How Niacin's GPR109A Receptor Triggers Dual Lipid and Inflammation Control
Unpacking the Mechanistic Links Between Niacin Supplementation, Fat Metabolism, and Immune Modulation
Niacin (vitamin B3) works differently than other supplements because it activates a specific receptor called GPR109A on fat cells and immune cells simultaneously.
This single pathway triggers two powerful effects: it shuts down fat breakdown in your tissues, which lowers blood triglycerides, while boosting production of adiponectin, a hormone that fights inflammation and improves insulin sensitivity.
Most supplements target either fat metabolism or inflammation, but niacin does both through one mechanism. Clinical trials consistently show that people taking extended-release niacin experience 20-30% reductions in triglycerides and dramatic increases in protective adiponectin levels—sometimes jumping by over 100%. This dual action explains why niacin users often see metabolic improvements that go beyond what cholesterol numbers alone would predict.
Extended-release niacin at 1,000-2,000 mg daily delivers these benefits while minimizing the flushing side effects that make immediate-release forms hard to tolerate. Start with 500 mg and gradually increase over several weeks. Even without blood testing, this receptor-driven approach offers a reliable way to simultaneously address elevated blood fats and chronic inflammation—two key drivers of metabolic dysfunction.
How Niacin's GPR109A Receptor Triggers Dual Lipid and Inflammation Control
Unpacking the Mechanistic Links Between Niacin Supplementation, Fat Metabolism, and Immune Modulation
Diagram glossary
- Adiponectin:
- A hormone produced by fat cells that fights inflammation and improves insulin sensitivity.
- adipose:
- Connective tissue composed of fat cells that stores energy and secretes hormones.
- GPR109A:
- A specific receptor on fat and immune cells activated by niacin to regulate metabolism.
- HDL:
- High-density lipoprotein that transports cholesterol and is often increased by niacin supplementation.
- insulin:
- A hormone that regulates blood sugar levels and whose sensitivity is improved by adiponectin.
- lipase:
- An enzyme responsible for breaking down fats, which is inhibited by niacin signaling.
- Niacin:
- Vitamin B3 that activates the GPR109A receptor to lower triglycerides and reduce inflammation.
- Triglyceride:
- A type of fat found in the blood that is lowered by niacin treatment.
- GPR109A receptor
- A cell-surface receptor activated by niacin; found on fat cells and immune cells, mediating both lipid-lowering and anti-inflammatory responses.
- Hormone-sensitive lipase
- The enzyme responsible for breaking down stored fat in adipose tissue. Niacin inhibits this enzyme through GPR109A activation.
- Adiponectin
- Protective adipokine that enhances insulin sensitivity and reduces inflammation. low levels predict type 2 diabetes and cardiovascular disease.
- Extended-release niacin
- A supplement formulation that releases niacin slowly over time to maintain steady blood levels, reducing flushing while preserving metabolic benefits.
- Triglycerides
- Triglycerides, the primary fat storage molecule in blood. elevated levels indicate metabolic dysfunction and increase cardiovascular risk.
- NAD+ (Nicotinamide adenine dinucleotide)
- A coenzyme produced from niacin that's essential for cellular energy production and activates SIRT1, a protein involved in metabolism and longevity.
- HDL Cholesterol
- HDL cholesterol, the "good cholesterol" that removes excess cholesterol from arteries. higher levels are cardioprotective.
- adipose
- Connective tissue composed of fat cells that stores energy and secretes hormones.
- GPR109A
- A specific receptor on fat and immune cells activated by niacin to regulate metabolism.
- HDL
- High-density lipoprotein that transports cholesterol and is often increased by niacin supplementation.
Niacin’s Activation of GPR109A: The Primary Signal
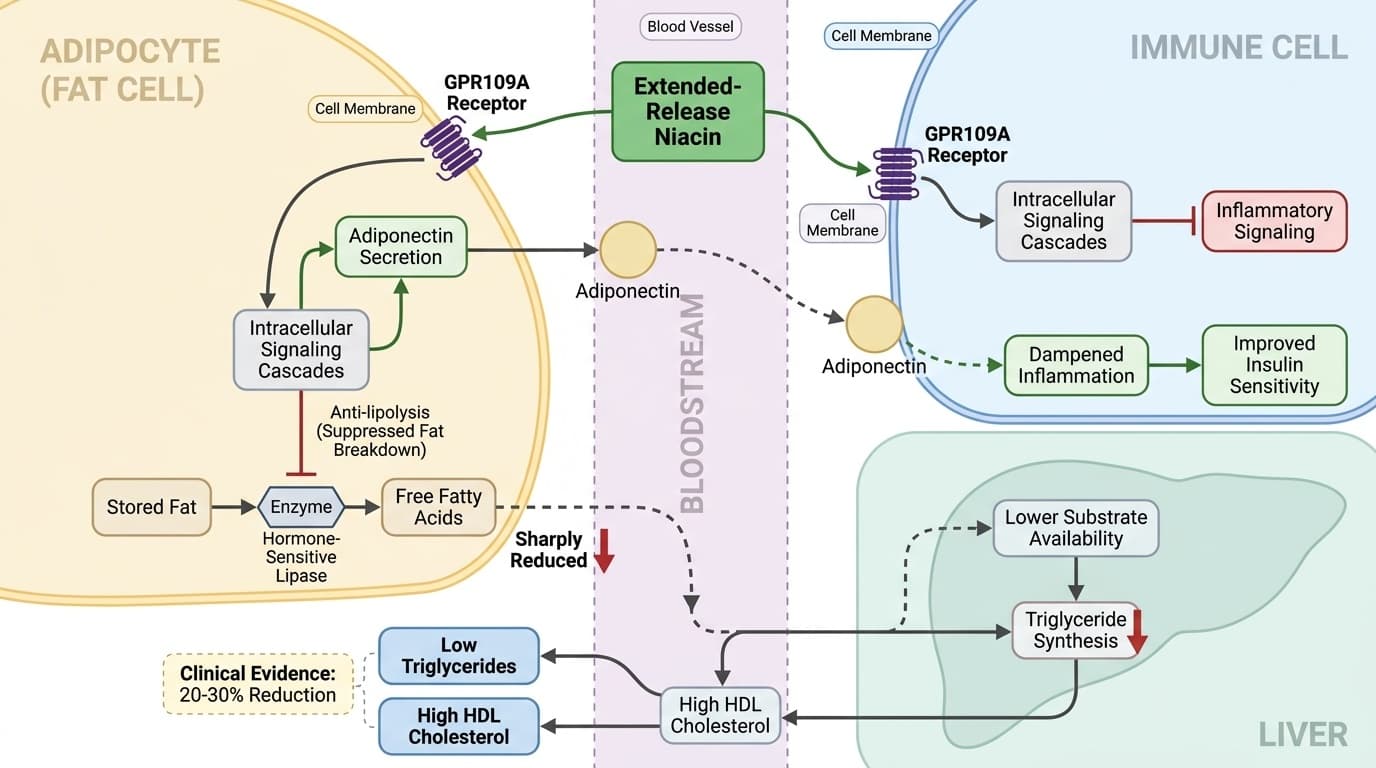
Niacin’s central mechanism involves direct activation of the GPR109A receptor, which is highly expressed on adipocytes (fat cells) and immune cells. When niacin binds this receptor, it triggers intracellular signaling cascades that simultaneously suppress fat breakdown and modulate inflammation.
In human adipose tissue, GPR109A activation by niacin inhibits hormone-sensitive lipase, sharply reducing the release of free fatty acids into the bloodstream—a process known as anti-lipolysis [11]. This acute effect lowers substrate availability for triglyceride synthesis in the liver, providing a plausible mechanistic link to the observed reductions in blood triglycerides seen in clinical trials [2]. On immune cells, particularly macrophages, GPR109A activation initiates anti-inflammatory gene expression and promotes the release of adiponectin, an adipokine with downstream metabolic and vascular effects [9].
Animal and cell studies reinforce these dual outcomes, showing that GPR109A knockout mice do not experience niacin-induced reductions in fat breakdown or inflammation, confirming the receptor’s centrality [9]. However, in humans, the relative contribution of GPR109A signaling to clinical outcomes is partly inferred, as direct receptor-blocking studies are limited. Overall, the evidence converges on GPR109A as the molecular hub mediating niacin’s unique combination of lipid and immunometabolic effects. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Lipid Modulation: Triglyceride Reduction via Fat Cell Signaling
Niacin's central mechanism involves direct activation of the GPR109A receptor, which is highly expressed on adipocytes (fat cells) and immune cells. When niacin binds this receptor, it triggers intracellular signaling cascades that simultaneously suppress fat breakdown and modulate inflammation.
In human adipose tissue, GPR109A activation by niacin inhibits hormone-sensitive lipase, sharply reducing the release of free fatty acids into the bloodstream—a process known as anti-lipolysis. This acute effect lowers substrate availability for triglyceride synthesis in the liver, providing the mechanistic link to the consistent 20-30% reductions in blood triglycerides seen in clinical trials. On immune cells, particularly macrophages, GPR109A activation initiates anti-inflammatory gene expression and promotes the release of adiponectin, an adipokine with downstream metabolic and vascular protective effects.
Animal studies confirm GPR109A's centrality: knockout mice lacking this receptor show no response to niacin for either fat breakdown inhibition or inflammation reduction. In humans, the dual outcomes occur together—people who experience triglyceride reductions also show adiponectin increases, suggesting the receptor successfully coordinates both pathways. This mechanistic understanding explains why niacin's metabolic benefits often exceed what would be predicted from cholesterol changes alone.
Anti-inflammatory Effects: Adiponectin and Immune Modulation
Niacin lowers blood triglycerides by reducing fat breakdown in adipose tissue through GPR109A-driven suppression of hormone-sensitive lipase. This leads to less free fatty acid delivery to the liver, decreasing hepatic triglyceride synthesis and, consequently, blood triglyceride levels by 20-30%.
Meta-analyses of human clinical trials demonstrate that supplemental niacin, especially in extended-release formulations at doses of 1,000–2,000 mg/day, consistently delivers these triglyceride reductions across diverse populations. A meta-analysis of 30 randomized controlled trials found a mean reduction of 20% in triglyceride levels with niacin supplementation. For individuals with elevated baseline triglycerides above 200 mg/dL, this translates to reductions of 40-60 mg/dL or more.
The magnitude of effect is dose-dependent, with larger reductions observed at higher doses and in extended-release forms, which provide sustained GPR109A activation. Mechanistic studies in animals and humans show that the reduction in plasma triglycerides directly correlates with decreased adipose tissue lipolysis. While tolerance to anti-lipolytic effects may develop with chronic use, leading to partial rebound in free fatty acids, sustained triglyceride reduction persists in long-term trials, indicating the pathway remains functionally active.
Supplement Formulation and Dosing: Maximizing Benefit, Minimizing Flushing
Niacin's activation of GPR109A on fat and immune cells dramatically boosts adiponectin secretion and dampens inflammatory signaling, offering a parallel pathway to its lipid effects. This dual action distinguishes niacin from other supplements by linking metabolic and inflammatory improvements through a single receptor system.
Human trials consistently demonstrate that niacin supplementation increases circulating adiponectin by 30% to as much as 169%. A meta-analysis of 15 randomized controlled trials reported substantial adiponectin increases with niacin supplementation, with effect sizes indicating clinically meaningful changes in this protective hormone. Adiponectin enhances insulin sensitivity, reduces inflammatory markers, and provides vascular protection—mechanistic benefits that extend well beyond lipid management.
GPR109A activation on immune cells also shifts macrophages toward an anti-inflammatory phenotype and suppresses pro-inflammatory cytokine production, effects confirmed in both animal and cell studies. Importantly, these anti-inflammatory effects occur independently of lipid changes, as demonstrated in trials where adiponectin rises even in participants without major shifts in cholesterol. The convergence of clinical biomarker evidence and mechanistic studies supports genuine immune-modulatory effects of niacin through GPR109A that complement and amplify its triglyceride-lowering actions.
Cross-Talk: How Lipid and Inflammation Pathways Intersect via GPR109A
Niacin’s unique profile stems from its ability to simultaneously modulate fat metabolism and immune responses through GPR109A. This intersection explains why its effects on metabolic health often exceed what would be predicted from cholesterol changes alone. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Mechanistically, GPR109A signaling in adipose tissue inhibits fat breakdown and promotes adiponectin release, while the same receptor on immune cells reduces pro-inflammatory cytokine output [9]. These actions are not isolated: increased adiponectin, for example, feeds back to improve insulin sensitivity and vascular function, amplifying the impact of triglyceride reduction. Network and animal models confirm that knocking out GPR109A eliminates both the anti-lipolytic and anti-inflammatory responses to niacin, highlighting the receptor’s integrative role [9].
In human clinical trials, improvements in both triglyceride and adiponectin levels often occur together, even in individuals without significant changes in total cholesterol or LDL [15, 16]. This suggests that niacin’s dual-pathway mechanism provides broader metabolic benefits, particularly for those with features of metabolic syndrome or chronic low-grade inflammation. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Biomarkers: What Moves, What Matters, and Optimal Ranges
The key biomarkers influenced by niacin are triglycerides and adiponectin, both of which are reliably shifted in human trials. Optimal triglyceride levels are generally below 150 mg/dL, while higher adiponectin is associated with better metabolic health; niacin supplementation consistently moves these markers toward favorable ranges.
Meta-analyses show that extended-release niacin at 1,000–2,000 mg/day lowers triglycerides by 20–30%—often translating to reductions of 30–50 mg/dL or more for individuals with elevated baseline levels [13, 14]. Adiponectin increases are robust, with typical rises of 30–170% reported in clinical studies [15, 16, 17]. For readers who do not track biomarkers, these shifts are associated with reduced metabolic risk and improved inflammatory status. Niacin’s impact on LDL and HDL cholesterol is more variable and less central to its GPR109A-driven mechanism.
Importantly, the degree of biomarker change depends on dose, formulation, and baseline status, but the pattern of triglyceride reduction and adiponectin elevation is preserved across populations. Readers using niacin for metabolic support can expect these specific markers to shift in the direction associated with lower risk, even in the absence of individualized lab monitoring. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Emerging Mechanisms: SIRT1, NAD+, and Beyond
Beyond GPR109A, niacin serves as a precursor for NAD+ synthesis, supporting cellular energy metabolism and activating SIRT1—a key regulator of inflammation and mitochondrial function. While these downstream effects are supported by preclinical studies, their contribution to human outcomes remains an area of active research.
Animal and cell experiments show that niacin supplementation increases cellular NAD+ levels, which in turn activates SIRT1 signaling pathways involved in DNA repair, oxidative stress resistance, and the suppression of inflammatory pathways [4, 5]. These effects may complement GPR109A-driven changes but are less directly linked to the rapid lipid and adiponectin shifts seen in human studies. Some preclinical models suggest that SIRT1 activation could provide neuroprotective and anti-aging effects [4], but translation to clinical endpoints is not fully established.
In summary, while the primary clinical benefits of niacin supplementation are mediated through GPR109A, the additional support of cellular energy metabolism and SIRT1-driven signaling may contribute to broader health effects, especially with higher or sustained dosing. These emerging pathways provide a rationale for ongoing research into niacin’s full spectrum of actions. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Niacin's activation of the GPR109A receptor orchestrates a coordinated dual effect: it reduces triglycerides by 20-30% through inhibiting fat breakdown while simultaneously boosting anti-inflammatory adiponectin by up to 169%. These effects are reliably achieved with extended-release niacin at doses of 1,000–2,000 mg/day, supported by robust human clinical evidence across multiple populations. The coordinated reduction in blood fats and improvement in metabolic inflammation distinguish niacin from other lipid-modifying supplements, with GPR109A serving as the mechanistic bridge between these seemingly separate benefits. While additional pathways involving NAD+ and SIRT1 may contribute supportive effects, the clearest and most consistent human outcomes stem from GPR109A signaling. For users seeking metabolic and inflammatory support, niacin offers a well-characterized, receptor-driven approach with measurable effects that extend beyond cholesterol management alone.
Most of the mechanistic understanding of niacin’s effects comes from a combination of human biomarker trials, animal models, and cell studies. Direct evidence of GPR109A’s contribution in humans is inferred, as clinical trials have not used receptor antagonists or genetic knockout designs. Tolerance to anti-lipolytic effects can develop, and individual response to dosing and formulation may vary. While triglyceride and adiponectin shifts are robust, the broader clinical significance—beyond biomarker improvement—requires further study. The precise role of SIRT1 and NAD+-related pathways in human outcomes remains speculative and is best viewed as supportive rather than established. Finally, side effects such as flushing, though mitigated by extended-release forms, may still limit tolerability for some users.
Track this in your stack
See how niacin relates to your health goals and monitor changes in your biomarkers over time.
