How Lactobacillus Strains Lower Liver Enzymes Through Gut Barrier Repair
Mechanisms, human trial evidence, and practical interpretation for liver health biomarkers
Your gut barrier controls more than digestion—it directly influences liver health through a pathway most people never consider.
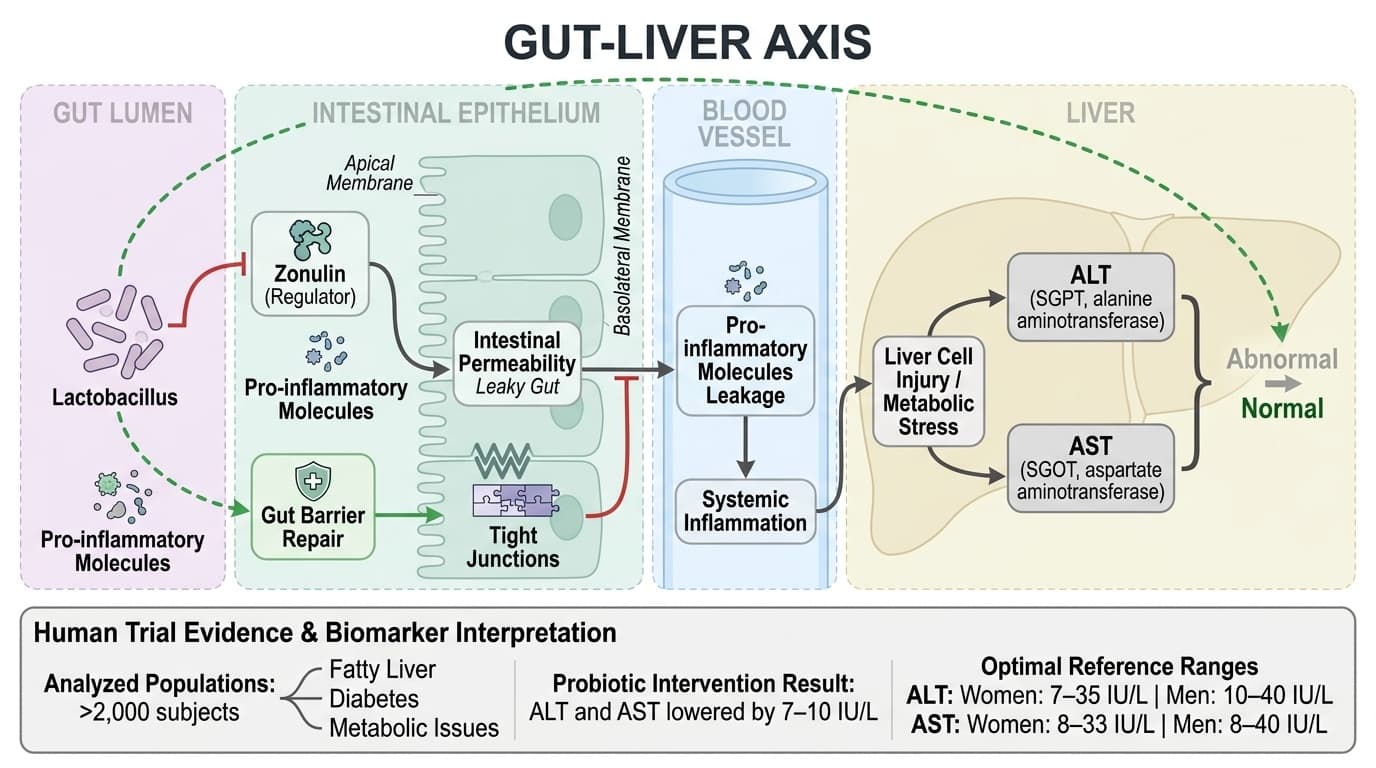
When intestinal permeability increases (measured by a protein called zonulin), inflammatory molecules leak into your bloodstream and stress your liver, causing enzyme levels like ALT and AST to rise. This gut-liver connection explains why probiotics can improve liver function even when you're not specifically targeting liver problems.
Multiple large-scale studies involving over 2,000 people with fatty liver, diabetes, or metabolic issues show that specific probiotic combinations lower liver enzymes by 7-10 IU/L within 8-12 weeks. The most effective approach uses three bacterial strains together: Lactobacillus, Bifidobacterium, and Streptococcus. This combination outperforms single-strain products by strengthening gut barrier function and reducing zonulin levels, which directly translates to less liver stress.
To support your liver health through gut barrier repair, choose a multi-strain probiotic containing all three bacterial types in enteric-coated capsules with 1-10 billion CFUs daily. Take it consistently for at least 8 weeks to see meaningful changes in liver enzymes. This approach works whether you currently have elevated liver markers or want to prevent future issues, since the mechanism operates through general improvements in gut permeability rather than requiring baseline dysfunction.
How Lactobacillus Strains Lower Liver Enzymes Through Gut Barrier Repair
Mechanisms, human trial evidence, and practical interpretation for liver health biomarkers
Diagram glossary
- alanine:
- An amino acid that is a key component of the liver enzyme ALT.
- ALT:
- A liver enzyme whose elevated levels indicate liver stress or damage.
- aminotransferase:
- A type of enzyme, like ALT or AST, that transfers amino groups.
- aspartate:
- An amino acid that is a key component of the liver enzyme AST.
- AST:
- A liver enzyme used as a biomarker to assess liver health and function.
- intestine:
- The digestive organ responsible for nutrient absorption and maintaining the gut barrier.
- IU/L:
- International Units per Liter, a standard unit used to measure blood enzyme levels.
- SGOT:
- An older medical term for AST, an enzyme indicating liver function.
- SGPT:
- An older medical term for ALT, a key enzyme biomarker for liver health.
- Zonulin:
- A protein biomarker that regulates and indicates the permeability of the intestinal barrier.
- Enteric-coated
- A dosage form designed to resist stomach acid and release later in the intestine.
- IL-10
- A branded lactobacillus product family name used to identify a specific extract or formulation in research and supplement labels.
- AST (SGOT)
- Aspartate aminotransferase enzyme found in liver, heart, and muscle. elevated in liver damage, heart attack, or muscle injury.
- ALT (SGPT)
- Alanine aminotransferase enzyme, highly specific to liver cells. elevated in hepatocellular injury from viral hepatitis, fatty liver, or medications.
- IL-6
- A branded lactobacillus product family name used to identify a specific extract or formulation in research and supplement labels.
- Lactobacillus
- A genus of beneficial bacteria commonly used in probiotic supplements, known for supporting gut and immune health.
- Gut-liver axis
- The bidirectional communication pathway between the intestine and liver through blood circulation, immune signaling, and bacterial metabolites.
- alanine
- An amino acid that is a key component of the liver enzyme ALT.
- ALT
- A liver enzyme whose elevated levels indicate liver stress or damage.
- aminotransferase
- A type of enzyme, like ALT or AST, that transfers amino groups.
Lactobacillus and the Gut-Liver Axis: Mechanistic Overview
Lactobacillus influences liver health by repairing the gut barrier, thereby reducing the leakage of pro-inflammatory molecules into the bloodstream and, ultimately, the liver. This process lowers systemic inflammation and directly impacts liver enzymes such as ALT and AST. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Human and preclinical studies increasingly focus on the gut-liver axis, recognizing the intestine as a primary gateway for substances affecting liver stress. When the gut barrier weakens—often measured by elevated zonulin—bacterial fragments and toxins enter the portal circulation and reach the liver, triggering inflammation and higher liver enzyme production. Lactobacillus strains reinforce tight junctions between gut cells, reducing permeability and decreasing exposure to these triggers. This mechanism is supported by human meta-analyses showing that probiotic supplementation can lower zonulin levels and improve gut barrier integrity, which correlates with reductions in ALT and AST in people with metabolic dysfunction [1,2,3].
Mechanistic animal and cell studies provide further detail: Lactobacillus produces short-chain fatty acids (SCFAs) like butyrate and acetate, which help regulate immune responses and suppress inflammatory signaling in both the gut and liver [11]. While much of this preclinical research highlights potential, the consistency of human biomarker changes in meta-analyses strengthens the plausibility of this pathway operating in real-world settings.
ALT and AST: Biomarkers of Liver Function and What Moves Them
ALT (alanine aminotransferase) and AST (aspartate aminotransferase) are key liver enzymes that rise in response to liver cell injury or metabolic stress. Probiotic interventions containing Lactobacillus consistently reduce these markers by 7-10 IU/L in at-risk populations, often shifting individuals from abnormal back to normal ranges.
Optimal reference ranges for ALT are typically 7–35 IU/L for women and 10–40 IU/L for men, while AST ranges are 8–33 IU/L (women) and 8–40 IU/L (men). Multiple meta-analyses show probiotic combinations lowered ALT by 7.5–10 IU/L and AST by 6–10 IU/L in adults and children with fatty liver or metabolic syndrome [1,3,6]. These reductions are clinically significant because they represent meaningful decreases in liver cell stress and inflammation. The most robust decreases occur in those with baseline elevations, with less pronounced changes in healthy individuals.
Lactobacillus acts on these biomarkers by reducing gut-derived inflammatory stimuli that reach the liver through portal circulation. Clinical trials and meta-analyses demonstrate the greatest effect occurs with multi-strain formulations, particularly when Lactobacillus combines with Bifidobacterium and Streptococcus [1,6]. These triple-strain formulas outperform single-strain supplements by 3-5 IU/L additional reduction, likely due to synergistic effects on gut barrier repair and immune modulation.
Zonulin and Gut Permeability: The Central Mechanism Linking Lactobacillus to Liver Health
Zonulin is a key regulator of intestinal permeability and serves as a biomarker for 'leaky gut.' Lactobacillus supplementation lowers zonulin levels, thereby tightening the gut barrier and reducing the inflammatory burden on the liver. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Elevated zonulin is associated with increased passage of bacterial endotoxins and dietary antigens into the bloodstream, fueling systemic inflammation and hepatic stress. In clinical studies, reductions in zonulin closely parallel improvements in liver enzyme levels, particularly in people with non-alcoholic fatty liver disease (NAFLD) or type 2 diabetes [1,3]. Probiotic interventions—including Lactobacillus—have been shown to decrease zonulin by 10–30 ng/mL in these populations, correlating with improved liver and metabolic markers [1].
Mechanistically, Lactobacillus enhances the integrity of tight junction proteins such as occludin and claudin, directly reducing zonulin-mediated permeability. Animal studies further confirm this pathway, showing that Lactobacillus supplementation limits endotoxin translocation and suppresses liver inflammation [11]. While human data is strongest in metabolic and liver-risk groups, the underlying pathway is likely relevant for broader populations with subclinical barrier dysfunction. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Strain Combinations and Formulation: What Works Best for Lowering Liver Enzymes?
Multi-strain probiotic formulas containing Lactobacillus, Bifidobacterium, and Streptococcus show superior effects on liver enzyme reduction compared to single-strain products. The form and viability of the probiotic also significantly influence outcomes. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A network meta-analysis of 35 RCTs (n=2,212) found that the triple-strain combination lowered ALT by 1.67 standard deviations and AST by 1.95, outperforming single-strain interventions [1]. These results suggest an additive or synergistic effect, likely due to broader coverage of gut microbial niches and multi-pathway support for barrier repair. Enteric-coated capsules and high-CFU (colony-forming unit) formulations ensure that viable bacteria reach the intestine, critical for efficacy. Most trials use doses ranging from 1 to 10 billion CFUs per day, with higher doses generally producing greater biomarker shifts in adults [2,3].
Table: Comparison of Probiotic Formulations | Formulation | ALT Reduction (IU/L) | AST Reduction (IU/L) | Notes | |-------------------------------------------|----------------------|----------------------|---------------------------| | Lactobacillus + Bifidobacterium + Streptococcus | Up to 10 | Up to 10 | Highest efficacy, network meta-analysis [1] | | Single-strain Lactobacillus | 3–5 | 2–4 | Modest effect, less robust | | Multi-strain, not enteric-coated | Variable | Variable | Dependent on survival rate |
Dose-response relationships are evident, particularly for combinations, supporting the use of well-formulated, high-viability supplements.
Dosage, Duration, and Practical Supplementation Guidelines
To achieve meaningful reductions in liver enzymes through gut barrier modulation, most evidence supports multi-strain probiotics containing Lactobacillus, Bifidobacterium, and Streptococcus, at daily doses of 1–10 billion CFUs for at least 8–12 weeks. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Meta-analyses and RCTs consistently use supplementation periods of 8–12 weeks, with some pediatric studies extending to 24 weeks for persistent biomarker shifts [1,3,6]. Enteric-coated formulations and refrigerated or otherwise stabilized products ensure maximum delivery to the gut. There is little evidence that higher doses (above 20 billion CFUs daily) further improve outcomes, but sub-therapeutic doses (<1 billion CFUs) may be insufficient for liver biomarker changes [2,3,4].
Human studies do not require baseline testing for effectiveness, but individuals with known elevations in ALT or AST—especially those with metabolic syndrome or fatty liver—are most likely to benefit. Consistency of daily intake appears to be more important than the time of day. For those who track biomarkers, improvements may be detectable as early as four weeks but are most reliably seen after 8–12 weeks of continuous use. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Indirect Mechanisms: Immune Modulation and Inflammatory Pathways
Beyond gut barrier repair, Lactobacillus exerts indirect effects on liver health by modulating immune responses and reducing low-grade inflammation. These pathways may further support reductions in ALT and AST, though human evidence is less direct. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Mechanistic animal and cell studies show Lactobacillus produces SCFAs and alters cytokine profiles, increasing anti-inflammatory IL-10 and reducing pro-inflammatory markers like TNF-α and IL-6 [11,12]. Some human studies in elderly populations suggest immune enhancement and reduced systemic inflammation with Lactobacillus supplementation [7]. However, these findings are less specific to liver enzymes and more indicative of broader metabolic and immune benefits.
While these indirect mechanisms are biologically plausible and supported by preclinical data, the strongest and most consistent human evidence remains with the gut barrier-zonulin-liver enzyme pathway. Further research is needed to clarify how much immune modulation contributes to liver biomarker changes in humans. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Lactobacillus-containing probiotic supplements, especially when combined with Bifidobacterium and Streptococcus, lower liver enzymes ALT and AST by improving gut barrier function and reducing systemic zonulin. The effect is most robust in individuals with elevated liver biomarkers due to metabolic or liver-related conditions. These findings are supported by multiple meta-analyses and RCTs, with clinically meaningful reductions in ALT and AST observed after 8–12 weeks of consistent supplementation. Optimal benefits are achieved with multi-strain, enteric-coated formulations in the 1–10 billion CFU daily dose range. While immune modulation and inflammatory pathways may also contribute, the gut barrier mechanism remains the primary driver of these effects. People seeking to support liver health can consider high-viability, multi-strain probiotic supplements as a plausible strategy for improving liver enzyme profiles. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
Most human studies focus on populations with elevated ALT, AST, or metabolic dysfunction, so effects in healthy individuals or those with normal biomarkers are less clear. The majority of trials use multi-strain combinations, making it difficult to isolate the unique contribution of Lactobacillus versus other genera. Biomarker improvements do not equate to disease treatment, and long-term safety beyond several months is not fully established. There is insufficient evidence to support highly personalized probiotic selection based on individual gut microbiome profiles. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how lactobacillus relates to your health goals and monitor changes in your biomarkers over time.
