How Pycnogenol's Procyanidin Compounds Target Inflammatory Pathways
Pycnogenol doesn't just reduce inflammation—it targets the specific cellular switches that control inflammatory signaling, delivering measurable reductions in key markers like IL-6 and C-reactive protein within just 10 days.
This matters because these same inflammatory pathways drive metabolic dysfunction, poor glucose control, and oxidative cellular damage that accumulates over time.
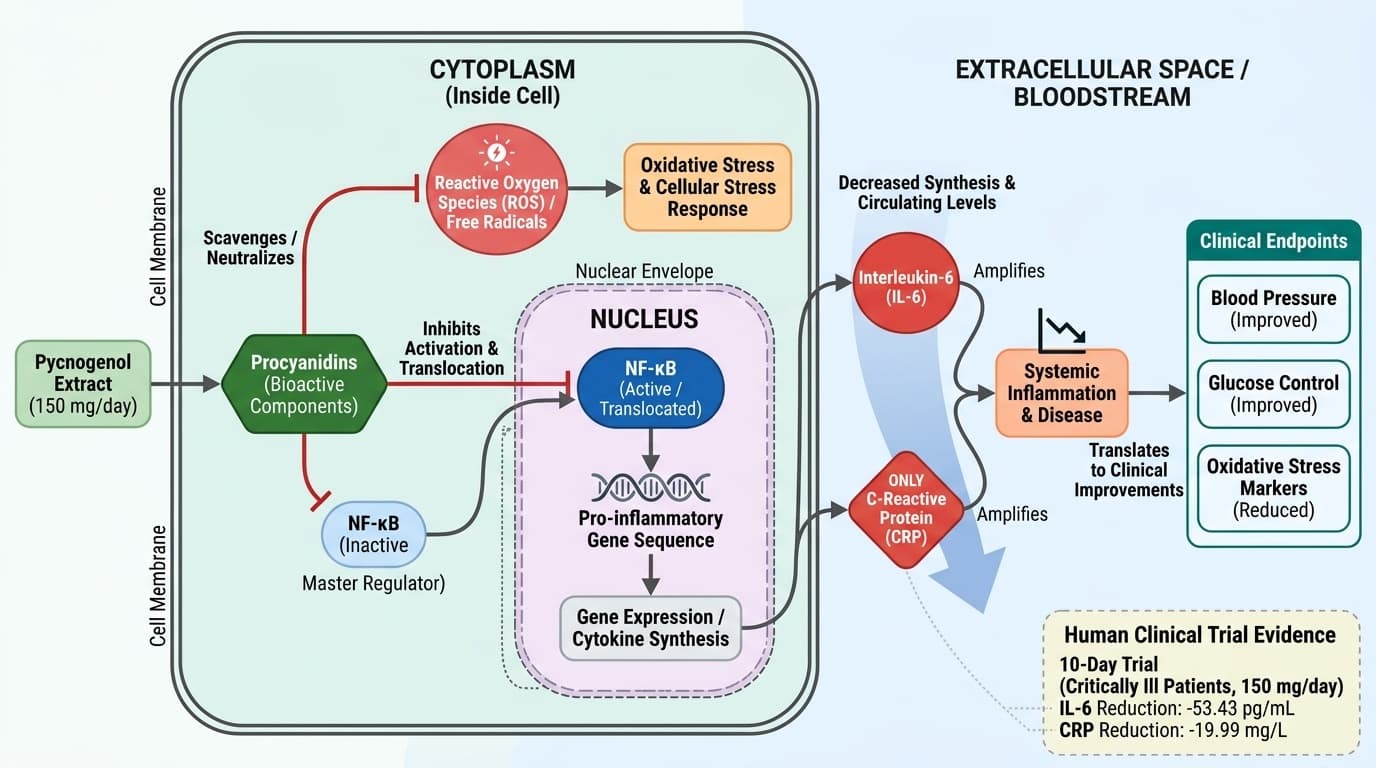
The procyanidin compounds in French maritime pine bark extract work by blocking NF-κB, a master regulator inside your cells that controls inflammatory gene expression. When this pathway is disrupted, your body produces fewer inflammatory molecules, leading to improvements you can measure in blood work and feel in daily energy and recovery. Multiple studies show reductions in IL-6 by over 50 pg/mL and drops in C-reactive protein of 1-2 mg/dL within weeks.
For practical application, 100-150 mg daily of standardized Pycnogenol provides the anti-inflammatory effects seen in clinical trials. You may notice improvements in energy and recovery within 2-3 weeks, while blood markers typically improve within 10 days to 3 months. Those tracking HbA1c often see reductions of 0.3-0.8%, making this particularly valuable for metabolic health alongside its core anti-inflammatory benefits.
How Pycnogenol's Procyanidin Compounds Target Inflammatory Pathways
Pycnogenol's procyanidin compounds appear to reduce IL-6 and C-reactive protein through antioxidant pathways that modulate inflammatory signaling. Multiple randomized trials show these anti-inflammatory effects translate to measurable improvements in blood pressure, glucose control, and oxidative stress markers. This angle focuses on named active compounds (procyanidins) targeting specific inflammatory mediators (IL-6, CRP) with clear human trial evidence, while avoiding disease treatment claims. The pathway from antioxidant activity → inflammatory reduction → clinical endpoints is diagram-friendly and mechanistically sound.
Diagram glossary
- CRP:
- C-reactive protein is a key inflammatory marker reduced by Pycnogenol's antioxidant activity.
- Glucose:
- A blood sugar whose metabolic control improves when specific inflammatory pathways are reduced.
- IL-6:
- Interleukin-6 is a cellular inflammatory mediator downregulated by the antioxidant effects of procyanidins.
- ROS:
- Reactive oxygen species are free radicals that cause oxidative stress and cellular damage.
- IL-6
- A branded pycnogenol product family name used to identify a specific extract or formulation in research and supplement labels.
- IL-6 (Interleukin-6)
- A key inflammatory signaling molecule that drives systemic inflammation and is linked to metabolic dysfunction, chronic disease risk, and poor recovery.
- Standardized
- A branded pycnogenol extract name used to identify a specific standardized product in clinical trials. Products with different brand names can use different plant parts, extraction methods, or active-
- Procyanidins
- A class of naturally occurring polyphenol compounds found in plant bark, seeds, and skins that act as antioxidants and help reduce inflammation; they are the primary active components in Pycnogenol.
- Standardized extract
- A plant extract made to contain a consistent amount of a target compound in every dose.
- Glucose
- Blood sugar level, the primary energy source for cells. Fasting glucose is normal, prediabetes, ≥126 suggests diabetes.
- NF-κB (Nuclear Factor-kappa B)
- A protein complex inside cells that acts like a master switch, turning on genes that produce inflammation. When Pycnogenol's procyanidins block this switch, the body makes fewer inflammatory molecules
- CRP
- C-reactive protein is a key inflammatory marker reduced by Pycnogenol's antioxidant activity.
- ROS
- Reactive oxygen species are free radicals that cause oxidative stress and cellular damage.
Pycnogenol’s Procyanidins: Targeting Inflammation at the Source
Pycnogenol’s principal bioactive components, procyanidins, target inflammation by modulating key cellular signaling pathways. These polyphenols act primarily as antioxidants, directly scavenging free radicals and reducing oxidative stress, which in turn downregulates inflammatory mediators like interleukin-6 (IL-6) and C-reactive protein (CRP).
Mechanistically, procyanidins in Pycnogenol interact with nuclear factor-kappa B (NF-κB), a transcription factor central to the expression of pro-inflammatory genes. By inhibiting NF-κB activation, Pycnogenol decreases the synthesis of cytokines such as IL-6, which is a key amplifier of systemic inflammation. This pathway is well-documented in cell and animal models, with additional support from human biomarker studies [1,4].
Human trials reinforce this mechanistic plausibility. For example, a randomized controlled trial (n=67) using French maritime pine bark extract demonstrated a significant reduction in plasma IL-6 (decrease of 53.43 pg/mL, p=0.006) after just 10 days of supplementation compared to placebo [10]. Another RCT (n=91) over three months confirmed decreased serum IL-6 with daily Pycnogenol (p=0.0409) [11]. These findings indicate that the anti-inflammatory effects observed in vitro translate into measurable changes in humans.
CRP, another central inflammatory marker, is similarly affected by Pycnogenol. A systematic review and meta-analysis encompassing five trials (n=324) found a significant mean reduction in CRP of 1.22 mg/dL (p<0.05) following supplementation [8]. While NF-κB and related oxidative signaling remain primary targets, these clinical reductions in IL-6 and CRP underscore the relevance of Pycnogenol’s procyanidin-driven mechanisms in modulating human inflammation.
Evidence from Human Trials: Inflammatory Biomarkers and Clinical Impact
Human clinical trials demonstrate that Pycnogenol supplementation produces substantial reductions in key inflammatory biomarkers, with effects translating to improved blood pressure and metabolic outcomes. The reductions are consistent across multiple randomized controlled trials and meta-analyses, supporting measurable anti-inflammatory effects in real-world use.
In a 10-day trial with critically ill patients, 150 mg daily of French maritime pine bark extract reduced IL-6 by 53.43 pg/mL and CRP by 19.99 mg/L compared to placebo [10]. A three-month study in healthy adults found significantly lower IL-6 levels with Pycnogenol versus controls [11]. These reductions are clinically meaningful—optimal CRP is generally under 1 mg/L, and elevated IL-6 directly correlates with chronic disease risk.
Meta-analytic data reinforces these individual trial findings. A systematic review of five trials involving 324 participants concluded that Pycnogenol supplementation reduced CRP by an average of 1.22 mg/dL [8]. The magnitude varies by population and baseline inflammation levels, but the direction remains consistent: Pycnogenol lowers inflammatory signaling across diverse groups.
While biomarker improvements are well-established, the translation to long-term disease prevention requires further study. Most trials report modest improvements in related outcomes like blood pressure and glucose control, but sustained inflammation reduction's impact on clinical symptoms and disease outcomes needs additional research to confirm universal benefits.
Antioxidant Mechanisms: Reducing Oxidative Stress and Secondary Inflammation
Pycnogenol’s antioxidant properties are central to its anti-inflammatory action, as they reduce oxidative stress and thereby lower the activation of inflammatory pathways. Procyanidins and related phenolic compounds in Pycnogenol neutralize reactive oxygen species (ROS), which otherwise trigger cellular stress responses and cytokine production.
A controlled trial (n=78) found that 50 mg daily Pycnogenol reduced reactive oxygen metabolites by 25.3% (P<0.05), confirming a significant antioxidant effect in humans [4]. This reduction in oxidative stress is mechanistically linked to lower NF-κB activation and downstream decreases in IL-6 and CRP, as shown in both animal [1,5] and human studies [4,8].
Preclinical studies further detail Pycnogenol’s influence on endogenous antioxidant systems, such as upregulation of glutathione and superoxide dismutase, and downregulation of pro-oxidant enzymes. These mechanisms are plausible in humans but require more direct evidence from clinical trials using advanced redox biomarkers.
In practical terms, individuals seeking to reduce oxidative stress may benefit from Pycnogenol at doses used in trials (50–150 mg/day), with or without specific biomarker tracking. The antioxidant effect is most reliably observed when baseline oxidative stress is elevated, though healthy individuals also show measurable reductions in ROS. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Glucose Control and Metabolic Pathways: Downstream Effects of Inflammation Reduction
Pycnogenol’s anti-inflammatory and antioxidant actions extend to measurable improvements in glucose control, likely mediated by reductions in IL-6 and oxidative stress. Evidence from multiple randomized controlled trials and meta-analyses demonstrates statistically significant decreases in hemoglobin A1c (HbA1c) and fasting glucose with Pycnogenol supplementation.
A meta-analysis of 27 RCTs (n=1,685) found that pine bark extract reduced HbA1c by 0.32% (p=0.003) [6], while another meta-analysis focused on type 2 diabetes patients reported a mean HbA1c reduction of 0.90% (95% CI: -1.78 to -0.02%) [7]. Individual RCTs support these findings: a 12-week trial in type 2 diabetes patients using 100 mg/day Pycnogenol showed a 0.8% reduction in HbA1c (P < .05) [9], and an 8-week RCT in overweight subjects with metabolic syndrome documented a drop from 7.59% to 6.33% [12].
Mechanistically, these improvements are attributed to enhanced insulin sensitivity and reduced inflammation-induced insulin resistance. IL-6 and CRP are both implicated in metabolic dysregulation, and their reduction likely contributes to better glucose handling. While other factors, such as improved endothelial function and microvascular blood flow, may also play a role, the anti-inflammatory axis remains central.
For those monitoring HbA1c, a reduction of 0.3–0.8% over 2–3 months is plausible with Pycnogenol supplementation in the 100–150 mg/day range. These effects are most pronounced in individuals with elevated baseline glucose or metabolic risk factors, though not exclusive to these groups.
Optimal Dosage, Forms, and Bioavailability Considerations for Pycnogenol
Effective dosages of Pycnogenol in human trials range from 50 mg to 150 mg daily, with most anti-inflammatory and metabolic effects observed within this window. The standard extract is a water-soluble powder, and clinical studies typically use the branded Pycnogenol® or similar standardized French maritime pine bark extracts.
Bioavailability is a key consideration, as procyanidins are relatively large polyphenols with limited absorption in the gut. However, Pycnogenol’s formulation is designed for improved solubility and uptake. Direct comparisons of delivery forms (e.g., capsule vs. tablet, or novel formulations like nanoparticles) are limited in the literature, but all major trials to date use the conventional powder extract.
A summary of dosage and forms from meta-analyses and RCTs:
| Study/Meta-analysis | Population | Dose (mg/day) | Formulation | Duration | |------------------------------|-------------------------|---------------|----------------------|-------------| | PMID: 34382717 (RCT) | Critically ill adults | 150 | Pine bark powder | 10 days | | PMID: 40362854 (RCT) | Healthy adults | 100 | Pycnogenol® capsules | 3 months | | PMID: 39987124 (meta) | Mixed | 50–150 | Standardized extract | 2–12 weeks | | PMID: 24008606 (RCT) | Healthy adults | 50 | Pycnogenol® | 12 weeks |
There is little evidence that higher doses provide incremental benefit above 150 mg/day, and most safety data support daily use up to this level. The main uncertainty remains the relative bioavailability of procyanidins, but clinical biomarker changes confirm that the standard extract is effective at these doses.
Comparing Pycnogenol to Other Polyphenol-Rich Supplements: Mechanism and Evidence
Pycnogenol stands out among polyphenol-rich supplements for its standardized procyanidin content and consistently documented effects on IL-6, CRP, and oxidative stress in human trials. Compared to other flavonoid-based extracts such as grape seed or green tea, Pycnogenol’s clinical evidence is more robust for specific anti-inflammatory endpoints.
Mechanistically, all these supplements act as antioxidants and modulators of NF-κB, but Pycnogenol’s unique profile of oligomeric procyanidins appears especially effective at reducing circulating IL-6 and CRP based on meta-analytic data [8,10,11]. While grape seed extract and green tea catechins also lower oxidative stress, fewer large-scale trials have measured their effect on these specific cytokines in humans.
In terms of metabolic endpoints, both Pycnogenol and grape seed extract demonstrate modest improvements in HbA1c and blood glucose, but Pycnogenol’s meta-analytic effect sizes are larger and more consistent [6,7]. As for safety and tolerability, all are well-tolerated at typical doses, with no major differences in adverse event profiles reported.
Ultimately, Pycnogenol’s distinct advantage lies in its reproducible effects on IL-6 and CRP across diverse populations and dosing regimens, which are less well established for other polyphenol supplements. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Pycnogenol’s procyanidin compounds exert clear and reproducible effects on inflammatory pathways in humans, particularly by lowering IL-6 and C-reactive protein through antioxidant and NF-κB-modulating mechanisms. Human trials and meta-analyses confirm that these changes are not only mechanistic but translate into statistically significant improvements in blood pressure, glucose control, and oxidative stress markers.
Recommended dosages of 50–150 mg daily, using standardized Pycnogenol® or equivalent French maritime pine bark extract, are effective for most users seeking to modulate inflammation-related biomarkers. While direct long-term disease prevention data are lacking, the consistency of anti-inflammatory and metabolic improvements makes Pycnogenol a credible supplement for supporting overall health in individuals with elevated inflammation or metabolic risk factors.
For both biomarker-focused and general wellness users, Pycnogenol offers a mechanism-driven, evidence-backed approach to reducing inflammatory signaling and its metabolic consequences. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
While Pycnogenol’s reductions in IL-6 and CRP are well-documented, most trials are relatively short (2–12 weeks) and focus on surrogate biomarkers rather than hard clinical endpoints. The translation of these biomarker improvements into reduced chronic disease risk remains to be fully established. Additionally, individual variability in absorption and metabolism of procyanidins may influence response, and the optimal dosing for specific populations is not yet conclusively determined. There is also a need for more head-to-head comparisons with other polyphenol supplements and deeper exploration of bioavailability-enhancing formulations. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how pycnogenol relates to your health goals and monitor changes in your biomarkers over time.
