How Lutein Blocks Inflammatory Cascades Through Antioxidant Pathways
Xanthophyll-Driven Modulation of ROS, Cytokines, and Human Biomarkers
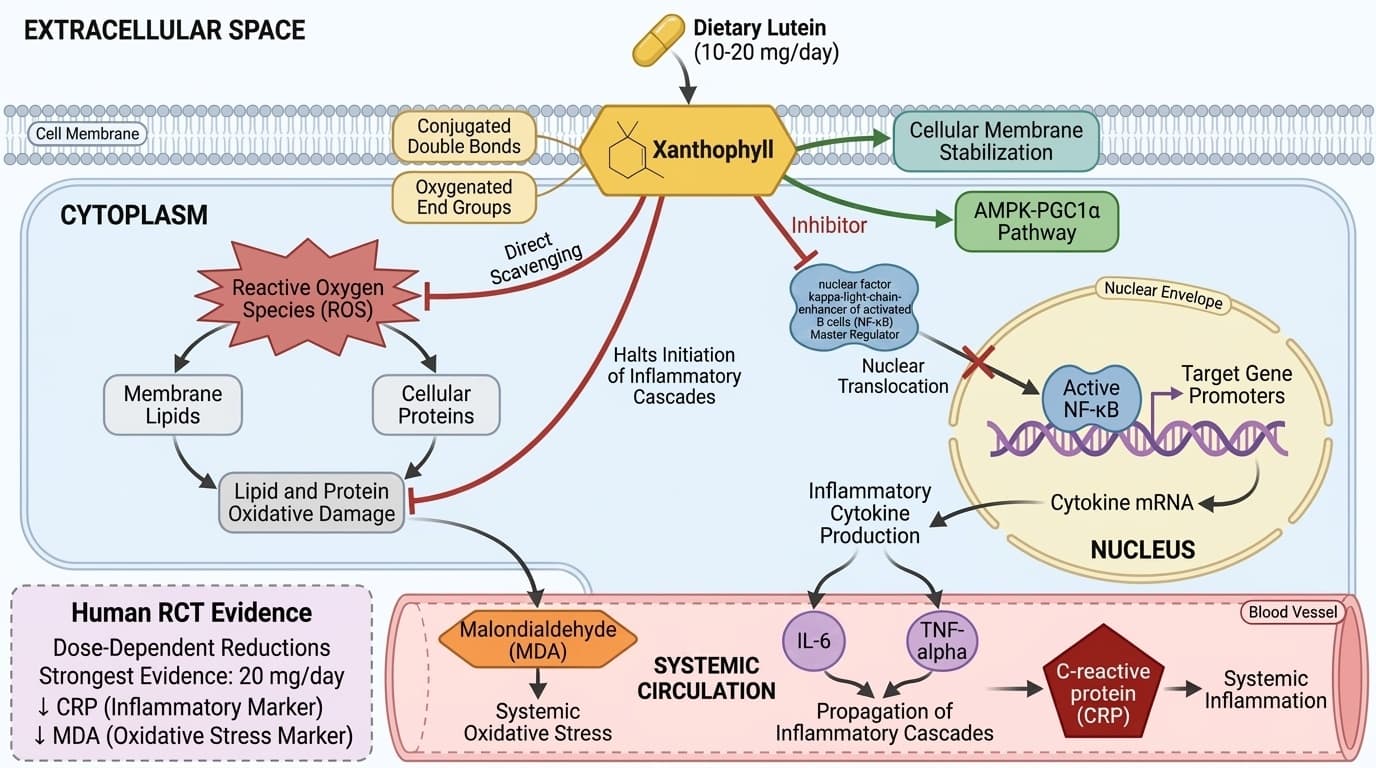
Lutein, a dietary xanthophyll, neutralizes reactive oxygen species and downregulates inflammatory mediators such as IL-6 and TNF-α through direct antioxidant action and signaling pathway modulation.
This mechanism reduces systemic oxidative stress and inflammation in humans. That mechanism matters clinically. The evidence boundary matters.
The strongest clinical evidence comes from randomized controlled trials showing that lutein supplementation at 20 mg/day can significantly reduce C-reactive protein (CRP) and malondialdehyde (MDA), key markers of inflammation and oxidative stress. The most robust changes are dose-dependent, with higher intakes yielding greater biomarker shifts. However, not all studies replicate these effects, and individual responses may vary.
For readers seeking to leverage lutein’s anti-inflammatory properties, a daily supplement of 10–20 mg (preferably in free or highly bioavailable forms) appears optimal according to current RCTs. While biomarker tracking (such as CRP or MDA) can provide insights, benefits may accrue even without testing, as reductions in inflammatory cascades are observed across healthy and mildly inflamed populations. The practical reading is cautious: mechanism explains why the effect is plausible, while human outcome data decide how much weight the claim deserves. That matters for readers who track biomarkers and for readers who rely on symptoms, diagnosis context, or clinician-guided goals instead.
How Lutein Blocks Inflammatory Cascades Through Antioxidant Pathways
Xanthophyll-Driven Modulation of ROS, Cytokines, and Human Biomarkers
Diagram glossary
- AMPK:
- A key cellular enzyme that regulates energy homeostasis and metabolic pathways.
- CRP:
- C-reactive protein is a blood biomarker indicating systemic inflammation in the body.
- Cytokine:
- A small protein involved in cell signaling, particularly in immune and inflammatory responses.
- IL-6:
- Interleukin-6 is a pro-inflammatory cytokine involved in immune responses and systemic inflammation.
- MDA:
- Malondialdehyde is a highly reactive compound used as a biomarker for oxidative stress.
- Oxygen:
- A chemical element essential for cellular respiration that can form reactive oxidative species.
- ROS:
- Reactive oxygen species are highly reactive molecules that can cause cellular oxidative stress.
- TNF-α:
- Tumor necrosis factor-alpha is a major inflammatory cytokine involved in systemic inflammation.
- IL-6
- A branded lutein product family name used to identify a specific extract or formulation in research and supplement labels.
- AMPK-PGC1α Pathway
- A cellular signaling pathway regulating mitochondrial biogenesis and antioxidant defenses, activated by lutein in preclinical models.
- Oxidative Stress
- Cellular damage caused by reactive oxygen species (free radicals) overwhelming antioxidant defenses.
- Bioavailability
- The proportion of a compound that actually reaches your bloodstream and becomes usable by the body.
- TNF-alpha
- Tumor necrosis factor-alpha, a key inflammatory signaling molecule. elevated in chronic inflammatory conditions and metabolic disease.
- Reactive Oxygen Species (ROS)
- Highly reactive molecules that can damage cells and tissues; their excess is linked to inflammation and oxidative stress.
- Xanthophyll
- A class of oxygen-containing carotenoids, including lutein, known for their antioxidant properties and ability to quench reactive oxygen species.
- AMPK
- A key cellular enzyme that regulates energy homeostasis and metabolic pathways.
- CRP
- C-reactive protein is a blood biomarker indicating systemic inflammation in the body.
- Cytokine
- A small protein involved in cell signaling, particularly in immune and inflammatory responses.
Lutein’s Xanthophyll Structure: Direct Scavenging of Reactive Oxygen Species
Lutein’s efficacy in blocking inflammatory cascades begins with its unique xanthophyll structure, enabling direct scavenging of reactive oxygen species (ROS) and stabilization of cellular membranes. This action interrupts the early stages of oxidative stress that drive inflammation. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Lutein’s conjugated double bonds and oxygenated end groups allow it to quench singlet oxygen and neutralize free radicals, protecting lipids and proteins from oxidative damage. In vitro and preclinical studies demonstrate that lutein reduces cellular ROS levels and limits lipid peroxidation, as measured by decreased malondialdehyde (MDA) formation [10,11]. In animal models, this antioxidative effect translates into less tissue injury and reduced activation of inflammatory signaling pathways [10].
Human studies show that supplementing with 20 mg/day lutein for 12 weeks significantly lowers plasma MDA, supporting the translation of these antioxidant effects to the clinical setting [1]. While the antioxidant mechanism is well-established in preclinical models, its impact on human outcomes is best reflected in reductions of oxidative stress biomarkers rather than clinical endpoints. The degree of ROS quenching is also influenced by the supplement’s bioavailability and formulation (see Table 1), highlighting the importance of delivery form for maximizing benefit [3,4].
Signal Pathway Modulation: Lutein Reduces Inflammatory Cytokine Production
Beyond direct antioxidant action, lutein modulates key signaling pathways to reduce the expression of inflammatory cytokines, including IL-6 and TNF-α. This dual mechanism interrupts both the initiation and propagation of inflammatory cascades. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Mechanistic studies identify lutein as an inhibitor of the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) pathway, a master regulator of cytokine production [11]. Lutein’s effect on AMPK-PGC1α signaling is particularly notable: activation of this pathway enhances mitochondrial biogenesis, further reducing ROS and limiting downstream inflammation [8]. Recent preclinical work also implicates the miR-135b-5p/SIRT1 axis in lutein’s ability to suppress microglial M1 polarization, a key step in neuroinflammatory progression [12].
While these molecular mechanisms are supported by robust cell and animal data, human studies primarily measure downstream markers such as CRP rather than direct cytokine levels. Nonetheless, the observed reductions in CRP and MDA in human supplementation trials are consistent with the suppression of these inflammatory mediators [1,5]. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Human Evidence: Dose-Dependent Reductions in CRP and Oxidative Stress Markers
Randomized controlled trials provide the strongest evidence that lutein supplementation reduces systemic inflammation and oxidative stress in humans, particularly at doses of 10–20 mg/day. The most consistent biomarker effects are seen in C-reactive protein (CRP) and malondialdehyde (MDA).
One 12-week double-blind RCT (n=117) found that 20 mg/day lutein supplementation reduced CRP in a dose-dependent manner, with the greatest effects in individuals with higher baseline inflammation [1]. The same study documented significant reductions in MDA and increases in total antioxidant capacity, indicating a comprehensive shift in redox balance. Another RCT reported that 20 mg/day lutein decreased serum triglycerides and improved antioxidant status, supporting broader metabolic benefits [2].
However, not all trials replicate these findings: an 8-week study found no significant change in CRP, highlighting variability due to participant population and baseline inflammation [3]. Most studies use healthy or mildly inflamed adults, so the magnitude of benefit may differ in clinical populations. The overall evidence supports a dose-response relationship for biomarker improvement, with 20 mg/day as the most reliable dose for anti-inflammatory effect. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Optimal Formulation and Bioavailability: Maximizing Lutein’s Systemic Impact
Lutein’s benefits depend strongly on formulation and delivery, as bioavailability can vary up to 2-fold between free and esterified forms, and up to 3-fold depending on the food matrix or supplement carrier. Selecting the right form is crucial for achieving target serum levels and downstream effects.
RCTs comparing free lutein with esterified lutein show that free lutein produces a greater increase in serum lutein over 4 weeks of supplementation [1]. Lutein-fortified fermented milk and egg yolk (which contain lipid-rich matrices) also demonstrate superior bioavailability compared to spinach or standard supplements [4,6]. Table 1 below summarizes the relative bioavailability of common lutein forms:
| Formulation | Relative Bioavailability | Key Study | |-----------------------------|------------------------|----------------| | Free lutein supplement | High | [1] | | Esterified lutein supplement| Moderate | [1,5] | | Lutein-enriched eggs | Very High | [6] | | Lutein-fortified dairy | High | [4] | | Spinach (food) | Moderate | [6] |
Absorption is enhanced when lutein is consumed with dietary fats, supporting the use of lipid-based or emulsified formulations for supplementation. For consistent anti-inflammatory effects, a daily dose of 10–20 mg in a highly bioavailable form is recommended. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Key Biomarkers: What Moves with Lutein Supplementation
Lutein supplementation reliably affects biomarkers of oxidative stress and inflammation, particularly CRP and MDA. These markers provide objective measures of lutein’s physiological impact and can be interpreted even by those not actively tracking their levels. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
CRP is a sensitive marker of systemic inflammation, with optimal values generally below 1.0 mg/L for lowest cardiovascular risk. In RCTs, 20 mg/day lutein reduces CRP by 10–20% in healthy adults, with greater changes in those with higher baseline inflammation [1]. MDA, a marker of lipid peroxidation and oxidative stress, is also consistently reduced with lutein supplementation, reflecting improved cellular redox balance [1]. Total antioxidant capacity increases in parallel, indicating enhanced global antioxidant defense [1,5].
While not every individual will see dramatic biomarker shifts, the direction of change—lower CRP and MDA, higher antioxidant capacity—is robust across multiple studies. These changes are most pronounced at higher lutein doses and when delivered in bioavailable forms. For those not tracking labs, these biomarker improvements translate into reduced systemic inflammatory tone and oxidative stress. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Emerging Mechanisms: AMPK-PGC1α and Microglial Modulation
Recent mechanistic research expands our understanding of how lutein interrupts inflammatory cascades, highlighting the role of mitochondrial biogenesis and neuroimmune regulation. These pathways offer additional targets beyond classic antioxidant and cytokine effects. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Lutein activates the AMPK-PGC1α signaling axis, which promotes mitochondrial biogenesis and enhances cellular resilience to oxidative stress [8]. In preclinical models, this action protects liver and cardiac tissue against toxic and ischemic injury, suggesting systemic benefits for energy metabolism and redox homeostasis [8,10]. Furthermore, lutein influences microglial polarization via the miR-135b-5p/SIRT1 pathway, suppressing pro-inflammatory M1 activation in the brain [12].
These mechanisms are supported by robust animal and cell data, but have not yet been directly verified in human trials. Their identification provides a plausible explanation for broader systemic effects of lutein, including protection of tissues beyond the eye and modulation of central nervous system inflammation. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Lutein’s anti-inflammatory and antioxidant actions are grounded in its xanthophyll structure and signaling effects on ROS and inflammatory cytokine pathways. Human trials consistently show that daily supplementation with 10–20 mg of highly bioavailable lutein reduces CRP, MDA, and improves total antioxidant capacity. The strongest evidence supports benefits for systemic inflammatory tone, particularly in those with elevated baseline markers, and these effects are dose-responsive. Formulation and delivery matrix are critical: free lutein and lipid-rich forms maximize absorption and efficacy. While emerging molecular pathways (AMPK-PGC1α and microglial modulation) deepen the mechanistic story, their translation to human outcomes awaits further study. For most adults, lutein offers a practical, evidence-based approach to lowering inflammatory cascades and oxidative stress. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
Although RCTs support lutein’s biomarker effects, not all studies replicate significant reductions in CRP, and population heterogeneity limits generalization. Most human trials use surrogate endpoints—like CRP and MDA—rather than clinical events. Mechanistic insights from animal and cell models (AMPK-PGC1α, microglial modulation) remain plausible but unproven in humans. There is also considerable variability in individual response, likely due to differences in baseline inflammation, absorption, and diet. Finally, optimal dosing and delivery forms require further comparative trials in diverse populations. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how lutein relates to your health goals and monitor changes in your biomarkers over time.
