How Vitamin B6 Converts to PLP to Power 150+ Enzyme Reactions
The Central Role of Pyridoxal 5-Phosphate in Human Metabolism and Health
Vitamin B6 supplements convert to pyridoxal 5-phosphate (PLP), the active coenzyme form that enables over 150 enzymatic reactions controlling amino acid metabolism, neurotransmitter synthesis, and homocysteine processing.
This core mechanism underlies all vitamin B6 effects in the body. That mechanism matters clinically.
The strongest human evidence shows that vitamin B6 supplementation reliably raises plasma PLP levels, which can influence oxidative stress markers, cholesterol, and blood glucose—especially in individuals with suboptimal status or elevated metabolic demands. However, downstream effects vary based on which PLP-dependent pathways are limited, and clinical outcomes do not always follow from higher PLP alone.
Practically, this means vitamin B6’s benefits are highly context-dependent: raising PLP is necessary but not always sufficient for health impact. For most, supplementing 10–50 mg/day of pyridoxine HCl or P-5-P is enough to normalize PLP. The biggest gains occur where a specific PLP-dependent enzyme or pathway is bottlenecked, such as in certain metabolic, neurotransmitter, or homocysteine-processing tasks. Biomarker tracking can help, but is not required for most to benefit from strategic B6 support. The practical reading is cautious: mechanism explains why the effect is plausible, while human outcome data decide how much weight the claim deserves. That matters for readers who track biomarkers and for readers who rely on symptoms, diagnosis context, or clinician-guided goals instead.
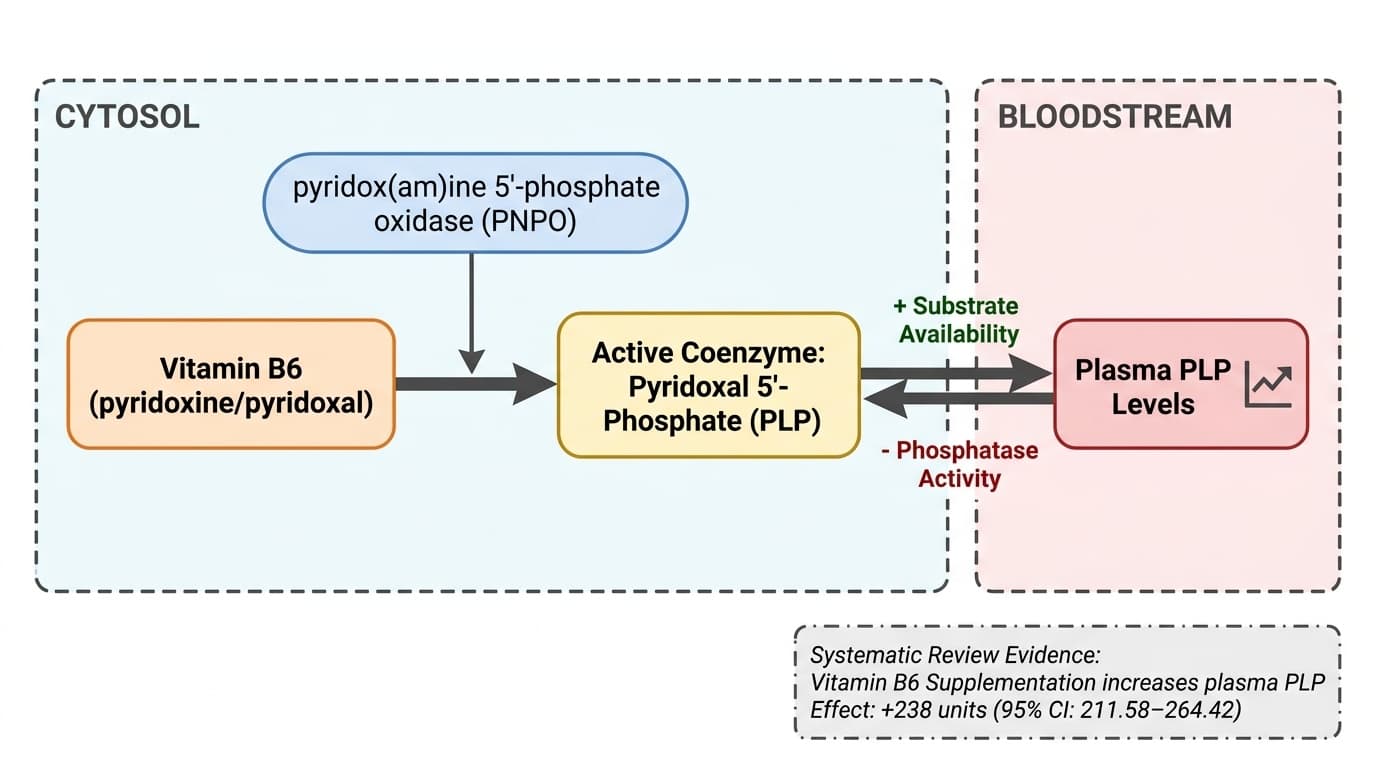
How Vitamin B6 Converts to PLP to Power 150+ Enzyme Reactions
A simple arrow diagram should depict Vitamin B6 (pyridoxine/pyridoxal) activating pyridox(am)ine 5'-phosphate oxidase (PNPO), which drives the conversion to the active coenzyme Pyridoxal 5'-Phosphate (PLP), with a bidirectional arrow indicating that plasma PLP levels rise or fall depending on substrate availability and phosphatase activity.
Diagram glossary
- B6:
- A vitamin ingested as precursors and converted to the active coenzyme PLP.
- phosphatase:
- An enzyme that removes phosphate groups, which can reduce active PLP levels.
- PLP:
- The biologically active coenzyme form of vitamin B6, also known as pyridoxal 5'-phosphate.
- PNPO:
- An enzyme catalyzing the rate-limiting oxidation of vitamin B6 precursors into active PLP.
- pyridoxamine:
- An ingested form of vitamin B6 that is oxidized to form active PLP.
- pyridoxine/pyridoxal:
- Primary ingested forms of vitamin B6 that are converted into the active coenzyme PLP.
- substrate:
- A molecule upon which an enzyme acts, driving metabolic pathways when available.
- Pyridoxine HCl
- A common, stable supplemental form of vitamin B6, converted in the liver to PLP. Used in most B6 supplements due to its bioavailability and cost-effectiveness.
- Pyridoxal 5-phosphate (PLP)
- The active coenzyme form of vitamin B6, essential for over 150 enzymatic reactions in the body, including amino acid metabolism and neurotransmitter synthesis. PLP is the main circulating and tissue-active B6 metabolite.
- Oxidative Stress
- Cellular damage caused by reactive oxygen species (free radicals) overwhelming antioxidant defenses.
- Neurotransmitter
- Chemical messengers that transmit signals between neurons.
- Homocysteine
- Homocysteine, an amino acid metabolite influenced by B vitamins. elevated levels damage blood vessels and increase cardiovascular and dementia risk.
- Neurotransmitter Synthesis
- The creation of brain-signaling chemicals such as serotonin, dopamine, and GABA. Several steps require PLP as a coenzyme.
- Glucose
- Blood sugar level, the primary energy source for cells. Fasting glucose is normal, prediabetes, ≥126 suggests diabetes.
- B6
- A vitamin ingested as precursors and converted to the active coenzyme PLP.
- phosphatase
- An enzyme that removes phosphate groups, which can reduce active PLP levels.
- PLP
- The biologically active coenzyme form of vitamin B6, also known as pyridoxal 5'-phosphate.
Vitamin B6 Metabolism: Conversion to Pyridoxal 5-Phosphate (PLP)
Vitamin B6 from supplements or food must be converted in the liver to pyridoxal 5-phosphate (PLP) to become biologically active. PLP acts as a universal coenzyme, enabling over 150 different enzymatic reactions crucial to human metabolism. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
The three principal natural forms of vitamin B6—pyridoxine, pyridoxal, and pyridoxamine—are all absorbed and then phosphorylated in the liver, but only PLP serves as the cofactor for essential enzymes. Human studies show that both pyridoxine HCl (the most common supplement form) and direct PLP (as P-5-P) supplementation effectively raise plasma PLP concentrations, though the process can be influenced by liver health, genetic factors, and competing nutrient status [9]. Optimal plasma PLP levels are generally considered to be 20–125 nmol/L, with deficiency defined below 20 nmol/L and possible toxicity above 200 nmol/L [9].
Conversion efficiency from pyridoxine to PLP may be impaired in some metabolic conditions or during chronic inflammation, but this is rare in healthy adults. PLP is distributed to all tissues, with particularly high requirements in the liver, brain, and muscle. Notably, recent research suggests that gut microbiota can synthesize pyridoxamine, which also serves as a precursor to PLP and may influence tissue-specific PLP pools [5].
In summary, supplementing with 10–50 mg/day of pyridoxine HCl or equivalent P-5-P reliably increases circulating PLP, supporting downstream metabolic functions. However, the tissue-specific demand for PLP and the relative activity of B6-dependent enzymes determine the magnitude of physiological effects.
PLP-Dependent Enzyme Reactions: Amino Acid and Neurotransmitter Metabolism
PLP acts as a required coenzyme for more than 150 enzymatic reactions, with the greatest impact on amino acid metabolism and neurotransmitter synthesis. This explains why vitamin B6 status can influence mood, cognition, and metabolic health. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Among its major roles, PLP is essential for transamination (the transfer of amino groups between amino acids), decarboxylation (generating neurotransmitters such as serotonin, dopamine, and GABA), and one-carbon metabolism (including folate and homocysteine cycles). Human clinical research confirms that B6 supplementation increases PLP and can improve biomarkers of neurotransmitter status, especially in populations at risk of deficiency or under high metabolic demand [9]. For example, B6 deficiency is associated with lower serotonin and GABA synthesis, which may manifest as mood or sleep disturbances. Restoring PLP through supplementation can normalize these pathways in most cases.
The table below summarizes key PLP-dependent pathways:
| Pathway | Key Enzyme (PLP-Dependent) | Physiological Role | |------------------------|------------------------------|---------------------------| | Transamination | Aminotransferases | Amino acid metabolism | | Decarboxylation | Aromatic L-amino acid decarboxylase | Neurotransmitter synthesis | | Transsulfuration | Cystathionine beta-synthase | Homocysteine detoxification | | Glycogenolysis | Glycogen phosphorylase | Glucose release from glycogen |
Most clinical outcomes depend on which of these pathways are PLP-limited in an individual. While normalizing PLP status supports these reactions, most people with adequate intake see only subtle changes unless specific enzyme bottlenecks exist.
Vitamin B6 and Homocysteine: Transsulfuration Pathways and Cardiometabolic Implications
Vitamin B6, through PLP, is necessary for the transsulfuration pathway that converts homocysteine to cysteine, thereby regulating homocysteine levels and related metabolic risk. This pathway relies on PLP-dependent enzymes such as cystathionine beta-synthase and cystathionine gamma-lyase. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Human studies indicate that vitamin B6 supplementation can lower homocysteine, but this effect is most pronounced when combined with other B vitamins (e.g., folate and B12) or in individuals with suboptimal status [9]. For example, in a randomized trial, vitamin B6 supplementation (with B12 and folate) led to significant reductions in plasma homocysteine, especially in those with baseline elevations [9]. However, B6 alone has a modest effect unless there is an underlying deficiency.
Elevated homocysteine is a recognized biomarker of cardiovascular risk, and PLP-dependent transsulfuration is the chief route for homocysteine detoxification. Therefore, ensuring sufficient B6 status (typically >30 nmol/L PLP) is a reasonable strategy for individuals with elevated homocysteine, particularly if intake of other B vitamins is also optimized. However, the direct impact on cardiovascular events remains less clear, as lowering homocysteine does not always translate to reduced disease risk in all populations.
In summary, vitamin B6’s mechanistic effect on homocysteine is clear and supported by both mechanistic and human data, but the translation to clinical endpoints depends on baseline status and other cofactors.
Vitamin B6, Oxidative Stress, and Inflammation: Evidence from Human Trials
Vitamin B6, via its active PLP form, has been studied for its potential to reduce oxidative stress and inflammation in humans. The main biomarker used in clinical studies is malondialdehyde (MDA), a marker of lipid peroxidation. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Several randomized controlled trials (RCTs) have examined the effect of vitamin B6 supplementation on MDA and related oxidative stress markers. For example, a 7-day RCT in sepsis patients found that intravenous vitamin B6 significantly reduced MDA levels compared to control (PMID: 38691102) [10]. In healthy adults, 6 weeks of B-complex supplementation (including B6) decreased oxidative stress markers (PMID: 31915511) [12]. However, results are not always consistent: in pregnant women with obesity, 18 mg/day of B6 for 24–36 weeks did not impact MDA levels (PMID: 38396126), and in stroke patients, high-dose B6 combination therapy was paradoxically associated with increased MDA (PMID: 37793337).
Mechanistic studies suggest that PLP modulates redox-sensitive signaling pathways and may directly scavenge reactive oxygen species [2,6]. Still, the magnitude and consistency of antioxidant effects in humans depends on context, baseline status, and co-supplementation. Practical takeaway: vitamin B6 can reduce oxidative stress markers in settings of deficiency or elevated demand, but effects are variable across populations and dosing regimens.
Tissue-Specific PLP Requirements and the Role of the Microbiome
Recent research reveals that PLP requirements differ substantially across tissues, and that gut microbiota play a role in vitamin B6 metabolism. These factors can influence who responds best to B6 supplementation. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
PLP concentrations are highest in the liver, skeletal muscle, and brain. Animal studies and emerging human evidence suggest that B6 deficiency most strongly affects neurotransmitter synthesis in the brain and insulin signaling in the pancreas—critical for mood and metabolic health [5]. For example, PLP-dependent serotonin synthesis in pancreatic islets is particularly sensitive to B6 status. Further, gut microbes can synthesize pyridoxamine, a natural B6 vitamer that is converted to PLP and may preferentially support certain tissue pools [5]. This highlights the potential for personalized or targeted B6 interventions depending on gut microbiome composition and tissue-specific demands.
In practical terms, most individuals meet systemic PLP needs through standard supplementation, but those with digestive disorders, high oxidative stress, or unique metabolic demands may require higher doses or forms that bypass liver conversion (e.g., direct P-5-P). The interplay between microbiome-derived B6 forms and host metabolism remains an active area of research. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Optimal Dosing, Forms, and Safety of Vitamin B6 Supplementation
Standard vitamin B6 dosing for metabolic support ranges from 10–50 mg/day, with both pyridoxine HCl and P-5-P (pyridoxal 5-phosphate) demonstrating efficacy in raising circulating PLP. Higher doses (100+ mg/day) are sometimes used in clinical research, but prolonged use above 200 mg/day can cause neuropathy and is not recommended for general supplementation [7].
Human trials indicate that pyridoxine HCl is efficiently converted to PLP in most adults, but P-5-P may be preferable for those with impaired liver function or certain genetic polymorphisms. Direct P-5-P supplementation can also be more predictable in raising plasma PLP, but both forms are effective for most people [9].
Safety is generally high at physiologic doses, but chronic excessive intake can lead to sensory neuropathy, emphasizing the importance of adhering to evidence-based ranges. The table below compares forms and uses:
| Form | Typical Dose | Conversion Efficiency | Clinical Use | |-----------------------|-------------|----------------------|-----------------------------| | Pyridoxine HCl | 10–50 mg | High | General population | | Pyridoxal 5-phosphate | 10–25 mg | Directly active | Impaired liver function, special cases | | Pyridoxamine | Rarely used | Microbiome-derived | Experimental, pending more evidence |
For most, a daily dose of 10–50 mg is sufficient to maintain optimal PLP and support the many enzymatic processes dependent on vitamin B6. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Vitamin B6’s central mechanism is its conversion to pyridoxal 5-phosphate (PLP), which powers over 150 enzymatic reactions essential to human health. Supplementation reliably increases plasma PLP, supporting amino acid metabolism, neurotransmitter synthesis, and homocysteine detoxification. However, the degree of benefit depends on baseline status, tissue-specific demands, and the presence of bottlenecks in PLP-dependent pathways.
Optimal B6 intake—typically 10–50 mg/day of pyridoxine HCl or P-5-P—supports most physiological needs and is well tolerated. Individuals with specific metabolic challenges, high oxidative stress, or impaired conversion may require tailored forms or doses. The strongest evidence supports B6’s role in normalizing PLP, improving homocysteine processing, and reducing oxidative stress in certain populations.
Ultimately, vitamin B6 is a foundational cofactor whose benefits are most apparent where deficiency, high demand, or specific enzyme limitations exist. Most people can optimize PLP and downstream metabolic health with standard, evidence-based B6 supplementation. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured.
While the core mechanism of vitamin B6 as a coenzyme (PLP) is well established, the translation from increased PLP to clinical outcomes is variable and context-dependent. Many studies are short-term, use combination supplements, or focus on populations with underlying health conditions. The role of microbiome-derived B6 forms and tissue-specific PLP needs remains incompletely understood. Finally, while B6 reliably raises PLP and influences metabolic markers, evidence for direct effects on disease endpoints (cardiovascular events, cognitive decline) is less robust and requires further long-term human trials. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how vitamin b6 relates to your health goals and monitor changes in your biomarkers over time.
