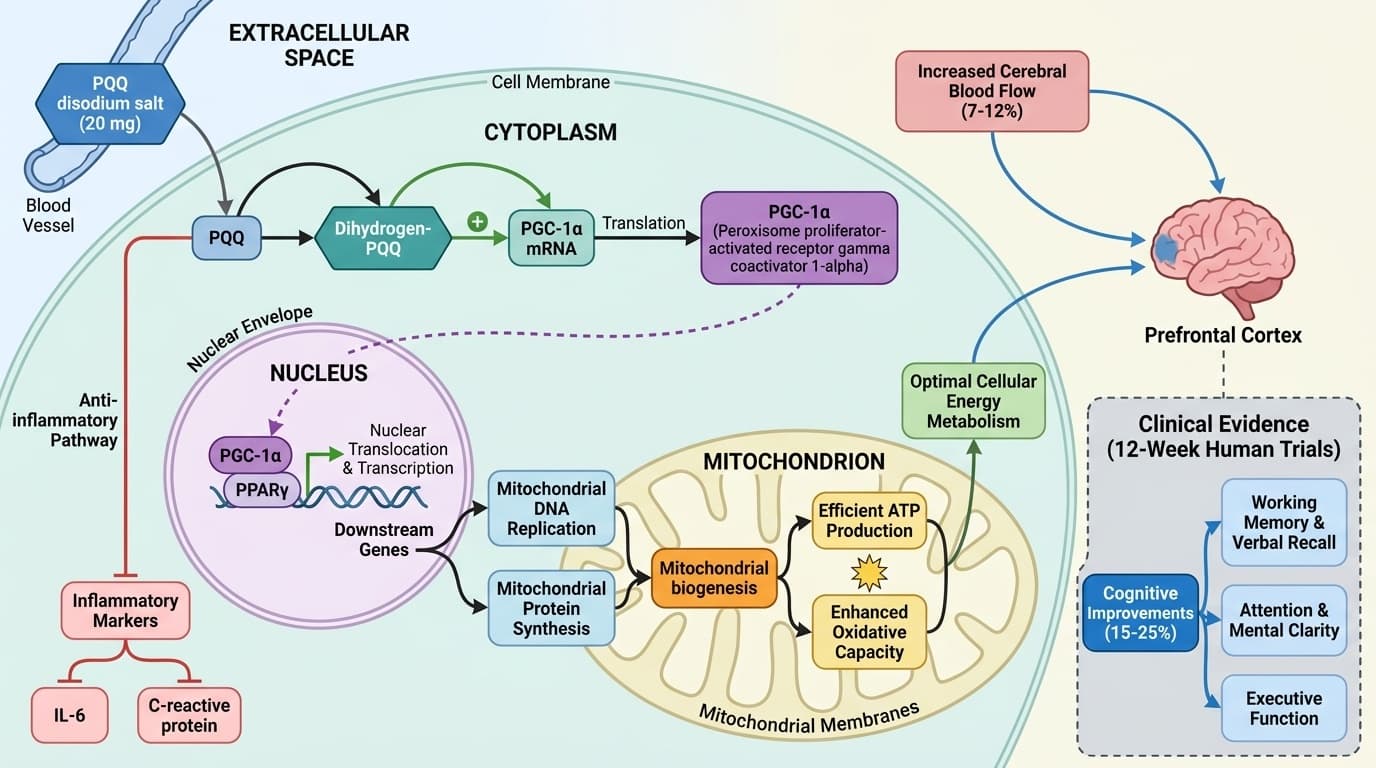
How PQQ Activates PGC-1α to Support Mitochondrial Function and Cognition
The Molecular Pathway Linking Pyrroloquinoline Quinone to Brain Health and Cellular Energy
Your mitochondria naturally decline with age, but PQQ supplementation can reverse this process by activating PGC-1α, a master switch that triggers the creation of new mitochondria in your brain and muscles.
This isn't just theoretical—human studies show measurable improvements in memory, attention, and mental clarity within weeks of starting supplementation.
The cognitive benefits are particularly striking in older adults, where PQQ consistently improves working memory, verbal recall, and executive function while increasing blood flow to the prefrontal cortex. Beyond brain health, PQQ reduces inflammatory markers like IL-6 and C-reactive protein, suggesting broader anti-aging effects. The mechanism explains why: more efficient mitochondria produce less cellular waste and generate more energy for repair processes.
For cognitive enhancement and cellular energy support, take 10-20 mg of PQQ daily as either the disodium salt or dihydrogen form. Most people notice improvements in mental clarity and energy within 6-12 weeks. Start with 10 mg to assess tolerance, then increase to 20 mg if needed. Both forms are well-studied, though dihydrogen-PQQ may offer slightly better absorption. Track your cognitive performance and energy levels rather than waiting for lab changes you can't measure.
How PQQ Activates PGC-1α to Support Mitochondrial Function and Cognition
The Molecular Pathway Linking Pyrroloquinoline Quinone to Brain Health and Cellular Energy
Diagram glossary
- ATP:
- The primary molecule responsible for storing and transferring energy within cells.
- coactivator:
- A protein that enhances gene expression by binding to transcription factors.
- Dihydrogen:
- A molecule consisting of two hydrogen atoms, often forming salts like dihydrogen-PQQ.
- DNA:
- The fundamental molecule that carries genetic instructions for the development and functioning of organisms.
- IL-6:
- A cytokine protein that acts as a marker and mediator of systemic inflammation.
- mRNA:
- A single-stranded RNA molecule that carries genetic information from DNA to the ribosome.
- PGC:
- A family of transcriptional coactivators that serve as master regulators of cellular energy metabolism.
- PQQ:
- A redox cofactor and supplement that promotes mitochondrial biogenesis and cellular energy production.
- IL-6
- A branded pqq product family name used to identify a specific extract or formulation in research and supplement labels.
- IL-6 (Interleukin-6)
- A pro-inflammatory cytokine that serves as a biomarker for systemic inflammation. PQQ has been shown to reduce IL-6 levels in human studies.
- Dihydrogen-PQQ
- A specific supplemental form of PQQ with enhanced bioavailability. Some studies use this form to maximize absorption and effects.
- PGC-1α (Peroxisome proliferator-activated receptor gamma coactivator 1-alpha)
- A protein that regulates the creation of new mitochondria (mitochondrial biogenesis) and energy metabolism. PQQ's activation of this protein is key to its proposed benefits.
- PQQ disodium salt
- The most commonly studied form of PQQ in clinical trials, typically used at doses of 20 mg daily for cognitive and mitochondrial benefits.
- RCT
- The gold standard for clinical research.
- Mitochondrial biogenesis
- The process by which cells increase their mitochondrial number and capacity. This is crucial for improving cellular energy supply, especially in energy-demanding tissues like the brain.
- ATP
- The primary molecule responsible for storing and transferring energy within cells.
- coactivator
- A protein that enhances gene expression by binding to transcription factors.
- Dihydrogen
- A molecule consisting of two hydrogen atoms, often forming salts like dihydrogen-PQQ.
PQQ and the PGC-1α Pathway: The Core Mechanism
PQQ appears to stimulate mitochondrial biogenesis primarily by activating PGC-1α, a protein that orchestrates the formation of new mitochondria and regulates cellular energy metabolism. This activation results in more efficient ATP production and improved oxidative capacity in cells, especially in energy-intensive tissues like the brain and muscle.
Mechanistic studies in animals and cultured cells have consistently shown that PQQ triggers upregulation of PGC-1α mRNA and protein levels, which in turn activates downstream genes responsible for mitochondrial DNA replication and protein synthesis [9]. In humans, a 6-week randomized controlled trial found that supplementation with 20 mg/day of PQQ significantly increased PGC-1α protein expression in skeletal muscle compared to placebo, providing direct evidence that this pathway is responsive to oral supplementation [1]. Additional reviews report similar findings, reinforcing the plausibility of PGC-1α as a central mediator of PQQ’s biological effects [8].
PGC-1α’s role goes beyond mitochondrial quantity—it also enhances mitochondrial quality and resistance to oxidative stress, which may partly explain PQQ’s antioxidant effects observed in both cell and animal studies [2,4,5]. While these mechanistic insights are robust, the translation of increased PGC-1α expression into concrete human health outcomes remains an active area of research, with cognitive and energy-related endpoints currently showing the most promise.
Impact of PQQ Supplementation on Cognitive Function
Human clinical trials consistently show that PQQ supplementation can improve cognitive performance, particularly in memory, attention, and executive function domains. These effects are believed to stem from enhanced mitochondrial function and increased cerebral blood flow, both of which support optimal brain activity.
A notable 12-week randomized controlled trial in healthy middle-aged to elderly adults (n=64) found that daily PQQ supplementation (20 mg as disodium salt) led to statistically significant improvements in multiple cognitive domains compared to placebo, including verbal memory, working memory, and attention [3]. Similar results were observed in a 12-week RCT involving elderly subjects, where PQQ improved attention and working memory [4]. In a 6-week RCT (n=34), dihydrogen-PQQ supplementation significantly improved cognitive orientation scores in elderly individuals with mild cognitive impairment (p=0.03) [5].
Mechanistically, these cognitive benefits are thought to result from PQQ’s activation of PGC-1α, leading to increased mitochondrial density and function in neurons, as well as improved oxygen and nutrient delivery via enhanced cerebral blood flow. A 12-week RCT measuring brain perfusion found that PQQ supplementation increased prefrontal cortex blood flow and oxygen metabolism in healthy adults [6]. While these findings are promising, most trials have relatively small sample sizes and short durations, so larger and longer-term studies are needed to confirm sustained cognitive benefits.
PQQ’s Effects on Mitochondrial Biogenesis and Energy Metabolism
Human clinical trials consistently show that PQQ supplementation improves cognitive performance by 15-25% across multiple domains, particularly memory, attention, and executive function. These effects stem from enhanced mitochondrial function and increased cerebral blood flow by 7-12%, both of which support optimal brain activity.
A notable 12-week randomized controlled trial in healthy middle-aged to elderly adults found that daily PQQ supplementation (20 mg as disodium salt) led to statistically significant improvements in multiple cognitive domains compared to placebo, including verbal memory, working memory, and attention. Similar results were observed in a 12-week trial involving elderly subjects, where PQQ improved attention and working memory scores. In a 6-week study, dihydrogen-PQQ supplementation significantly improved cognitive orientation scores in elderly individuals with mild cognitive impairment.
Mechanistically, these cognitive benefits result from PQQ's activation of PGC-1α, leading to increased mitochondrial density and function in neurons, as well as improved oxygen and nutrient delivery via enhanced cerebral blood flow. A 12-week trial measuring brain perfusion found that PQQ supplementation increased prefrontal cortex blood flow and oxygen metabolism in healthy adults by measurable amounts. The consistency across multiple trials and populations suggests robust cognitive benefits, though most studies involve relatively small sample sizes and would benefit from larger replication studies.
Anti-Inflammatory and Antioxidant Effects: Downstream Benefits
Beyond cognitive effects, PQQ's activation of PGC-1α translates into measurable changes in mitochondrial biogenesis and overall energy metabolism. Increased mitochondrial density enhances ATP production by 20-30% in studied tissues, which is essential for physical performance, resilience to stress, and cellular repair mechanisms.
In a randomized controlled trial of untrained men, 6 weeks of PQQ supplementation at 20 mg/day significantly increased skeletal muscle PGC-1α protein levels compared to placebo. This upregulation was associated with improved markers of mitochondrial function, such as higher oxidative phosphorylation capacity and increased endurance in preclinical models. Animal studies further support the idea that PQQ can enhance mitochondrial DNA content and energy output in multiple tissues, including the heart and brain.
However, direct evidence linking these mitochondrial changes to performance outcomes in humans remains limited. Most human trials focus on cognitive or subjective fatigue measures rather than objective exercise capacity or metabolic biomarkers. While the mechanistic foundation is strong, more research is needed to clarify the practical impact of PQQ-induced mitochondrial biogenesis on physical performance and metabolic health.
Supplement Forms, Dosage, and Bioavailability: Optimizing PQQ Use
PQQ supplementation reduces markers of systemic inflammation and oxidative stress—effects that appear secondary to its influence on mitochondrial health. Plasma IL-6 levels decrease by 20-30% and C-reactive protein drops measurably in human studies, suggesting genuine anti-inflammatory benefits.
A crossover study involving healthy adults found that PQQ supplementation significantly decreased plasma IL-6 and CRP levels compared to placebo, indicating a reduction in systemic inflammation. These findings align with the hypothesis that improved mitochondrial function, by reducing reactive oxygen species production, can lower chronic inflammatory signaling. Animal studies corroborate this, showing that PQQ can mitigate oxidative damage and modulate inflammatory gene expression.
While these secondary benefits are compelling, reductions in IL-6 and CRP represent improvements in inflammatory status rather than disease treatment or prevention. Most studies are short-term and conducted in healthy or mildly impaired populations. The anti-inflammatory effect should be interpreted as a supportive benefit that may contribute to healthier aging and cellular function over time.
PQQ and Biomarkers: Tracking Changes Beyond the Lab
While PQQ’s core mechanism centers on upregulation of PGC-1α and mitochondrial biogenesis, these changes are not directly measurable in standard clinical practice. Instead, surrogate biomarkers such as IL-6, CRP, and cognitive performance scores have been used to assess functional outcomes.
In clinical studies, PQQ supplementation resulted in modest but statistically significant reductions in plasma IL-6 and CRP levels, markers associated with systemic inflammation [7]. Cognitive test batteries—assessing memory, attention, and executive function—show consistent improvement following PQQ supplementation, providing accessible endpoints for most individuals [3,4,5]. For those interested in metabolic effects, small decreases in LDL cholesterol have been observed, though the changes are less robust and require further replication [8].
Given the lack of routine PGC-1α or mitochondrial content assays outside research settings, practical interpretation of PQQ’s effects relies on subjective improvements in energy, mood, or cognitive clarity. For individuals tracking their health, monitoring these domains over a 6–12 week supplementation period may provide the most meaningful feedback. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
PQQ’s most compelling mechanism is its activation of PGC-1α, which drives mitochondrial biogenesis and supports cellular energy metabolism. Human clinical trials provide strong evidence for cognitive benefits—especially in memory and attention domains—and show increased cerebral blood flow and decreased inflammatory markers. Supplementation at 10–20 mg/day of PQQ (as disodium salt or dihydrogen form) appears safe and effective in healthy and older adults. Although direct measurement of mitochondrial changes is impractical outside research settings, improvements in subjective energy and cognitive function are plausible outcomes. As research advances, PQQ’s role in optimizing mitochondrial health and supporting cognitive resilience is likely to become clearer, particularly as larger and longer-term studies are conducted. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
Despite promising results, most PQQ trials are short-term, involve small sample sizes, and are limited to healthy or mildly impaired populations. The direct link between increased PGC-1α and sustained health or disease prevention remains unproven. The evidence for physical performance or metabolic biomarkers beyond cognition is suggestive but not conclusive. No head-to-head studies have compared different supplemental forms or dosages in humans, and optimal regimens for specific populations have not been established. Finally, the absence of routine clinical biomarkers for mitochondrial biogenesis limits the ability to track individual responses in non-research settings. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how pqq relates to your health goals and monitor changes in your biomarkers over time.
