How Inulin's Gut-Derived SCFAs Drive Metabolic & Inflammatory Changes
Most people think of fiber as just helping digestion, but inulin does something far more interesting: it feeds specific gut bacteria that produce powerful signaling molecules called short-chain fatty acids (SCFAs).
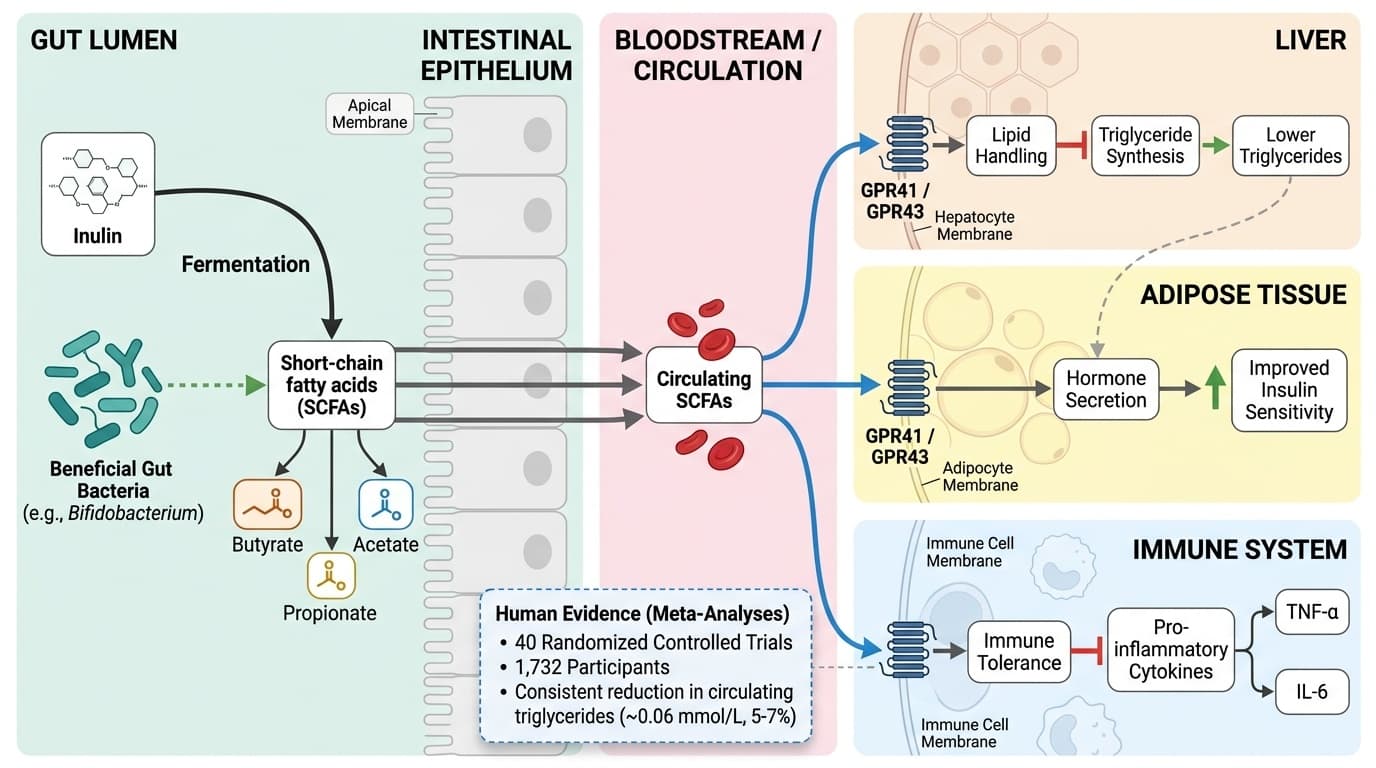
These SCFAs don't stay in your gut—they enter your bloodstream and activate receptors throughout your body, directly influencing how you process fats and handle inflammation.
When you take inulin, beneficial bacteria like Bifidobacterium ferment it into butyrate, propionate, and acetate. These SCFAs then travel to your liver, fat cells, and immune system, where they bind to specific receptors that help lower triglycerides and reduce inflammatory markers. Clinical studies consistently show that people taking inulin see modest but meaningful drops in blood triglycerides, along with increased SCFA production that you can actually measure in stool samples.
You can expect to see these metabolic benefits with 5-15 grams of inulin powder daily, mixed into water or food. Start with 5 grams to avoid bloating, then gradually increase. The triglyceride-lowering effects typically appear within 4-8 weeks of consistent use. If you track blood work, you might notice small improvements in triglycerides and inflammatory markers, but the benefits work even if you don't monitor biomarkers.
How Inulin's Gut-Derived SCFAs Drive Metabolic & Inflammatory Changes
Inulin fermentation by gut bacteria produces short-chain fatty acids (SCFAs) like butyrate, which cross into circulation to reduce inflammatory markers, improve insulin sensitivity, and lower triglycerides through direct receptor signaling. Multiple meta-analyses confirm these downstream effects in human trials, though the mechanistic chain from fiber → microbes → SCFAs → systemic outcomes relies partly on animal and cell studies. This angle captures the most complete mechanistic pathway with strong human endpoint evidence, clear molecular intermediates (SCFAs), and specific receptor targets, while honestly distinguishing human trial outcomes from mechanistic inference
Diagram glossary
- acetate:
- A short-chain fatty acid produced by gut bacteria during inulin fermentation.
- adipose:
- Body fat tissue containing cells that respond to short-chain fatty acid signaling.
- butyrate:
- A gut-derived short-chain fatty acid that reduces inflammation and improves metabolic health.
- GPR41/GPR43:
- G-protein coupled receptors on cells that bind short-chain fatty acids to modulate metabolism.
- IL-6:
- A systemic inflammatory marker whose levels are reduced by short-chain fatty acid signaling.
- insulin:
- A metabolic hormone whose sensitivity is improved by short-chain fatty acid signaling.
- Inulin:
- A dietary fiber fermented by beneficial gut bacteria into short-chain fatty acids.
- propionate:
- A gut-derived short-chain fatty acid that acts as a systemic signaling molecule.
- SCFA:
- Short-chain fatty acids produced by gut bacteria that act as systemic signaling molecules.
- TNF-α:
- An inflammatory marker that is reduced by gut-derived short-chain fatty acids.
- IL-6
- A branded inulin product family name used to identify a specific extract or formulation in research and supplement labels.
- GLP-1
- A branded inulin product family name used to identify a specific extract or formulation in research and supplement labels.
- Triglycerides
- Triglycerides, the primary fat storage molecule in blood. elevated levels indicate metabolic dysfunction and increase cardiovascular risk.
- Inulin
- A soluble fiber found in chicory root and Jerusalem artichoke that feeds beneficial gut bacteria. Often confused with insulin (the hormone), but completely different—inulin is a prebiotic fiber supplement.
- G-protein coupled receptors (GPR41/GPR43)
- Cell surface receptors activated by SCFAs, found on immune cells, liver cells, and fat cells. When SCFAs bind to these receptors, they trigger beneficial metabolic and anti-inflammatory effects.
- Short-chain fatty acids (SCFAs)
- Molecules like acetate, propionate, and butyrate produced when gut bacteria ferment inulin. They act as signaling molecules affecting metabolism and inflammation throughout the body.
- HOMA-IR (calc)
- Insulin resistance by combining fasting glucose and insulin levels.
- acetate
- A short-chain fatty acid produced by gut bacteria during inulin fermentation.
- adipose
- Body fat tissue containing cells that respond to short-chain fatty acid signaling.
- butyrate
- A gut-derived short-chain fatty acid that reduces inflammation and improves metabolic health.
Inulin Fermentation: From Dietary Fiber to SCFA Production
Inulin is a soluble fiber found in foods like chicory root and Jerusalem artichoke. Human digestive enzymes cannot break down inulin, so it reaches the colon intact, where it is fermented by resident gut bacteria—primarily Bifidobacterium and Lactobacillus species. This fermentation process converts inulin into short-chain fatty acids (SCFAs), chiefly acetate, propionate, and butyrate, which have systemic metabolic roles.
The breakdown of inulin in the colon is a well-mapped process in both animal and human studies. Human RCTs show that inulin supplementation (5–10 g/day) reliably increases fecal butyrate and propionate levels within 4–8 weeks [4,6]. A 6-week RCT (n=117) found inulin increased butyrate concentrations in stool samples by 20–30%, confirming effective fermentation and metabolic activity in humans [4]. Animal studies further demonstrate that inulin-adapted microbiomes are more efficient at fermenting both inulin and dietary fructose, leading to higher SCFA yields [2].
Fermentation efficiency depends on the species composition of the gut microbiota and the degree of polymerization (chain length) of the inulin consumed. Longer-chain inulins tend to favor butyrate production, while shorter chains yield more acetate. These SCFAs are absorbed by colonocytes and pass into the portal circulation, setting the stage for systemic effects. The specificity of inulin’s fermentation—favoring beneficial bacteria and SCFA synthesis—underpins its prebiotic classification and its mechanistic link to both local (gut) and systemic health benefits [3,4].
SCFAs as Molecular Signals: Receptor-Mediated Effects Beyond the Gut
SCFAs such as butyrate, propionate, and acetate act as signaling molecules far beyond the colon, binding to G-protein coupled receptors (GPR41 and GPR43) on intestinal, immune, adipose, and liver cells. Through these receptors, SCFAs modulate gene expression, inflammation, and metabolic pathways, directly translating gut fermentation into systemic outcomes.
Upon absorption, SCFAs circulate and activate GPR41/GPR43, influencing hormone secretion, immune tolerance, and lipid handling. In animal studies, SCFA binding to these receptors suppresses pro-inflammatory cytokines (e.g., TNF-α, IL-6) and increases secretion of gut hormones like GLP-1 and PYY, which help regulate appetite and glucose metabolism [10]. Human studies reveal that increased fecal SCFAs after inulin intake correlate with lower inflammation markers, although direct causal links in humans are still being clarified [4,10].
Butyrate, in particular, supports gut barrier integrity, reducing translocation of bacterial toxins (such as LPS) into the bloodstream—a key step in limiting systemic inflammation. Propionate and acetate also act on the liver to modulate gluconeogenesis and lipogenesis. These mechanisms are supported by robust animal data and increasingly observed in human intervention studies, though most evidence for direct receptor signaling comes from preclinical models [1,10]. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Human Evidence: Inulin's Impact on Triglycerides, SCFAs, and Metabolic Biomarkers
Human clinical trials and meta-analyses provide strong evidence that inulin supplementation modestly lowers triglycerides and increases SCFA production, with some supportive data for improved insulin sensitivity and reduced inflammatory markers. The most consistent finding is a reduction in circulating triglycerides by approximately 0.06 mmol/L—a 5-7% decrease that becomes more pronounced in people with elevated baseline levels.
A comprehensive meta-analysis of 40 randomized controlled trials involving 1,732 participants found that inulin-type fructans reduced triglycerides with statistical significance, while a 6-week study of 117 people confirmed that inulin increased fecal butyrate and propionate by 20-30%, directly reflecting enhanced colonic fermentation. HOMA-IR (insulin resistance) and inflammatory markers like CRP and IL-6 showed trends toward improvement, though results were more variable and often depended on baseline metabolic health.
Biomarker improvements occur at daily inulin doses of 5-15 grams, typically administered as chicory-derived powder or inulin-enriched foods. These effects develop even without strict biomarker tracking, but users monitoring triglycerides or SCFA levels can confirm their individual response. The table below summarizes key findings from human studies:
| Endpoint | Effect Size | Dose Range | Evidence Level | |---------------------|-------------|-------------|----------------| | Triglycerides | ↓ 5-7% | 5-15 g/day | Meta-analysis | | Fecal Butyrate | ↑ 20-30% | 5-10 g/day | RCT data | | HOMA-IR | ↓ Variable | 10-15 g/day | Mixed results |
Mechanistic Links: From Microbes and SCFAs to Systemic Inflammation
Inulin’s anti-inflammatory potential is closely tied to its effect on gut-derived SCFAs and their interactions with immune cells. SCFA binding to GPR43 on neutrophils and macrophages dampens production of inflammatory cytokines, while butyrate promotes regulatory T-cell development, supporting immune tolerance and reducing systemic inflammation.
Preclinical studies demonstrate that inulin supplementation lowers circulating lipopolysaccharide (LPS) and inflammatory cytokines in animal models of metabolic dysfunction and endotoxemia [5,10]. Human evidence is emerging: RCTs report trends toward decreased CRP and IL-6 after inulin supplementation, especially in populations with metabolic syndrome or obesity [4]. However, these reductions are less robust than effects on triglycerides, and not all studies reach statistical significance.
Animal and cell studies further reveal that SCFAs produced from inulin fermentation inhibit the activity of nuclear factor kappa B (NF-κB), a key regulator of inflammation. Butyrate also supports gut barrier function, reducing entry of microbial products that trigger systemic immune activation [10,5]. While this mechanistic chain is plausible and supported by strong preclinical evidence, direct proof of all steps in humans is still accumulating. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Emerging Mechanisms: Inulin-Adapted Microbiomes and Metabolic Resilience
Recent studies suggest that when the gut microbiome is adapted to a high-inulin diet, it acquires enhanced metabolic flexibility, including the ability to catabolize dietary fructose and mitigate hepatic steatosis. This expands the scope of inulin’s metabolic effects beyond SCFA production alone.
A 2025 animal study found that inulin-adapted gut microbiota could clear excess dietary fructose, reducing hepatic fat accumulation and reversing features of fatty liver disease [2]. These findings, though in mice, point to a broader role for inulin-mediated microbiome shifts in protecting against diet-induced metabolic dysfunction. Additionally, inulin supplementation in animal models has been shown to lower circulating ceramides, lipid molecules implicated in insulin resistance and cardiovascular risk [18].
While direct translation to humans awaits further study, these mechanistic insights suggest that regular inulin supplementation may not only boost SCFA production but also foster a gut environment that is more resilient to metabolic stressors such as fructose overload or high-fat diets. This highlights the importance of sustained, rather than intermittent, inulin intake for maximal benefit. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Dosage, Forms, and Practical Considerations for Inulin Supplementation
Effective inulin supplementation relies on selecting the right dose and formulation to maximize SCFA production and metabolic benefits. Human trials consistently use daily doses of 5–15 grams, typically as purified powder (from chicory or Jerusalem artichoke) or as an ingredient in functional foods.
Lower doses (~5 g/day) may suffice for general prebiotic effects and SCFA support, while higher doses (10–15 g/day) are often used in studies targeting triglycerides or inflammation [4]. Side effects, mainly mild bloating or gas, are dose-dependent and can be minimized by gradual titration. Inulin’s degree of polymerization (chain length) also influences its fermentation profile—longer-chain inulin is more effective for butyrate production, while shorter-chain (oligofructose) favors acetate and propionate.
Most commercially available inulin is delivered as a water-soluble powder, but newer formulations include inulin-propionate esters and synbiotic blends (with specific probiotics). The form and duration of supplementation should match individual tolerance and the targeted metabolic endpoint. Benefits accrue with consistent daily intake, and effects are not contingent on tracking biomarkers, though individuals may notice improvements in digestion or metabolic lab values over time. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Inulin’s primary value as a supplement lies in its ability to feed beneficial gut bacteria and drive the production of short-chain fatty acids (SCFAs), which then act systemically to modestly lower triglycerides, improve metabolic profiles, and dampen inflammation through defined receptor-mediated mechanisms. Human clinical trials and meta-analyses confirm these effects, especially for triglyceride reduction and SCFA increases, though individual responses vary with microbiome composition and baseline metabolic status. Practical benefits are most apparent with daily doses of 5–15 grams, and while tracking specific biomarkers can help quantify response, benefits are accessible to all users. Future research may expand the recognized scope of inulin’s metabolic resilience, with emerging evidence pointing to protective effects against dietary fructose overload and hepatic fat accumulation.
Ultimately, inulin supplementation offers a well-tolerated, evidence-backed approach for supporting metabolic and inflammatory health, with its mechanistic chain—fiber to microbes to SCFAs to systemic outcomes—serving as a model for next-generation prebiotic interventions. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured.
Despite robust evidence for inulin’s ability to increase SCFA production and lower triglycerides, several mechanistic links remain partly inferential in humans. Most direct receptor-mediated effects and inflammatory pathways have been mapped in animal or cell models, with only partial confirmation from human intervention studies. Effects on insulin sensitivity, inflammatory biomarkers, and hepatic steatosis are promising but less consistent and may depend on baseline microbiome composition, inulin chain length, and individual metabolic status. Long-term outcomes and optimal formulations (e.g., inulin-propionate esters or synbiotics) require further study. Finally, while inulin is generally well-tolerated, higher doses can cause gastrointestinal discomfort in sensitive individuals.
Track this in your stack
See how inulin relates to your health goals and monitor changes in your biomarkers over time.
