oral and topical HMW-HA (≥1,000 kDa) supplementation significantly increased skin hydration (standardized mean difference = 1.34, 95% CI: 0.14-2.54) compared to placebo, with benefits observed after 4-12 weeks of use [5]. Products containing LMW-HA showed mixed results, sometimes improving penetration but also raising local IL-6 and transient redness [1]. In clinical practice, injectable HMW-HA fillers are preferred for long-lasting hydration and support of skin structure, while lower-weight forms are used selectively for deeper tissue penetration or in combination with other agents.
For readers seeking to optimize skin hydration, formulations specifying HMW-HA content and applied at dosages used in RCTs (topical 0.1-0.3%, oral 120-240 mg/day) are most reliable. These effects are independent of biomarker testing but can be tracked via improvements in skin texture and reduced dryness. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Joint Health: Variable Effects of Hyaluronic Acid Linked to Molecular Weight
Joint health outcomes with hyaluronic acid supplementation are highly variable and linked to the molecular size of the administered HA. HMW-HA provides superior joint lubrication and inflammation control compared to LMW-HA, which may be less effective or even pro-inflammatory. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Human RCTs confirm that intra-articular injections of HMW-HA (20-40 mg/injection) improve pain and function in osteoarthritis, with repeated dosing providing sustained benefits [6,7]. The anti-inflammatory action is attributed to HMW-HA’s ability to increase IL-10 and reduce IL-6 in synovial fluid, while LMW-HA fragments, produced by degradation, may promote synovitis and joint swelling [1,4]. Animal models further reveal that HMW-HA preserves cartilage and inhibits inflammatory cell infiltration, whereas LMW-HA can exacerbate joint inflammation [8].
For joint support, products specifying cross-linked or high molecular weight HA are best supported by evidence. Single-dose regimens are convenient, but repeated treatments may yield better outcomes. The lack of molecular weight labeling in many over-the-counter products remains a major limitation for consumer decision-making. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
The molecular weight of hyaluronic acid is the primary determinant of whether it acts as an anti- or pro-inflammatory agent in the body. High molecular weight forms upregulate IL-10 and support tissue hydration and repair, while low molecular weight fragments stimulate IL-6 and can provoke inflammation. These size-dependent mechanisms, now confirmed in systematic reviews and RCTs, provide a mechanistic explanation for the wide variability in clinical results with hyaluronic acid supplementation. For the greatest anti-inflammatory and hydration benefits, products specifying high molecular weight or cross-linked HA are preferred, particularly for joint and skin applications. As research advances, clearer labeling and formulation standards will further optimize outcomes for users. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
Despite compelling mechanistic and clinical evidence, major limitations persist. Many commercial hyaluronic acid products do not specify molecular weight, making it difficult to predict their effects. Most RCTs focus on short- to medium-term outcomes, with limited data on long-term safety or efficacy. The influence of delivery route, individual metabolism, and potential for LMW-HA generation in vivo are not fully understood. Further human studies directly comparing defined molecular weight fractions are needed to refine recommendations and confirm optimal dosing strategies. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how hyaluronic acid relates to your health goals and monitor changes in your biomarkers over time.
