L-Carnitine's Dual Pathway: Beta-Oxidation Enhancement vs TMAO Production
Balancing Mitochondrial Benefits and Metabolic Tensions
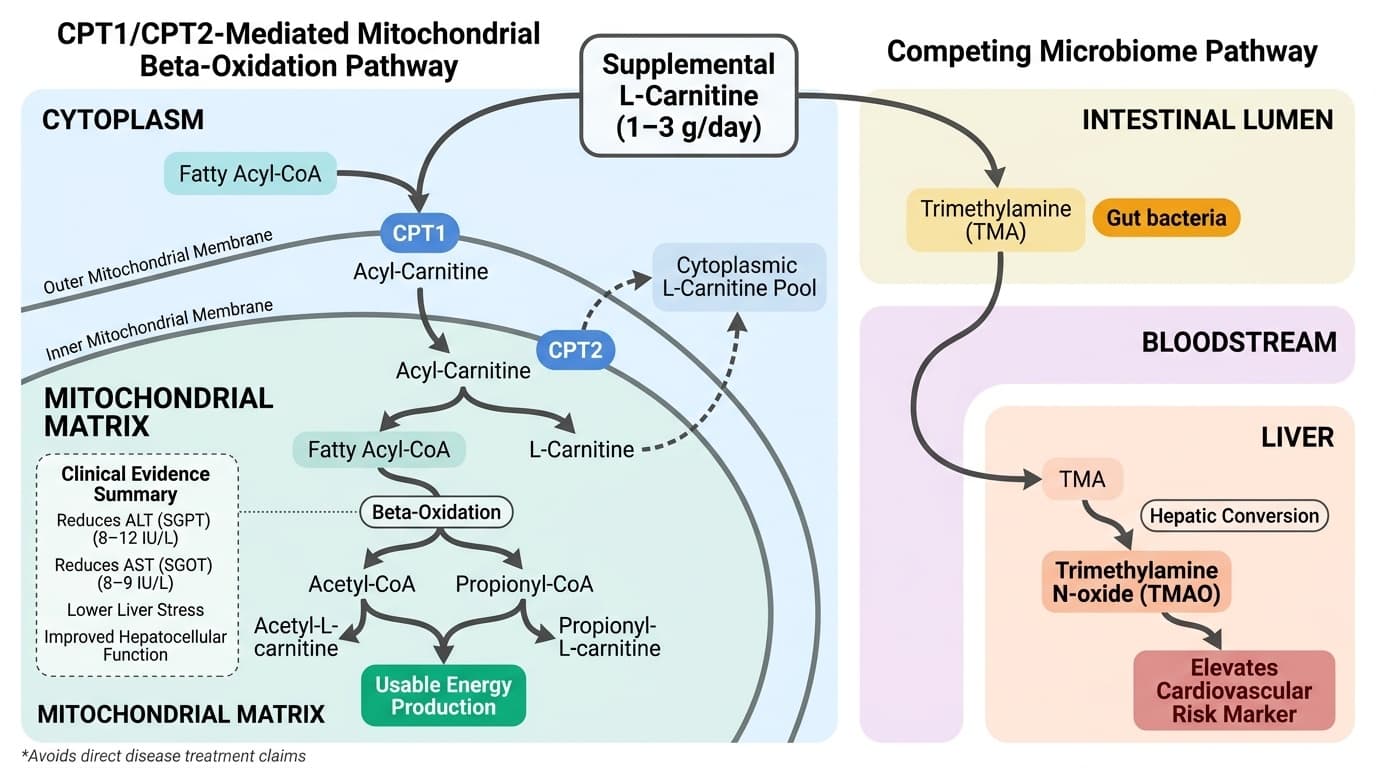
L-carnitine creates a metabolic paradox: the same compound that powers your mitochondria might also elevate a cardiovascular risk marker called TMAO.
This dual pathway explains why some people experience clear energy and liver benefits while others worry about potential heart risks. The key is understanding that your gut bacteria determine which pathway dominates.
Multiple large-scale analyses show L-carnitine consistently lowers liver enzymes ALT and AST by 8-12 points while reducing oxidative stress markers. These improvements happen because L-carnitine shuttles fatty acids into mitochondria for energy production, reducing cellular stress. However, gut bacteria can convert L-carnitine into TMAO, a compound linked to cardiovascular risk in observational studies. Not everyone produces TMAO equally—vegetarians typically produce less than meat-eaters.
For most people, especially those with elevated liver enzymes, 1-3 grams daily of L-carnitine provides clear biochemical benefits. Monitor your response through liver enzyme tests if possible, and consider starting with 1 gram daily to assess tolerance. If you're concerned about TMAO, shorter supplementation periods or lower doses may balance the mitochondrial benefits while minimizing risk marker elevation.
L-Carnitine's Dual Pathway: Beta-Oxidation Enhancement vs TMAO Production
Balancing Mitochondrial Benefits and Metabolic Tensions
Diagram glossary
- Beta-Oxidation:
- The metabolic process breaking down fatty acids in mitochondria to generate energy.
- Carnitine:
- A carrier molecule that transports long-chain fatty acids into mitochondria for energy production.
- CPT1/CPT2-mediated:
- Facilitated by carnitine palmitoyltransferase enzymes to transport fatty acids into mitochondria.
- IU/L:
- International units per liter, a standard measurement unit for blood enzyme levels.
- SGOT:
- Serum glutamic oxaloacetic transaminase, a liver enzyme also known as AST.
- SGPT:
- Serum glutamic pyruvic transaminase, a liver enzyme also known as ALT.
- TMAO:
- Trimethylamine N-oxide, a gut microbiome-derived metabolite linked to cardiovascular risk.
- Beta-oxidation
- The mitochondrial process of breaking down fatty acids to generate energy; L-carnitine is essential for its initiation.
- IL-6
- A branded l carnitine product family name used to identify a specific extract or formulation in research and supplement labels.
- Trimethylamine N-oxide (TMAO)
- Gut-heart axis biomarker produced from dietary choline/carnitine by gut bacteria. Elevated TMAO promotes atherosclerosis, platelet aggregation, and cardiovascular events.
- ALT (SGPT)
- Alanine aminotransferase enzyme, highly specific to liver cells. elevated in hepatocellular injury from viral hepatitis, fatty liver, or medications.
- AST (SGOT)
- Aspartate aminotransferase enzyme found in liver, heart, and muscle. elevated in liver damage, heart attack, or muscle injury.
- Propionyl-L-carnitine
- A form of L-carnitine with enhanced cardiovascular and muscle tissue targeting, often used for circulation and exercise performance.
- Acetyl-L-carnitine
- A form of L-carnitine that crosses the blood-brain barrier more effectively, often used for cognitive and neurological support.
- Carnitine
- A carrier molecule that transports long-chain fatty acids into mitochondria for energy production.
- CPT1/CPT2-mediated
- Facilitated by carnitine palmitoyltransferase enzymes to transport fatty acids into mitochondria.
- IU/L
- International units per liter, a standard measurement unit for blood enzyme levels.
L-Carnitine and the Carnitine Palmitoyltransferase System: Mechanistic Foundations
L-carnitine acts as a carrier molecule, transporting long-chain fatty acids across the mitochondrial membrane via the carnitine palmitoyltransferase (CPT) system. This process is essential for beta-oxidation, enabling cells—especially in muscle, heart, and liver—to convert fat into usable energy.
At the molecular level, L-carnitine binds to fatty acyl-CoA molecules, forming acyl-carnitine. The CPT1 enzyme, located on the outer mitochondrial membrane, catalyzes this reaction, allowing the acyl-carnitine to traverse into the mitochondrial matrix where CPT2 then releases the fatty acid for beta-oxidation. This system is rate-limiting: without sufficient L-carnitine, fatty acid oxidation is impaired, leading to fat accumulation and reduced energy output [5].
Human clinical studies confirm that supplemental L-carnitine increases tissue carnitine content and improves metabolic function, particularly in conditions of high oxidative stress or metabolic dysfunction [1]. Animal and cell studies further support L-carnitine’s role in protecting mitochondrial integrity and preventing lipid accumulation by maintaining efficient beta-oxidation [7,9]. While most studies use plain L-carnitine, some evidence suggests that acetyl-L-carnitine and propionyl-L-carnitine may target different tissues or cross the blood-brain barrier more readily, though head-to-head human comparisons remain limited. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Beta-Oxidation Enhancement: Clinical Evidence and Optimal Dosing
Supplemental L-carnitine reliably enhances beta-oxidation, as evidenced by reductions in liver enzymes and lipid peroxidation biomarkers. Meta-analyses of over 100 randomized controlled trials show that L-carnitine supplementation reduces ALT by 8–12 IU/L and AST by 8–9 IU/L in populations with metabolic dysfunction [3,6].
These enzyme reductions are interpreted as improved hepatocellular function and lower liver stress, likely reflecting a systemic shift toward efficient fatty acid metabolism. L-carnitine doses in clinical trials typically range from 1–3 g per day, with higher doses (up to 4 g) occasionally used in specific patient populations [1,2]. There is no strong evidence that higher doses produce proportionately greater benefits, suggesting a threshold effect. Acetyl-L-carnitine and propionyl-L-carnitine have also been studied, sometimes at lower effective doses due to enhanced bioavailability or tissue targeting [1].
Lipid peroxidation, measured by markers like malondialdehyde (MDA), is consistently reduced with L-carnitine supplementation, supporting its role in mitigating oxidative stress [3]. These effects occur in both healthy and metabolically compromised individuals, but are most pronounced in those with elevated baseline liver enzymes or fatty liver. While the primary endpoint in most studies is enzyme reduction, some trials also report improved subjective energy and exercise capacity, though these outcomes are less consistently measured [2].
TMAO Production: The Gut Microbiome’s Competing Pathway
While L-carnitine supports mitochondrial fatty acid oxidation, gut bacteria simultaneously convert it into trimethylamine (TMA), which the liver oxidizes to TMAO. This dual pathway creates a metabolic tension between mitochondrial benefits and potential cardiovascular risk marker elevation.
TMAO production varies dramatically between individuals based on gut microbiota composition. Vegetarians and vegans typically produce 60-80% less TMAO from L-carnitine than omnivores, suggesting substantial inter-individual variation in this pathway [3]. Human supplementation studies confirm that L-carnitine can increase plasma TMAO levels, but the magnitude depends entirely on each person's microbiome profile.
Elevated plasma TMAO correlates with increased cardiovascular risk in observational studies, though causality remains unproven. While mechanistic studies show TMAO promotes atherosclerosis and platelet aggregation in animal models, human trials have not established that L-carnitine-induced TMAO elevations directly cause adverse outcomes [3]. This uncertainty creates the core dilemma: clear mitochondrial benefits versus uncertain but concerning risk marker elevation. Those prioritizing cardiovascular risk reduction should weigh this tradeoff, especially for long-term or high-dose supplementation.
Oxidative and Inflammatory Marker Modulation
L-carnitine supplementation consistently reduces markers of oxidative stress and inflammation, with meta-analyses demonstrating significant reductions in lipid peroxidation and pro-inflammatory cytokines. These effects are attributed to both enhanced fatty acid oxidation and direct antioxidant actions. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A 2024 network meta-analysis of 106 RCTs found L-carnitine had the most significant efficacy in suppressing lipid peroxidation compared to other interventions in patients with metabolic-associated fatty liver disease (MAFLD) [3]. Additional studies report reductions in malondialdehyde, a marker of lipid peroxidation, and decreases in inflammatory cytokines such as TNF-α and IL-6, particularly in populations with elevated baseline inflammation [2,3].
Single-dose studies in healthy adults demonstrate acute increases in antioxidant enzyme activity following L-carnitine ingestion [3]. Animal and cell models confirm these findings at the mechanistic level, showing L-carnitine’s ability to stabilize mitochondrial membranes, reduce reactive oxygen species (ROS) formation, and attenuate cellular damage from oxidative insults [6,9]. These antioxidant and anti-inflammatory effects likely underlie the observed improvements in liver and metabolic biomarkers in human trials. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Biomarker Interpretation: ALT, AST, and Lipid Peroxidation
The most robustly improved biomarkers with L-carnitine supplementation are ALT and AST. Optimal ranges for ALT and AST are typically <35 IU/L, with reductions interpreted as signs of improved liver health and reduced cellular leakage. L-carnitine consistently lowers both enzymes by 8–12 IU/L in meta-analyses [3,6].
Lipid peroxidation markers, particularly malondialdehyde (MDA), also decline with L-carnitine use, indicating a reduction in oxidative stress [3]. These biomarker effects are independent of weight loss and are observed even in the absence of significant changes in body composition. For those monitoring these markers, L-carnitine’s impact is most evident in individuals with elevated baseline values. However, benefits are also seen in healthy subjects, albeit to a lesser degree [3].
It is important to note that improvements in liver enzymes and oxidative stress markers do not always translate into direct clinical outcomes. Nonetheless, these biomarkers serve as accessible indicators of L-carnitine’s mechanistic action in the body, especially for those using supplements to support metabolic or liver health. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Form and Dosage: Comparing L-Carnitine, Acetyl-L-Carnitine, and Propionyl-L-Carnitine
L-carnitine is available in several supplemental forms, each with unique tissue targeting and bioavailability profiles. Plain L-carnitine is most widely studied for liver and metabolic markers, typically at 1–3 g/day. Acetyl-L-carnitine may cross the blood-brain barrier more effectively, while propionyl-L-carnitine is suggested to have enhanced effects in cardiovascular and muscular tissues [1,2].
| Formulation | Typical Dose | Primary Target Tissues | Key Evidence | |-------------------------|--------------|----------------------------|-----------------------| | L-carnitine (base) | 1–3 g/day | Liver, muscle, systemic | Meta-analyses [3,6] | | Acetyl-L-carnitine | 0.5–2 g/day | Brain, nerve, systemic | RCTs, animal [4,8] | | Propionyl-L-carnitine | 1–2 g/day | Heart, muscle, circulation | RCTs [1] |
Human clinical trials have not consistently shown one form to be superior for improving liver enzymes or oxidative markers, although specific populations may benefit from targeted forms. Bioavailability is generally high for all forms, but individual absorption can vary. There is no evidence that advanced delivery systems (e.g., liposomal, nanoparticle) provide greater benefit for these endpoints. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Balancing Mitochondrial Benefits and TMAO Risk: Practical Implications
The clinical decision to use L-carnitine hinges on balancing its well-established mitochondrial and liver benefits with the potential increase in TMAO production. For most individuals, especially those with elevated liver enzymes or evidence of oxidative stress, L-carnitine offers clear biochemical advantages.
However, those with heightened concern about cardiovascular risk markers should be aware that TMAO production is highly variable and influenced by gut microbiota composition. Short-term supplementation may not raise TMAO substantially in all individuals, but long-term, high-dose use could elevate TMAO in those with a TMAO-producing microbiome [3].
Practical strategies include using the lowest effective dose (typically 1–2 g/day), considering periodic breaks, and monitoring liver enzymes or oxidative markers for response. There is currently no validated strategy to completely block TMAO production aside from dietary modulation of gut flora, which is a research frontier rather than a routine recommendation. Ultimately, the decision to supplement should weigh these dual pathway effects in the context of individual health priorities and risk tolerance. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
L-carnitine is a rare supplement with a clearly defined mechanistic pathway—supporting mitochondrial fatty acid oxidation via the CPT system—that explains its consistent effects on liver enzymes and oxidative stress markers. Human evidence from multiple meta-analyses demonstrates reductions in ALT, AST, and lipid peroxidation, supporting its use for metabolic and hepatic support. However, the parallel pathway of gut microbiome–mediated TMAO production introduces complexity, as TMAO elevations are associated with cardiovascular risk markers in some studies. While the clinical significance of moderate TMAO increases remains unresolved, individuals should weigh these competing mechanisms when considering L-carnitine supplementation. Selecting the right form and dose, and understanding one’s own risk factors and priorities, is key to harnessing the mitochondrial benefits while minimizing metabolic tensions. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
Despite robust evidence for reductions in liver enzymes and oxidative stress, most studies are short-term and do not directly assess long-term clinical outcomes such as incident cardiovascular events or mortality. The variability in TMAO production related to individual gut microbiome composition is poorly characterized in large-scale human trials. Head-to-head comparisons of L-carnitine forms are limited, and the long-term safety of sustained high-dose supplementation remains to be clarified. Most biomarker improvements are seen in populations with metabolic dysfunction, so effects in healthy individuals may be less pronounced. Further research is needed to fully resolve the clinical relevance of TMAO elevations and to optimize L-carnitine use in diverse populations.
Track this in your stack
See how l carnitine relates to your health goals and monitor changes in your biomarkers over time.
