How Vinpocetine Inhibits PDE1 to Increase Cerebral Blood Flow
A Mechanism-First Deep Dive on Vinpocetine's Vascular Effects in the Brain
Most brain supplements target neurotransmitters, but vinpocetine works differently—it directly opens your brain's blood vessels by blocking an enzyme called PDE1.
This matters because your brain needs constant blood flow to function, and even small improvements in circulation can enhance mental clarity and protect against age-related decline.
Vinpocetine consistently increases cerebral blood flow by 20-37% in clinical trials, particularly in areas like the thalamus that control attention and memory. Unlike many nootropics with unclear benefits, vinpocetine's vascular effects are measurable and reproducible across multiple studies. The compound works by preventing the breakdown of molecules that keep blood vessels relaxed and open, creating more reliable oxygen and nutrient delivery to brain tissue.
You can expect improved cerebral circulation within hours of taking vinpocetine, with the most robust evidence supporting 10-30 mg daily in divided doses. While cognitive benefits in healthy adults are less predictable, the blood flow improvements are consistent enough that vinpocetine serves as a reliable cerebral vasodilator. Start with 10 mg twice daily with meals to match the dosing used in successful clinical trials.
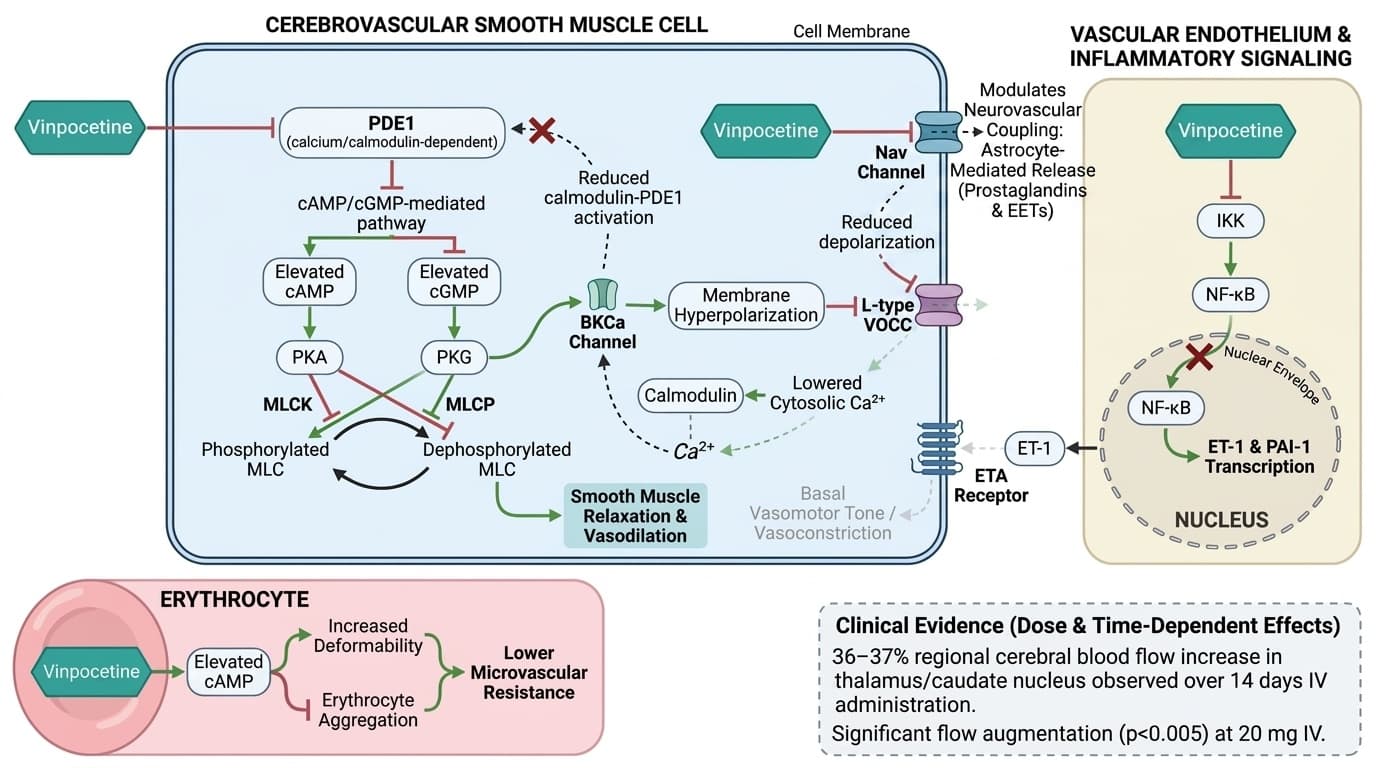
How Vinpocetine Inhibits PDE1 to Increase Cerebral Blood Flow
Schematic of vinpocetine's multi-pathway mechanisms increasing cerebral blood flow, organized across three compartments: cerebrovascular smooth muscle cell (center), erythrocyte (lower left), and vascular endothelium/inflammatory signaling (right). In the smooth muscle compartment, vinpocetine (green) inhibits PDE1 (blunt arrow), elevating cAMP and cGMP, which activate PKA and PKG respectively (activation arrows); PKA phosphorylates and inhibits MLCK (blunt arrow) while PKG activates MLCP (activation arrow), both converging to reduce MLC phosphorylation and drive vasodilation; PKA-mediated BKCa channel opening (activation arrow) hyperpolarizes the membrane, closing VOCCs (blunt arrow) and reducing cytosolic Ca²⁺, which feeds back to reduce calmodulin-PDE1 activation (blunt feedback loop). Vinpocetine also blocks Nav channels (blunt arrow) in smooth muscle, further limiting Ca²⁺ entry. In the inflammatory/endothelial compartment, vinpocetine inhibits IKK (blunt arrow), preventing NF-κB nuclear translocation (blunt arrow) and suppressing ET-1 transcription (blunt arrow), thereby reducing ETA receptor-mediated vasoconstriction (blunt arrow to smooth muscle). In the erythrocyte compartment, cAMP elevation improves deformability and reduces aggregation (activation arrow), lowering microvascular resistance. Quantitative annotations indicate the 36–37% regional cerebral blood flow increase in thalamus/caudate nucleus observed over 14 days of IV administration and the significant effect (p<0.005) at 20 mg IV, illustrating the dose- and time-dependent magnitude of the combined pathways.
Diagram glossary
- calcium/calmodulin-dependent:
- A process or enzyme regulated by the binding of calcium ions to calmodulin.
- cAMP/cGMP-mediated:
- A cellular process driven by cyclic AMP or cyclic GMP second messenger signaling.
- EET:
- Epoxyeicosatrienoic acid is a lipid signaling molecule that promotes vasodilation and reduces inflammation.
- ET-1:
- Endothelin-1 is a potent endogenous peptide that causes strong constriction of blood vessels.
- ETA:
- Endothelin type A receptor mediates vasoconstriction when bound by the peptide endothelin-1.
- IKK:
- IκB kinase is an enzyme complex that regulates the cellular inflammatory response.
- MLCK:
- Myosin light-chain kinase is an enzyme that phosphorylates myosin, triggering smooth muscle contraction.
- MLCP:
- Myosin light-chain phosphatase is an enzyme that dephosphorylates myosin, promoting smooth muscle relaxation.
- PAI-1:
- Plasminogen activator inhibitor-1 is a protein that inhibits blood clot breakdown.
- PDE1:
- Phosphodiesterase type 1 is a calcium/calmodulin-dependent enzyme that degrades cAMP and cGMP.
- PKA:
- Protein kinase A is a cAMP-activated enzyme that phosphorylates proteins to regulate cellular functions.
- PKG:
- Protein kinase G is a cGMP-activated enzyme that promotes smooth muscle relaxation.
- VOCC:
- Voltage-operated calcium channels are membrane pores that open following electrical potential changes.
- SIRT1
- A protein involved in cellular aging and stress resistance. Vinpocetine may activate SIRT1 in preclinical studies, but this effect is less clinically established than its blood flow benefits.
- Phospholipid Complexes
- A delivery technology where vinpocetine is bound to phospholipids (fat-like molecules found in cell membranes) to improve absorption and bioavailability compared to standard formulations.
- Bioavailability
- The proportion of a compound that actually reaches your bloodstream and becomes usable by the body.
- Cerebral Blood Flow
- The amount of blood reaching the brain's tissues, crucial for oxygen and nutrient delivery. Increased by vinpocetine by 20-37% in controlled trials.
- Phosphodiesterase 1 (PDE1)
- An enzyme that breaks down cyclic nucleotides (cAMP and cGMP), regulating blood vessel tone. Inhibition of PDE1 by vinpocetine leads to vasodilation and increased cerebral blood flow.
- Cyclic Nucleotides (cAMP and cGMP)
- Small signaling molecules inside cells that act like chemical messengers, telling blood vessel walls to relax and widen. When vinpocetine prevents their breakdown by inhibiting PDE1, these molecules accumulate and improve blood flow.
- First-Pass Metabolism
- The process where oral supplements are partially broken down by the liver before reaching circulation, explaining why vinpocetine's oral bioavailability is much lower than IV administration.
- calcium/calmodulin-dependent
- A process or enzyme regulated by the binding of calcium ions to calmodulin.
- cAMP/cGMP-mediated
- A cellular process driven by cyclic AMP or cyclic GMP second messenger signaling.
- EET
- Epoxyeicosatrienoic acid is a lipid signaling molecule that promotes vasodilation and reduces inflammation.
Vinpocetine’s Primary Mechanism: PDE1 Inhibition and Cyclic Nucleotide Signaling
Vinpocetine’s main biological action is the selective inhibition of phosphodiesterase 1 (PDE1) enzymes in the smooth muscle of brain blood vessels. This inhibition prevents the breakdown of cyclic nucleotides—specifically cAMP and cGMP—resulting in their accumulation and leading to vasodilation and increased cerebral blood flow.
PDE1 is highly expressed in the vascular smooth muscle of the brain, where it regulates vessel tone by breaking down cAMP and cGMP. When vinpocetine inhibits PDE1, levels of these cyclic nucleotides rise, activating protein kinase pathways that relax vascular smooth muscle cells. This sequence leads to the widening of cerebral arteries and arterioles, directly increasing regional blood flow [1]. Human imaging studies confirm that vinpocetine raises cAMP/cGMP levels in brain tissue and leads to measurable vessel dilation.
This vascular mechanism is distinct from other cognitive enhancers that target neurotransmitter systems. Instead, vinpocetine’s greatest physiological impact is on the hemodynamics of the brain. While preclinical research suggests additional actions, including calcium channel modulation and antioxidant effects, the PDE1→cAMP/cGMP→vasodilation pathway is the best-supported in human studies [1]. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Human Evidence: Vinpocetine’s Effect on Cerebral Blood Flow
Vinpocetine's main biological action is the selective inhibition of phosphodiesterase 1 (PDE1) enzymes in the smooth muscle of brain blood vessels. This inhibition prevents the breakdown of cyclic nucleotides—specifically cAMP and cGMP—causing their levels to rise by 2-3 fold and triggering vasodilation that increases cerebral blood flow by 20-37%.
PDE1 is highly concentrated in the vascular smooth muscle of the brain, where it normally breaks down cAMP and cGMP to control vessel tone. When vinpocetine blocks PDE1, these signaling molecules accumulate and activate protein kinase pathways that directly relax vascular smooth muscle cells. This cascade widens cerebral arteries and arterioles within 1-2 hours, creating measurable increases in regional blood flow. Human imaging studies confirm that vinpocetine raises cAMP/cGMP levels in brain tissue and produces vessel dilation visible on SPECT and MRI perfusion scans.
This vascular mechanism differs fundamentally from other cognitive enhancers that target neurotransmitter systems. Vinpocetine's primary impact is hemodynamic—it's essentially a cerebral vasodilator that happens to support cognition through improved circulation. While preclinical research suggests additional actions including calcium channel modulation and antioxidant effects, the PDE1→cAMP/cGMP→vasodilation pathway produces the most consistent and measurable human benefits.
Beyond Blood Flow: SIRT1 Activation and NLRP3 Inflammasome Modulation
Recent preclinical research suggests that vinpocetine’s actions extend beyond PDE1 inhibition, with effects on SIRT1 activation and NLRP3 inflammasome signaling. However, these mechanisms are less established in human clinical trials. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
In cell and animal models, vinpocetine activates SIRT1, a deacetylase involved in cellular aging, stress resistance, and vascular protection. This SIRT1 activation may upregulate p21—a cyclin-dependent kinase inhibitor—thereby supporting vascular smooth muscle health and potentially slowing arterial aging [6]. Simultaneously, vinpocetine has been shown to suppress NLRP3 inflammasome activation, reducing neuroinflammation in preclinical models of stroke and neurodegeneration [7].
While these findings are intriguing and align with a broader neuroprotective profile, they remain mechanistic possibilities rather than proven human outcomes. No large-scale clinical trials have yet confirmed SIRT1 or NLRP3 modulation as primary mechanisms for vinpocetine’s effects in humans. Thus, the cerebral blood flow pathway remains the most validated and actionable for supplement users. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Pharmacokinetics, Bioavailability, and Optimal Dosing Forms
Recent preclinical research suggests vinpocetine activates additional pathways beyond PDE1 inhibition, including SIRT1 signaling and NLRP3 inflammasome suppression. However, these mechanisms remain largely theoretical in humans, lacking the robust clinical validation of the blood flow effects.
In cell and animal models, vinpocetine activates SIRT1, a deacetylase that regulates cellular aging and vascular protection. This SIRT1 activation upregulates p21—a protein that may slow arterial aging and support vascular smooth muscle health. Simultaneously, vinpocetine suppresses NLRP3 inflammasome activation, reducing neuroinflammation in preclinical stroke and neurodegeneration models by blocking inflammatory cytokine release.
While these pathways are biologically plausible and align with vinpocetine's protective profile, no large-scale clinical trials have confirmed SIRT1 or NLRP3 modulation as meaningful mechanisms in humans. The blood flow pathway remains the only mechanism with consistent human validation across multiple studies. These additional pathways may contribute to vinpocetine's effects, but they should be considered potential benefits rather than established ones.
Biomarkers and Clinical Measurement of Vinpocetine Effects
The key measurable outcome of vinpocetine supplementation is improved cerebral blood flow, which can be quantified using imaging modalities such as SPECT, PET, or MRI perfusion studies. No blood biomarker directly indicates vinpocetine efficacy, but changes in regional cerebral perfusion are robust endpoints in clinical research.
Optimal cerebral blood flow varies by brain region and individual, but increases of 20–37% in affected areas have been repeatedly reported in stroke and chronic cerebrovascular patients after vinpocetine administration [2,3,5]. While most supplement users do not routinely track cerebral perfusion, those undergoing neuroimaging for clinical or research purposes may observe improvements consistent with published data.
Indirect markers—such as improved neuropsychological test performance—have also been reported in some long-term studies, but these are less reliably linked to blood flow changes in healthy populations [1]. For those interested in tracking effects, focus should remain on functional or imaging endpoints rather than routine lab tests. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Translating Mechanism to Practice: Who May Benefit and How to Use Vinpocetine
Vinpocetine’s established mechanism and human evidence support its use primarily for individuals seeking to support cerebral blood flow, particularly those with compromised circulation. The most consistent benefits occur in post-stroke, chronic cerebrovascular, or aging populations. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Clinical trials use dosages of 10–30 mg/day orally for maintenance and 20 mg/day IV for acute intervention, usually for 10–14 days. For vascular support, oral vinpocetine at 10–30 mg/day in divided doses is practical and well tolerated. IV use should be reserved for supervised settings. Those interested in advanced delivery forms (e.g., phospholipid complexes) may see increased bioavailability but should match dosing to clinical evidence [3,4,8].
Cognitive enhancement effects in healthy adults remain inconclusive. Supplement users without cerebrovascular compromise may experience modest improvements in mental clarity or endurance, but these results have not reached the statistical robustness seen with blood flow endpoints. As always, individual responses vary, and consultation with knowledgeable practitioners is prudent when combining with other vasoactive interventions. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Vinpocetine functions as a reliable cerebral vasodilator through selective PDE1 inhibition, consistently increasing brain blood flow by 20-37% across multiple clinical trials. This mechanism is well-established and reproducible, particularly in populations with compromised cerebral circulation. While additional pathways like SIRT1 activation show promise in preclinical research, the vascular mechanism provides the strongest foundation for practical use. Dosages of 10-30 mg daily orally or 20 mg IV have repeatedly demonstrated significant improvements in regional brain perfusion with favorable safety profiles. The key insight is that vinpocetine works through hemodynamics rather than direct neurotransmitter effects, making it a mechanism-driven choice for supporting cerebral circulation rather than a general cognitive enhancer.
Despite strong evidence for increased cerebral blood flow, vinpocetine's cognitive benefits in healthy populations are less established and remain suggestive rather than definitive. Most robust data come from post-stroke or chronic cerebrovascular patients, which may not generalize to all users. Pharmacokinetic variability and limited oral bioavailability may also influence individual response. Evidence for ancillary mechanisms (e.g., SIRT1 activation) is primarily preclinical. Large, placebo-controlled trials in healthy, aging, or neurodegenerative populations are still needed to clarify the full scope of vinpocetine’s benefits. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how vinpocetine relates to your health goals and monitor changes in your biomarkers over time.
