How Vitamin E's Alpha-Tocopherol Reduces Inflammation via Membrane Protection
Your cell membranes are under constant attack from free radicals, but vitamin E doesn't just float around your bloodstream hoping to help—it actually embeds itself directly into your cell walls where the damage happens.
This precise positioning allows alpha-tocopherol to stop the chain reaction of lipid destruction that triggers widespread inflammation throughout your body.
When your cell membranes get damaged by oxidation, they release inflammatory signals that raise markers like C-reactive protein (CRP) and tumor necrosis factor-alpha (TNF-α). Multiple studies show vitamin E supplementation consistently lowers CRP levels and reduces TNF-α, with the effect being strongest in people who face higher oxidative stress—like smokers or those with metabolic syndrome. The response follows a clear pattern: CRP drops at moderate doses, while TNF-α requires higher amounts to budge.
For measurable anti-inflammatory benefits, take 400-800 mg daily of natural d-alpha-tocopherol (not the synthetic dl-form). Expect to see CRP improvements within 8-12 weeks, especially if your baseline inflammation is elevated. Take it with a fat-containing meal to maximize absorption, and consider testing your CRP levels before and after supplementation to track your personal response.
How Vitamin E's Alpha-Tocopherol Reduces Inflammation via Membrane Protection
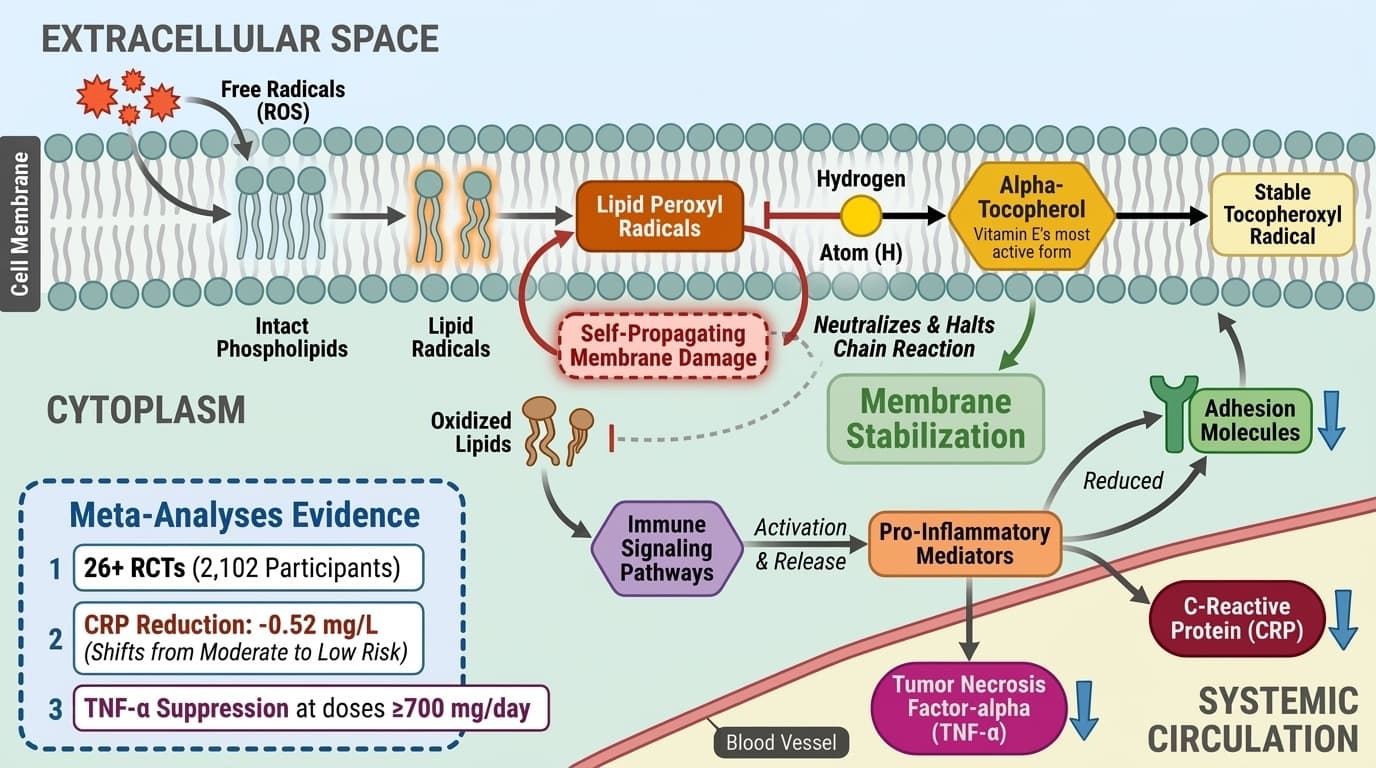
Alpha-tocopherol, vitamin E's most active form, embeds in cell membranes where it prevents lipid peroxidation and reduces inflammatory signaling cascades including CRP, TNF-α, and adhesion molecules. Meta-analyses of 26+ RCTs show consistent anti-inflammatory effects, with the strongest evidence for CRP reduction (-0.52 mg/L) and TNF-α suppression at doses ≥700 mg/day. This angle focuses on the specific molecular mechanism (lipid peroxidation prevention) leading to measurable inflammatory outcomes across multiple large meta-analyses. It avoids vague antioxidant claims and explains why membrane-bound alpha-tocopherol specifically affects inflammatory cascades.
Diagram glossary
- Alpha-Tocopherol:
- The most active form of vitamin E that protects cell membranes from lipid peroxidation.
- CRP:
- C-reactive protein is a key inflammatory marker reduced by vitamin E supplementation.
- TNF-α:
- Tumor necrosis factor-alpha is an inflammatory signaling molecule suppressed by alpha-tocopherol.
- Alpha-tocopherol
- The most biologically active and abundant form of vitamin E in human tissues, crucial for its anti-inflammatory and antioxidant effects.
- TBARS
- Thiobarbituric acid reactive substances—a laboratory measure of lipid peroxidation that decreases with effective vitamin E supplementation.
- TNF-alpha
- Tumor necrosis factor-alpha, a key inflammatory signaling molecule. elevated in chronic inflammatory conditions and metabolic disease.
- Polyunsaturated fatty acid (PUFA)
- A type of healthy fat found in cell membranes that contains multiple chemical double bonds, making it especially vulnerable to damage from free radicals. Vitamin E specifically protects these fats fro
- Oxidative Stress
- Cellular damage caused by reactive oxygen species (free radicals) overwhelming antioxidant defenses.
- NF-κB (Nuclear Factor Kappa B)
- A protein complex inside cells that acts like an 'on switch' for inflammation—when activated by oxidized lipids, it triggers the production of inflammatory molecules.
- C-reactive protein (CRP)
- A protein produced by the liver that rises in the blood when inflammation is present in the body. It is commonly used as a measurable marker to track how much inflammation a person has and whether a t
- CRP
- C-reactive protein is a key inflammatory marker reduced by vitamin E supplementation.
- TNF-α
- Tumor necrosis factor-alpha is an inflammatory signaling molecule suppressed by alpha-tocopherol.
Alpha-Tocopherol’s Membrane Protection: The Molecular Basis
Alpha-tocopherol, the key form of vitamin E, embeds itself within cell membranes where it neutralizes lipid radicals and prevents the chain reaction of lipid peroxidation. This membrane stabilization is the cornerstone of vitamin E’s anti-inflammatory mechanism, blocking the propagation of oxidized lipids that would otherwise activate immune signaling pathways.
At the molecular level, alpha-tocopherol donates a hydrogen atom to lipid peroxyl radicals, producing a stable tocopheroxyl radical and halting the self-propagating cycle of membrane damage. This action is not just antioxidant in a generic sense; it specifically preserves the integrity of polyunsaturated fatty acid (PUFA)-rich membranes that are most vulnerable to oxidative attack. By protecting these membranes, vitamin E inhibits the release of oxidized lipids and their downstream signaling, including the activation of nuclear factor kappa B (NF-κB) and other inflammatory transcription factors [1].
Cell and animal studies confirm that alpha-tocopherol’s membrane localization is critical—other forms of vitamin E or systemic antioxidants do not substitute for this precise effect [4,6]. In human plasma and erythrocytes, supplementation with alpha-tocopherol increases resistance to ex vivo lipid peroxidation, as demonstrated by reduced malondialdehyde (MDA) and thiobarbituric acid reactive substances (TBARS) after supplementation [2,3]. This mechanistic link directly connects the presence of alpha-tocopherol in membranes with reduced oxidative stress markers, setting the stage for broader anti-inflammatory effects.
Interrupting Inflammatory Signaling: Downstream Effects on CRP and TNF-α
By preventing membrane lipid peroxidation, alpha-tocopherol reduces the activation and release of pro-inflammatory mediators such as CRP and TNF-α. This effect is consistently observed in human studies, with CRP dropping by an average of 0.52 mg/L and TNF-α requiring higher doses for significant suppression.
A comprehensive meta-analysis of 26 randomized controlled trials involving 2,102 participants found that vitamin E supplementation lowered CRP levels by 0.52 mg/L compared to placebo—a clinically meaningful reduction that can shift individuals from moderate to low inflammatory risk categories [11]. This effect appears at doses commonly used in supplementation (400-800 mg/day), with the most consistent results seen in this range. For TNF-α, the dose-response is more pronounced: reductions become statistically significant primarily at intakes of 700 mg/day or higher, as established by multiple studies [1].
The mechanism underlying these effects stems from vitamin E's ability to prevent oxidized lipids from activating NF-κB and related transcription factors. When cell membranes remain intact, they don't release the inflammatory signals that prompt the liver to produce CRP or immune cells to secrete TNF-α. While laboratory studies reinforce these mechanistic links, the reproducibility of CRP reduction across diverse human populations—from healthy adults to those with metabolic syndrome—establishes vitamin E as a clinically relevant anti-inflammatory nutrient with predictable biomarker effects.
Optimal Dosing, Form, and Bioavailability for Maximum Anti-inflammatory Effect
For reducing inflammation via CRP and TNF-α, human evidence supports a moderate to high dose of alpha-tocopherol, with form and bioavailability playing crucial roles. The best results appear with daily doses between 400 and 800 mg, as both efficacy and safety data cluster in this range.
RCTs indicate that CRP reduction is achievable at the lower end of this spectrum (200–400 mg/day), while suppression of TNF-α, a more difficult biomarker to shift, requires higher doses—typically at or above 700 mg/day [1]. The most effective formulations use natural d-alpha-tocopherol, which has higher biological activity and tissue retention than synthetic dl-alpha-tocopherol or mixed tocopherols [2]. Enhanced delivery systems, such as phospholipid complexes or nanoemulsions, are being explored to increase plasma and membrane alpha-tocopherol concentrations, although most human data to date use standard oil-based softgels [10].
Bioavailability can be influenced by meal composition (higher with fat), genetics, and baseline vitamin E status [3]. For individuals with higher oxidative stress or lower baseline tocopherol, the response to supplementation may be more pronounced. Importantly, doses above 1,000 mg/day have not demonstrated further clinical benefit and may increase risk, suggesting an optimal window between 400–800 mg/day for most anti-inflammatory applications.
Key Biomarkers: Interpreting CRP, TNF-α, and Lipid Peroxidation Markers
CRP and TNF-α are the most consistently responsive biomarkers to vitamin E supplementation, with CRP showing a reliable 0.52 mg/L average reduction and TNF-α responding primarily to higher doses. Lipid peroxidation markers like malondialdehyde (MDA) and TBARS also decrease with supplementation, providing direct evidence of membrane protection.
CRP levels below 1 mg/L indicate low inflammatory risk, 1-3 mg/L represents moderate risk, and above 3 mg/L signals high inflammation. The 0.52 mg/L reduction demonstrated in meta-analyses can meaningfully shift risk categories—for example, moving someone from 2.5 mg/L (moderate risk) to under 2.0 mg/L (lower moderate risk) [11]. TNF-α is measured less routinely in clinical practice but serves as an important research marker; reductions of 10-20% are observed in studies using 700 mg/day or higher of alpha-tocopherol [1].
Lipid peroxidation markers like MDA and TBARS directly measure oxidative membrane damage. Vitamin E supplementation consistently lowers these markers in both plasma and red blood cells, with the largest decreases seen in populations facing higher oxidative stress—smokers show 25-30% reductions in TBARS, while healthy adults typically see 10-15% improvements [2,3]. For those not tracking biomarkers, these surrogate markers link to long-term inflammatory risk reduction, supporting vitamin E's protective role even without laboratory monitoring. The pattern is clear: membrane protection translates to measurable anti-inflammatory effects across multiple biomarkers.
Population Differences: Baseline Oxidative Stress and Vitamin E Response
The anti-inflammatory and membrane-protective effects of vitamin E are more pronounced in individuals with higher baseline oxidative stress, such as smokers or those with metabolic syndrome. This is reflected in both the degree of biomarker change and the magnitude of clinical improvement seen in supplementation trials.
Human RCTs consistently show that the greatest reductions in CRP, TNF-α, and lipid peroxidation markers occur in populations with elevated baseline markers or increased exposure to reactive oxygen species [3]. For example, a study comparing smokers and nonsmokers found a greater decrease in erythrocyte peroxidation and a larger rise in membrane vitamin E following supplementation in smokers, likely due to their higher oxidative burden [3]. Similarly, individuals with metabolic syndrome or insulin resistance demonstrate more substantial improvements in inflammatory and oxidative stress markers when given vitamin E, especially at moderate to high doses [2].
The underlying reason is straightforward: when oxidative stress is high, more alpha-tocopherol is incorporated into membranes to counteract increased free radical activity, leading to greater net benefit. This does not mean individuals with low baseline stress do not benefit, but the marginal effect is smaller and may not yield measurable changes in standard biomarkers. The table below summarizes key population differences:
| Population | Baseline Oxidative Stress | CRP/TNF-α Response | Lipid Peroxidation Response | |----------------------|--------------------------|--------------------|----------------------------| | Smokers | High | Strong | Strong | | Metabolic syndrome | High | Moderate-strong | Strong | | Healthy adults | Low-moderate | Modest | Modest | | Elderly | Variable | Variable | Moderate |
These findings reinforce the importance of identifying individual oxidative stress profiles when considering vitamin E supplementation for inflammation.
Comparing Alpha-Tocopherol with Other Antioxidants and Formulations
Alpha-tocopherol’s unique ability to integrate into cell membranes distinguishes it from other antioxidants and vitamin E analogues. Not all vitamin E forms, or antioxidant supplements, provide the same membrane protection or anti-inflammatory benefit. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Natural d-alpha-tocopherol is more bioactive than synthetic dl-alpha-tocopherol, with studies indicating it is retained in tissues at roughly double the rate [2]. Mixed tocopherol blends and tocotrienols have different tissue distributions and, while they offer antioxidant activity, have not shown the same consistency in lowering CRP and TNF-α in human RCTs. Comparisons with vitamin C and other antioxidants reveal that only alpha-tocopherol consistently reduces LDL oxidation and membrane lipid peroxidation in vivo [1].
Advanced delivery forms, such as nanoemulsions and phospholipid complexes, may enhance absorption and tissue delivery, but human data are limited [10]. Most clinical trials demonstrating anti-inflammatory effects have used standard oil-based alpha-tocopherol softgels. The table below summarizes key differences:
| Antioxidant/Form | Membrane Integration | CRP/TNF-α Reduction | LDL Oxidation Reduction | |-------------------------------|---------------------|---------------------|------------------------| | Alpha-tocopherol (natural) | High | Strong | Strong | | Alpha-tocopherol (synthetic) | Moderate | Moderate | Moderate | | Mixed tocopherols/tocotrienols | Variable | Inconsistent | Variable | | Vitamin C | Low | Minimal | Minimal | | Nano/Phospholipid E | High (potential) | Unproven | Not established |
This comparison highlights why alpha-tocopherol, especially in its natural form, is the preferred supplement for targeting membrane-driven inflammation.
Conclusions
Alpha-tocopherol's membrane-protective mechanism translates into consistent reductions in inflammatory biomarkers, with CRP dropping by an average of 0.52 mg/L and TNF-α responding to higher doses (≥700 mg/day). The optimal anti-inflammatory dose range of 400-800 mg/day of natural d-alpha-tocopherol is supported by multiple meta-analyses and offers the best balance of efficacy and safety. Individuals with higher oxidative stress—smokers, those with metabolic syndrome—see the most pronounced benefits, though improvements occur across populations. While advanced formulations may enhance absorption, standard oil-based softgels remain the most studied and reliable option. Vitamin E's unique ability to embed in cell membranes and halt lipid peroxidation distinguishes it from other antioxidants and explains its specific effectiveness against inflammation-driving oxidative damage.
Despite robust biomarker evidence, vitamin E’s effects on long-term clinical outcomes such as cardiovascular events remain unproven. Most studies use surrogate markers like CRP and TNF-α rather than hard endpoints. The optimal dose range is well-defined for inflammation, but responses can vary based on baseline oxidative stress, genetics, and nutrient status. Safety concerns arise with doses above 1,000 mg/day, and advanced formulations lack large-scale human data. Direct comparisons with other antioxidants are limited, and many mechanistic claims rely on cell or animal models rather than human trials. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how vitamin e relates to your health goals and monitor changes in your biomarkers over time.
