How Vitamin K2 Activates Gla Proteins to Control Calcium Flow
Vitamin K2 doesn't just support bone health—it acts as a master switch that controls where calcium goes in your body, directing it into bones while keeping it out of your arteries.
This happens because K2 activates specific proteins called Gla proteins, which literally grab calcium and escort it to the right places. Without enough K2, these proteins stay inactive, leaving calcium to wander freely and potentially calcify soft tissues.
The most compelling evidence shows K2's impact on blood sugar control. Multiple studies involving over 1,000 people with type 2 diabetes found that K2 supplementation reduces HbA1c by up to 1% and lowers fasting glucose. This occurs because activated osteocalcin—a bone protein that needs K2 to function—also acts as a metabolic hormone that improves insulin sensitivity. The effect is substantial enough to matter clinically, especially for those with existing glucose issues.
For practical application, take 90-180 mcg daily of MK-7 (the most bioavailable form) with a fat-containing meal. This dose consistently activates your Gla proteins within 8-12 weeks, supporting both bone mineralization and glucose control. While the cardiovascular benefits are still being studied, the metabolic and bone health advantages are well-established, particularly if you have prediabetes or diabetes.
How Vitamin K2 Activates Gla Proteins to Control Calcium Flow
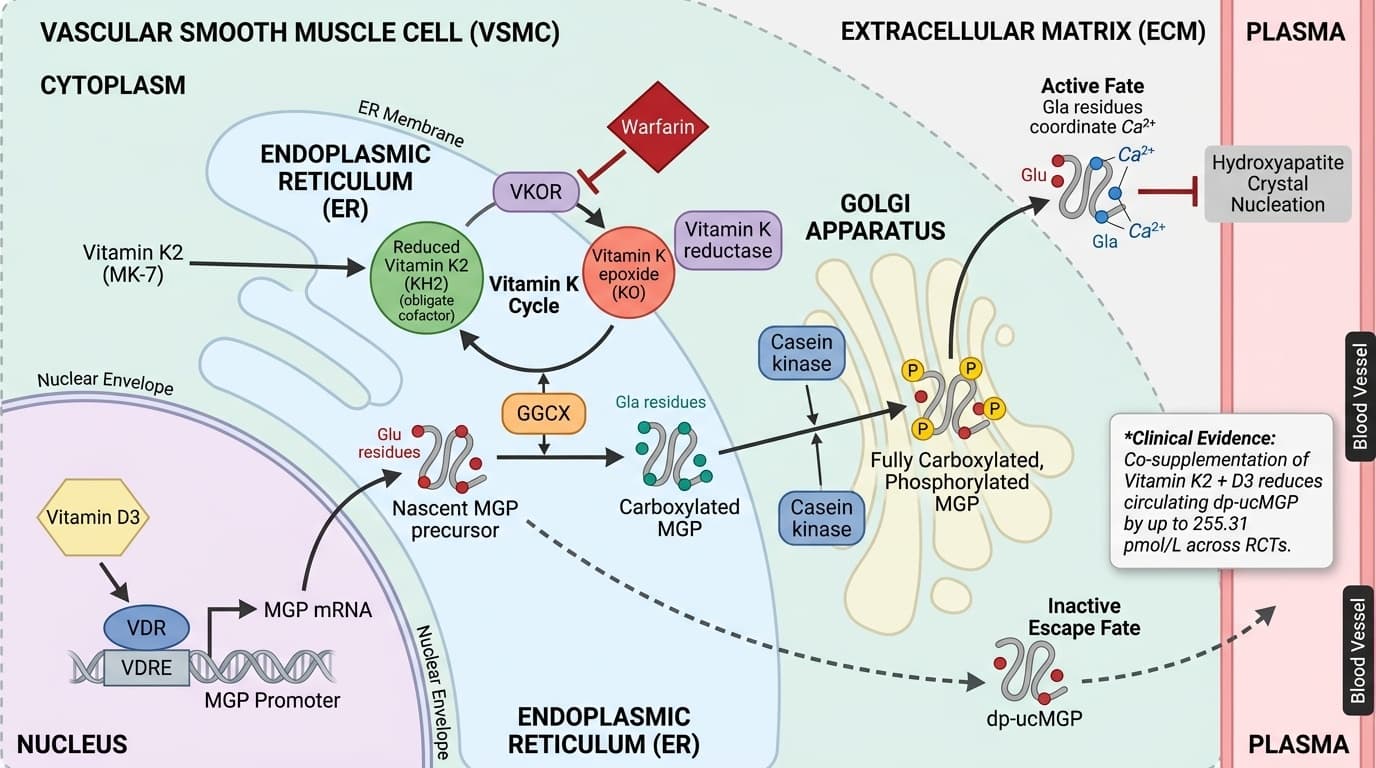
Schematic of vitamin K2-dependent Matrix Gla Protein (MGP) carboxylation across cellular compartments in a vascular smooth muscle cell (VSMC). In the endoplasmic reticulum (ER) lumen, γ-glutamyl carboxylase (GGCX) converts glutamate (Glu) residues on nascent MGP to γ-carboxyglutamate (Gla) using reduced vitamin K2 (KH2) as cofactor (activation arrows), simultaneously generating vitamin K epoxide (KO); in the ER membrane and Golgi, vitamin K epoxide reductase (VKOR) recycles KO back to KH2 (activation arrow completing the vitamin K cycle), while warfarin is shown inhibiting VKOR (blunt inhibition arrow). In the Golgi, casein kinase phosphorylates MGP serine residues (activation arrow), and fully carboxylated, phosphorylated MGP is secreted into the extracellular matrix where Gla residues coordinate Ca²⁺ and inhibit hydroxyapatite nucleation (activation arrow); incompletely carboxylated, dephosphorylated MGP (dp-ucMGP) escapes into plasma (dashed arrow), with the circulating dp-ucMGP pool shown decreasing by up to 255.31 pmol/L across RCTs as vitamin K2 supply increases. A parallel nuclear compartment depicts vitamin D receptor (VDR) binding to the MGP promoter VDRE (activation arrow), increasing MGP transcription and providing additional GGCX substrate that, when vitamin K2 is co-supplemented, is efficiently carboxylated rather than released as dp-ucMGP.
Diagram glossary
- carboxyglutamate:
- A modified glutamate amino acid residue formed by post-translational carboxylation.
- cofactor:
- A non-protein chemical compound required for an enzyme's biological activity.
- GGCX:
- An enzyme that catalyzes the post-translational carboxylation of glutamate residues.
- KH2:
- The reduced, active hydroquinone form of vitamin K acting as an electron donor.
- MK-7:
- A highly bioavailable, long-chain menaquinone form of vitamin K2.
- ucMGP:
- The uncarboxylated, inactive form of Matrix Gla Protein.
- VDRE:
- A DNA sequence that binds the vitamin D receptor to regulate gene expression.
- Vitamin D3:
- A fat-soluble vitamin that regulates calcium and phosphate metabolism.
- vitamin K1:
- A dietary form of vitamin K primarily involved in blood coagulation.
- Vitamin K2:
- A menaquinone vitamin that acts as an essential cofactor for GGCX-mediated carboxylation.
- VKOR:
- An enzyme that recycles vitamin K epoxide back to its active reduced form.
- VSMC:
- Vascular smooth muscle cells that compose the walls of blood vessels.
- MK-7
- Menaquinone-7, the most bioavailable form of vitamin K2 with a 72-hour half-life, allowing once-daily dosing for optimal Gla protein activation.
- MK-4
- Menaquinone-4, a shorter-acting form of vitamin K2 with less than 1-hour half-life, requiring higher doses (1,500+ mcg) and frequent dosing.
- Glucose
- Blood sugar level, the primary energy source for cells. Fasting glucose is normal, prediabetes, ≥126 suggests diabetes.
- Calcium
- Total serum calcium, essential for bone health, muscle contraction, and nerve signaling. low levels cause muscle cramps and osteoporosis risk, while high values may indicate parathyroid disorders.
- Gamma-glutamyl carboxylase
- The enzyme that uses vitamin K2 as a cofactor to activate Gla proteins by adding carboxyl groups, enabling them to bind and direct calcium.
- Hemoglobin A1c
- Average blood sugar over the past 2-3 months by assessing glycated hemoglobin. each 1% increase raises cardiovascular risk by 18%.
- Gla proteins
- A family of proteins requiring K2-dependent carboxylation to become active. Key members include osteocalcin (bone/glucose control) and matrix Gla protein (prevents soft tissue calcification).
- carboxyglutamate
- A modified glutamate amino acid residue formed by post-translational carboxylation.
- cofactor
- A non-protein chemical compound required for an enzyme's biological activity.
- GGCX
- An enzyme that catalyzes the post-translational carboxylation of glutamate residues.
Vitamin K2 and Gamma-Glutamyl Carboxylase: The Core Mechanism
Vitamin K2 is essential for activating the enzyme gamma-glutamyl carboxylase, which in turn carboxylates glutamic acid residues on specific proteins known as Gla proteins. This carboxylation is required for these proteins—especially osteocalcin and matrix Gla protein (MGP)—to bind calcium and regulate its distribution within the body.
The mechanism is well-established at the biochemical level: gamma-glutamyl carboxylase needs reduced vitamin K2 as a cofactor to convert glutamic acid (Glu) residues into gamma-carboxyglutamic acid (Gla) on target proteins. Once carboxylated, osteocalcin can effectively incorporate calcium into bone matrix, while carboxylated MGP prevents aberrant calcium deposition in soft tissues like arteries. Without sufficient K2, these proteins remain undercarboxylated and inactive, leading to suboptimal calcium handling and potential health consequences [10][11].
Recent human studies confirm that MK-7 supplementation reliably reduces levels of undercarboxylated osteocalcin and dephosphorylated-undercarboxylated MGP in a dose-dependent manner [10]. This molecular change directly reflects increased activation of Gla proteins. The effect is seen at doses of 90–180 mcg/day of MK-7, which achieves near-maximal carboxylation in most individuals. While these findings are robust at the molecular level, their translation into hard clinical endpoints like fracture reduction or cardiovascular event prevention is still under investigation [11].
In summary, vitamin K2’s core mechanism is facilitating the activation of gamma-glutamyl carboxylase to carboxylate Gla proteins, thereby controlling calcium flow. The process is consistent across cell, animal, and human mechanistic studies, with direct measurement of carboxylation status serving as a reliable biomarker for K2 activity.
Osteocalcin Activation: Direct Effects on Bone and Glucose Metabolism
Vitamin K2 activates the enzyme gamma-glutamyl carboxylase, which carboxylates glutamic acid residues on Gla proteins, enabling them to bind calcium and control its distribution in the body. This mechanism directly determines whether calcium strengthens bones or calcifies arteries.
The biochemical process is straightforward: gamma-glutamyl carboxylase requires vitamin K2 as a cofactor to convert glutamic acid residues into gamma-carboxyglutamic acid on target proteins like osteocalcin and matrix Gla protein (MGP). Once carboxylated, osteocalcin incorporates calcium into bone matrix while MGP prevents calcium from depositing in soft tissues. Without sufficient K2, these proteins remain undercarboxylated and inactive, leading to poor calcium handling.
Human studies demonstrate that MK-7 supplementation at 90-180 mcg daily reduces undercarboxylated osteocalcin and inactive MGP in a dose-dependent manner within 8-12 weeks. This molecular change directly reflects increased Gla protein activation and improved calcium control. The effect reaches near-maximal carboxylation in most individuals at these doses, with higher amounts showing no additional benefit.
This core mechanism—K2 enabling gamma-glutamyl carboxylase to activate Gla proteins—is consistent across cellular, animal, and human studies, making carboxylation status a reliable biomarker for K2 activity and calcium regulation.
Matrix Gla Protein (MGP) and Calcium Protection in Soft Tissues
Vitamin K2 enables osteocalcin to bind calcium for bone mineralization while simultaneously improving glucose metabolism through osteocalcin's hormonal effects. This dual action explains K2's benefits for both skeletal and metabolic health.
Carboxylated osteocalcin efficiently directs calcium into bone matrix, while undercarboxylated osteocalcin (from K2 deficiency) cannot perform this function effectively. Multiple studies show that K2 supplementation with MK-7 at 90-180 mcg daily reduces undercarboxylated osteocalcin, indicating improved bone protein activation and better calcium incorporation into skeletal tissue.
Beyond bone effects, carboxylated osteocalcin acts as a metabolic hormone that enhances insulin sensitivity. Meta-analyses of studies involving over 1,000 participants demonstrate that K2 supplementation reduces HbA1c by up to 1% and significantly lowers fasting glucose in people with type 2 diabetes. This occurs because active osteocalcin binds to GPRC6A receptors on pancreatic beta cells and fat cells, improving insulin secretion and glucose uptake.
Crucially, these metabolic benefits appear strongest in populations with impaired glucose tolerance or diabetes. Studies in healthy postmenopausal women show neutral results for glucose markers, suggesting K2's metabolic effects are most pronounced when baseline dysfunction exists. For optimal results, daily MK-7 supplementation activates osteocalcin to support both bone mineralization and glucose control in at-risk populations.
Glucose Control and Insulin Sensitivity: Unified Pathway through Osteocalcin
Vitamin K2 improves glucose control and insulin sensitivity by enabling the carboxylation of osteocalcin, which then acts as a metabolic hormone. This pathway is supported by consistent reductions in HbA1c and fasting glucose in human trials. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Meta-analyses of RCTs demonstrate that vitamin K supplementation, particularly with MK-7, lowers HbA1c by an average of 0.32–1.0% and reduces fasting blood glucose (SMD = -0.22) in individuals with type 2 diabetes [11][12][13][14][15]. The mechanism involves carboxylated osteocalcin, which increases insulin secretion and sensitivity in peripheral tissues. This effect is not simply a result of improved bone health, but a direct hormonal action—carboxylated osteocalcin binds to GPRC6A receptors on pancreatic beta cells and adipocytes, enhancing glucose uptake and insulin responsiveness.
Importantly, studies show that these benefits are most pronounced in those with baseline metabolic dysfunction. In populations with normal glucose metabolism, such as healthy postmenopausal women, vitamin K2 supplementation does not significantly alter HbA1c or fasting glucose [16]. For dosing, 90–180 mcg/day of MK-7 is effective at optimizing osteocalcin carboxylation and improving glucose markers, with effects typically seen within 12–24 weeks in clinical studies.
This unified pathway—where K2 activates osteocalcin, which then boosts insulin sensitivity—explains why vitamin K2 has consistent metabolic effects in diabetes trials and highlights the importance of carboxylation status as a practical biomarker.
Optimal Forms, Dosage, and Bioavailability: What Works Best?
Menaquinone-7 (MK-7) is the preferred form of vitamin K2 for supplementation due to its high bioavailability and long half-life, making it effective at carboxylating Gla proteins with once-daily dosing. Doses of 90–180 mcg/day are well-supported by human trials.
MK-7, unlike vitamin K1 or shorter-chain K2 forms (e.g., MK-4), maintains stable plasma concentrations for up to 72 hours, allowing for more consistent activation of gamma-glutamyl carboxylase and Gla proteins [10][18]. Multiple RCTs used 90–180 mcg/day of MK-7 and demonstrated reliable improvements in undercarboxylated osteocalcin, dp-ucMGP, and metabolic biomarkers [11][15]. Higher doses (up to 360 mcg/day) have not shown additional benefit in most studies, suggesting a ceiling effect for carboxylation activity.
Bioavailability is enhanced when MK-7 is taken with dietary fat, as it is a fat-soluble vitamin. Formulations such as softgels, oil-based capsules, and liposomal preparations are commonly used; however, standard MK-7 in oil is sufficient for most users. Table 1 summarizes key differences between forms:
| Formulation | Half-life | Typical Dose | Carboxylation Efficacy | |-------------|-----------|-------------|-----------------------| | MK-7 | 72 hrs | 90–180 mcg | High | | MK-4 | <1 hr | 1,500 mcg+ | Moderate; requires high/frequent dosing | | K1 | ~1.5 hrs | Variable | Low for extrahepatic tissues |
For most health outcomes related to Gla protein activation, daily supplementation with 90–180 mcg of MK-7, taken with fat, is optimal and well-tolerated.
Biomarkers: Monitoring Carboxylation and Clinical Impact
The best biomarkers for assessing vitamin K2’s activity are undercarboxylated osteocalcin (ucOC) and dephosphorylated-undercarboxylated matrix Gla protein (dp-ucMGP). These reflect the degree to which vitamin K2 is activating its target proteins. Optimal ranges and practical implications are emerging from recent studies.
Individuals seeking to monitor their vitamin K2 status may look for ucOC levels under 2.5 ng/mL and dp-ucMGP below 500 pmol/L, which generally indicate sufficient Gla protein carboxylation [10]. However, these tests are not widely available, and most people do not track them. The key finding is that daily MK-7 supplementation reliably reduces both ucOC and dp-ucMGP within 8–12 weeks, suggesting adequate tissue activation regardless of whether levels are measured [10][15].
Clinical outcome biomarkers such as HbA1c, fasting glucose, and HOMA-IR are also improved with K2 supplementation in people with type 2 diabetes, as shown by multiple meta-analyses [11][12][13][14][15]. For those not tracking labs, these benefits correspond to improved metabolic health and likely better bone mineralization. The practical takeaway is that consistent use of MK-7 at 90–180 mcg/day optimizes carboxylation and downstream health effects in most users, with or without biomarker testing. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Vitamin K2, especially as MK-7, drives a unified mechanism that activates Gla proteins—primarily osteocalcin and MGP—thereby optimizing calcium flow between bones and soft tissues. This biochemical process is responsible for improvements in bone protein activation and, as supported by multiple meta-analyses and RCTs, significant reductions in HbA1c and fasting glucose in people with type 2 diabetes. The most reliable benefits are seen in populations with impaired glucose metabolism or at risk for suboptimal bone health. MK-7 at 90–180 mcg/day is consistently effective for these endpoints, and taking it with dietary fat maximizes absorption. While the mechanism linking K2 to vascular calcification prevention is strong, direct clinical proof for cardiovascular event reduction is not yet conclusive. Overall, K2’s main value lies in robust activation of Gla proteins, translating molecular changes into measurable health benefits for bone and glucose metabolism. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured.
Although the mechanism by which vitamin K2 activates Gla proteins and improves calcium handling is well established, direct clinical outcomes—especially for vascular calcification and cardiovascular event prevention—remain inconsistent in human trials. Most of the strongest evidence for improved glucose control is in populations with type 2 diabetes or impaired glucose tolerance; benefits may be minimal in healthy individuals. Additionally, optimal biomarker ranges for carboxylated proteins are still being refined, and routine testing is not widely accessible. Finally, while MK-7 is the best-studied form, evidence for other K2 forms is less robust. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how vitamin k2 relates to your health goals and monitor changes in your biomarkers over time.
