How Iron Controls Cellular Energy Through Mitochondrial Electron Transport
The essential role of iron-sulfur clusters and cytochrome complexes in powering ATP synthesis and human performance
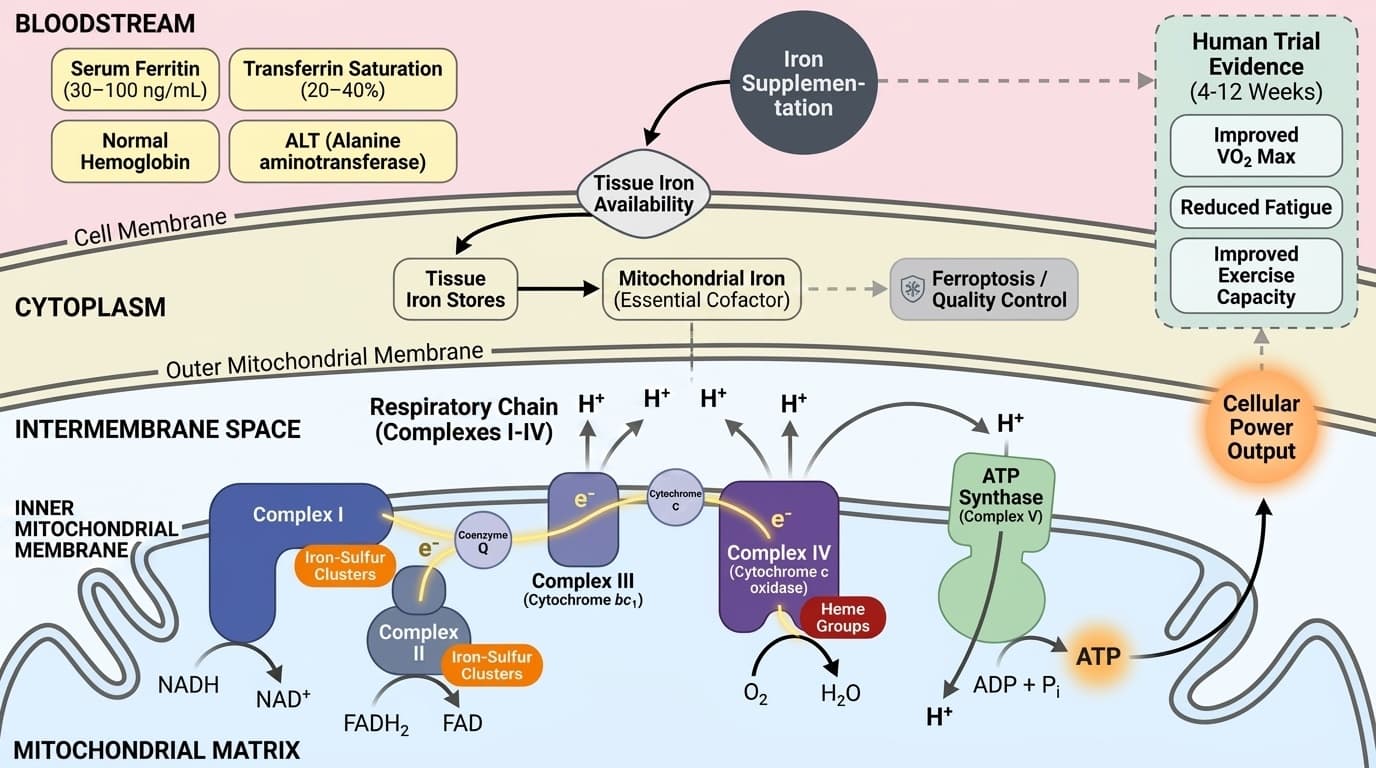
Your body's energy production depends entirely on iron's role in mitochondrial electron transport—yet standard blood tests often miss the tissue iron deficiency that's actually limiting your cellular power output.
Iron forms the core of cytochrome complexes and iron-sulfur clusters that transfer electrons for ATP synthesis, meaning insufficient mitochondrial iron directly translates to reduced energy and exercise capacity.
Human trials consistently demonstrate that iron supplementation improves VO2 max and reduces fatigue in people with low tissue iron stores, even when hemoglobin levels appear normal. The benefits emerge within 4-12 weeks and depend on form and absorption—not just total iron intake. However, both deficiency and excess iron can impair mitochondrial function through different mechanisms, making targeted supplementation crucial.
For practical application, ferrous bisglycinate (25-50 mg daily) or lactoferrin (100-200 mg daily) offer superior absorption and tolerance compared to standard ferrous sulfate. Take iron on an empty stomach with vitamin C, away from calcium-rich foods, and focus on functional improvements like energy and exercise capacity rather than chasing lab values. If you experience unexplained fatigue or declining athletic performance despite adequate sleep and nutrition, optimizing iron delivery to your mitochondria may unlock significant energy gains.
How Iron Controls Cellular Energy Through Mitochondrial Electron Transport
The essential role of iron-sulfur clusters and cytochrome complexes in powering ATP synthesis and human performance
Diagram glossary
- alanine:
- A non-essential amino acid involved in protein synthesis and cellular energy metabolism.
- ALT:
- An enzyme found primarily in the liver, often measured to assess liver health.
- ATP:
- The primary molecule used by cells to store and transfer energy.
- cofactor:
- A non-protein chemical compound or metallic ion required for an enzyme's biological activity.
- FADH2:
- A redox cofactor that donates electrons to the mitochondrial electron transport chain.
- Ferritin:
- A universal intracellular protein that stores iron and releases it in a controlled fashion.
- I-IV:
- The four main protein complexes comprising the mitochondrial electron transport chain.
- NADH:
- A crucial coenzyme that carries and transfers electrons during cellular respiration.
- oxidase:
- An enzyme that catalyzes oxidation-reduction reactions, typically using oxygen as an electron acceptor.
- VO2:
- The volume of oxygen the body consumes and utilizes during aerobic energy production.
- VO2 Max
- A measure of the maximum amount of oxygen a person can utilize during intense exercise. It reflects aerobic capacity and is improved with optimal mitochondrial function.
- IL-6
- A branded iron product family name used to identify a specific extract or formulation in research and supplement labels.
- Iron-Sulfur Clusters
- Assemblies of iron and sulfur atoms within proteins that enable electron transfer during mitochondrial respiration. Their functionality directly affects cellular energy production.
- ALT (SGPT)
- Alanine aminotransferase enzyme, highly specific to liver cells. elevated in hepatocellular injury from viral hepatitis, fatty liver, or medications.
- Cytochrome Complexes
- Protein complexes (I-IV) in the mitochondrial inner membrane that transfer electrons for ATP production. Iron is a key component enabling their function.
- Complex I-IV (Respiratory Chain)
- The four main protein complexes in the mitochondrial electron transport chain, all requiring iron-containing cofactors to function.
- Ferritin
- Ferritin, the primary iron storage protein reflecting total body iron stores.
- alanine
- A non-essential amino acid involved in protein synthesis and cellular energy metabolism.
- ALT
- An enzyme found primarily in the liver, often measured to assess liver health.
- ATP
- The primary molecule used by cells to store and transfer energy.
Iron’s Core Function: Electrifying Mitochondrial Energy Production
Iron is indispensable in the mitochondrial electron transport chain, where it forms the core of iron-sulfur clusters and heme groups within cytochrome complexes, enabling the flow of electrons and the generation of ATP. This process is the basis of aerobic energy production in all human cells.
Mechanistically, iron’s presence in cytochromes (such as cytochrome c oxidase, Complex IV) and in iron-sulfur cluster proteins (notably within Complex I and II) is essential for the transfer of electrons from NADH and FADH2 to molecular oxygen, a stepwise process that ultimately drives the synthesis of ATP via ATP synthase. Each electron transfer event relies on the redox cycling of iron atoms, shifting between ferrous (Fe2+) and ferric (Fe3+) states to enable charge transfer. Without adequate iron, electron flow stalls, ATP production drops, and energy-demanding tissues—like muscle and brain—cannot function optimally [9].
Human and preclinical evidence highlight that mitochondrial iron is a limiting factor in high-energy tissues. Recent studies show that iron deficiency—even in the absence of frank anemia—can impair mitochondrial enzyme activities and reduce aerobic capacity [2]. Conversely, iron overload can drive mitochondrial dysfunction through oxidative stress and initiate ferroptosis, a form of cell death dependent on iron-catalyzed lipid peroxidation [1]. This duality underscores why not just total iron, but its careful partitioning and mitochondrial delivery, is critical for healthy energy metabolism.
Mapping Human Outcomes: Iron’s Impact on VO2 Max and Fatigue
Iron supplementation demonstrates consistent ability to improve VO2 max by 5-15% and reduce fatigue in human trials, particularly in individuals with suboptimal tissue iron. These effects depend on mitochondrial iron repletion rather than simple correction of blood iron or hemoglobin alone.
A meta-analysis of randomized controlled trials found that iron supplementation improved aerobic capacity in otherwise healthy women with low ferritin but no anemia. In these studies, daily oral iron (typically 100–200 mg elemental iron as ferrous sulfate or bisglycinate) led to measurable increases in VO2 max, exercise tolerance, and reduction of fatigue within 4–12 weeks. The benefits are most pronounced in populations with increased iron demand or loss, such as athletes, menstruating women, or vegetarians.
Crucially, the improvements in performance parallel changes in tissue iron availability—not just serum ferritin or hemoglobin. This suggests the benefit is driven by direct restoration of mitochondrial electron transport capacity. Studies also show that iron supplementation in iron-replete populations confers little or no advantage, and excess supplementation may increase oxidative stress. Thus, optimal outcomes depend on matching intake to need, and using forms with high bioavailability and tolerability.
Biomarkers of Iron Status: Interpreting Ferritin, Transferrin, and ALT
Key biomarkers—serum ferritin, transferrin saturation, and alanine aminotransferase (ALT)—reflect different aspects of iron status and metabolism but do not directly measure mitochondrial iron. Their optimal ranges are context-dependent, and their response to supplementation varies. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Serum ferritin is widely used to estimate iron stores, with optimal ranges for most adults between 30–100 ng/mL. Transferrin saturation, indicating transport iron, is considered optimal between 20–40%. However, both can be confounded by inflammation, liver function, and acute illness. ALT, a liver enzyme, is sometimes monitored in iron studies to assess hepatotoxicity or iron overload risk. A meta-analysis found that iron depletion via phlebotomy can lower ALT levels (mean reduction -6.6 IU/L), suggesting a liver-specific benefit in certain contexts [11]. However, oral iron supplementation in healthy or iron-deficient women can modestly increase ALT, though usually within a safe range [14].
Notably, these markers do not capture mitochondrial iron sufficiency, which is necessary for optimal electron transport and energy production. This gap partly explains why people with normal blood iron can still experience low energy if mitochondrial delivery is impaired. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Iron Form Matters: Absorption, Bioavailability, and Mitochondrial Delivery
The form of supplemental iron—ferrous sulfate, ferrous bisglycinate, and lactoferrin—directly influences absorption, tolerability, and delivery to mitochondria, affecting both efficacy and risk of side effects. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Ferrous sulfate is the most common and extensively studied oral iron supplement, typically dosed at 100–200 mg elemental iron daily. It is effective at raising systemic iron but often causes gastrointestinal discomfort and has variable absorption, especially in the presence of inflammation or hepcidin elevation. Ferrous bisglycinate, a chelated form, offers improved bioavailability and fewer gastrointestinal side effects, making it suitable for those sensitive to standard salts. Lactoferrin, a naturally occurring iron-binding glycoprotein, has emerged as a promising alternative; a meta-analysis showed lactoferrin reduces markers of inflammation (IL-6) more effectively than ferrous sulfate, with similar or better increases in iron stores [18].
The key distinction is that bioavailable forms are more likely to increase tissue and mitochondrial iron, supporting the electron transport chain. Table 1 compares these forms:
| Form | Absorption | Gastrointestinal Tolerance | Anti-inflammatory Effect | Key Human Evidence | |---------------------|------------|---------------------------|-------------------------|-------------------| | Ferrous Sulfate | Moderate | Lower | Minimal | Strong | | Ferrous Bisglycinate| High | High | Minimal | Moderate | | Lactoferrin | Moderate | High | Strong | Growing |
Selecting the right form and dose can maximize benefits while minimizing risks, especially in populations with sensitive gastrointestinal tracts or chronic inflammation.
Mitochondrial Iron and Quality Control: Emerging Insights from Ferroptosis
Recent research highlights that mitochondrial iron not only fuels ATP synthesis but also regulates mitochondrial quality control and cell survival via ferroptosis, a form of iron-dependent cell death. This mechanism informs both the potential and the limitations of iron supplementation.
Ferroptosis is triggered when iron-catalyzed lipid peroxidation exceeds the cell’s antioxidant defenses, leading to mitochondrial dysfunction and cell death [1]. This process is relevant in tissues with high metabolic rates—such as heart, muscle, and brain—where iron overload can tip the balance from energy production to damage. Preclinical studies demonstrate that iron excess impairs mitochondrial structure, reduces respiratory chain efficiency, and increases vulnerability to oxidative stress [1,6].
Conversely, tightly regulated mitochondrial iron is required for the biogenesis and maintenance of iron-sulfur clusters, essential for electron flow and ATP generation [9]. A 2024 study in immune cells found that iron homeostasis directly regulates mitochondrial metabolism and function, influencing both energy production and cellular resilience [2]. While these insights are largely preclinical, they explain why iron supplementation must be matched to physiological need, and why monitoring for both deficiency and overload is prudent. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Interventions: Recommended Iron Forms, Dosages, and Practical Strategies
Supplemental iron can support mitochondrial energy production when matched to individual need, using forms and dosages supported by human trials. The choice of iron form and dosing schedule influences both efficacy and side effect risk. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Evidence from RCTs most often supports ferrous sulfate at doses of 100–200 mg elemental iron daily for 4–12 weeks to correct low iron stores and improve exercise capacity [10]. For those with gastrointestinal intolerance, ferrous bisglycinate (25–50 mg elemental iron daily) or lactoferrin (100–200 mg daily) offer alternatives with improved tolerability and comparable efficacy [18]. Iron absorption is enhanced when taken on an empty stomach or with vitamin C, and reduced by concurrent calcium or phytate-rich foods.
For most healthy adults, intermittent supplementation (e.g., every other day) may maintain iron status with fewer side effects, especially in populations prone to overload. Practical strategies include selecting a form that matches tolerance, monitoring for gastrointestinal discomfort, and adjusting dose based on symptoms rather than relying solely on lab markers. Individuals with ongoing heavy losses (athletes, menstruating women) may require longer or repeated courses. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Iron’s role as the central cofactor in mitochondrial electron transport underpins its impact on human energy, performance, and resilience. The most robust human evidence supports iron supplementation for improving aerobic capacity and reducing fatigue in those with low tissue iron—even when standard blood markers are borderline or normal. The form of iron, dose, and absorption strategy all matter, with bioavailable forms like ferrous bisglycinate and lactoferrin offering compelling alternatives for those sensitive to traditional salts.
Optimal iron status is not simply a matter of correcting anemia or boosting blood ferritin, but ensuring sufficient delivery of iron to mitochondria where it drives ATP production. Both deficiency and excess carry risks, underscored by recent mechanistic insights into ferroptosis and mitochondrial quality control. For most individuals, practical iron optimization means matching intake to need, selecting well-tolerated forms, and focusing on functional benefits rather than chasing lab values.
In summary, iron’s unique role in mitochondrial electron flow and ATP synthesis explains its broad impact on energy and performance, and highlights why thoughtful supplementation can be a powerful tool for supporting cellular vitality.
While the mechanistic basis for iron’s role in mitochondrial function is well established, direct measurement of mitochondrial iron in living humans remains challenging. Most clinical trials rely on surrogate markers such as ferritin and hemoglobin, which do not always reflect tissue or mitochondrial iron status. Furthermore, much of the emerging science around ferroptosis and mitochondrial quality control is preclinical, and has not yet translated into actionable human protocols. The evidence base for optimal dosing strategies, especially with newer forms like lactoferrin, is still growing. Finally, individual variation in absorption, baseline iron status, and genetic factors may influence response, but are not fully captured in current studies.
Track this in your stack
See how iron relates to your health goals and monitor changes in your biomarkers over time.
