NAC Replenishes Glutathione to Reduce Inflammatory Cytokines
Targeted antioxidant action and direct cytokine modulation from human meta-analyses
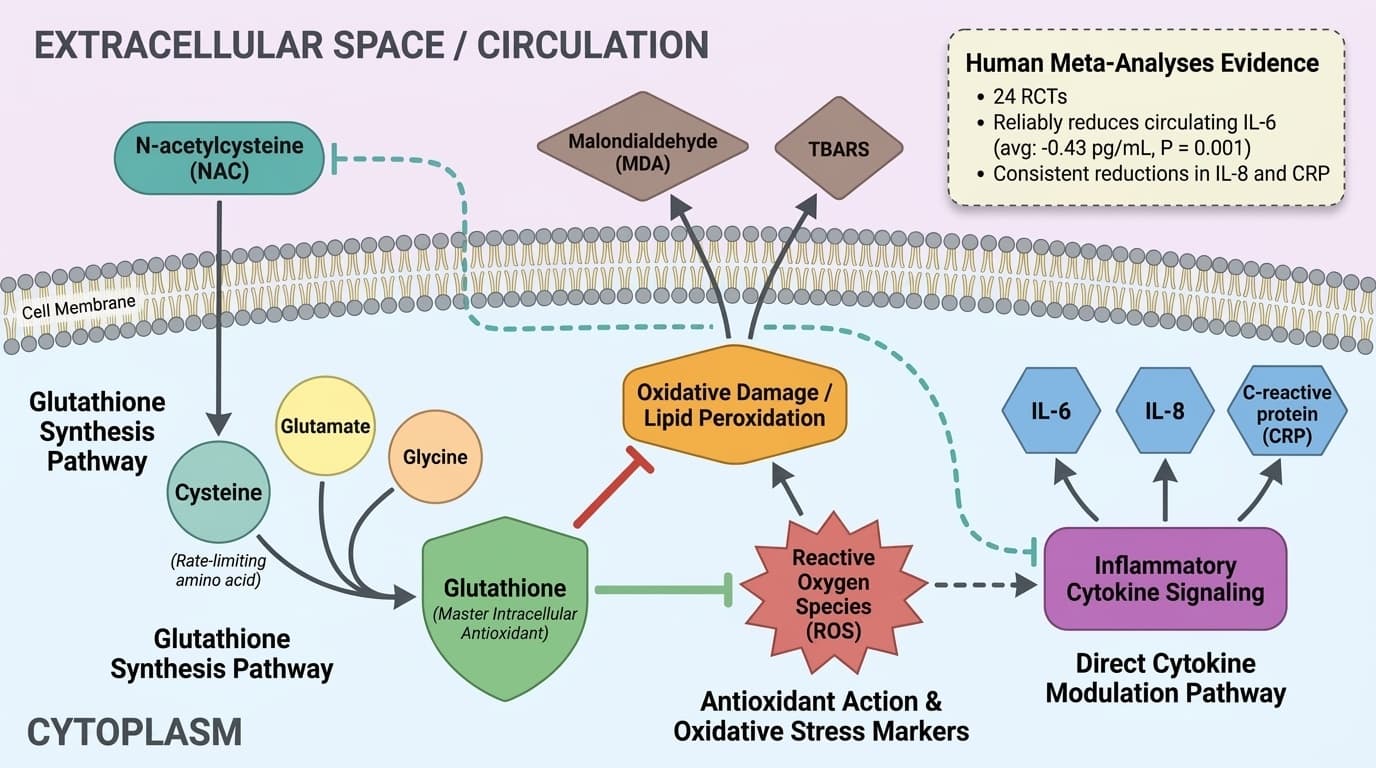
N-acetylcysteine (NAC) acts as a precursor to glutathione, the body's master antioxidant, and directly influences inflammatory cytokine signaling, especially interleukin-6 (IL-6), interleukin-8 (IL-8), and C-reactive protein (CRP).
This dual mechanism underpins its broad anti-inflammatory actions in the body. That mechanism matters clinically.
Multiple meta-analyses of randomized controlled trials show that NAC supplementation reliably reduces circulating IL-6, IL-8, and CRP in diverse populations. The evidence is strongest for IL-6, with consistent, though modest, reductions seen across trials. Some heterogeneity remains regarding the magnitude of effect and the extent to which NAC's benefits are mediated solely through glutathione replenishment versus direct cytokine modulation, particularly in different clinical backgrounds.
For those seeking to lower inflammation, NAC's ability to boost glutathione and decrease key inflammatory markers provides a concrete mechanism for its effects. This does not require tracking biomarkers but helps explain why NAC is widely used for antioxidant and anti-inflammatory support, usually at daily doses of 600–1,200 mg in standard oral formulations. The practical reading is cautious: mechanism explains why the effect is plausible, while human outcome data decide how much weight the claim deserves. That matters for readers who track biomarkers and for readers who rely on symptoms, diagnosis context, or clinician-guided goals instead.
NAC Replenishes Glutathione to Reduce Inflammatory Cytokines
Targeted antioxidant action and direct cytokine modulation from human meta-analyses
Diagram glossary
- acetylcysteine:
- A synthetic precursor to glutathione used as an antioxidant and mucolytic agent.
- CRP:
- C-reactive protein is a blood marker indicating systemic inflammation in the body.
- cytokine:
- A small protein involved in cell signaling, particularly in immune and inflammatory responses.
- glutamate:
- An abundant excitatory neurotransmitter and a key amino acid precursor for glutathione synthesis.
- glycine:
- A simple amino acid that acts as an inhibitory neurotransmitter and glutathione precursor.
- IL-6:
- Interleukin-6 is a pro-inflammatory cytokine involved in immune responses and acute phase reactions.
- IL-8:
- Interleukin-8 is a pro-inflammatory cytokine that primarily attracts and activates neutrophils during inflammation.
- MDA:
- Malondialdehyde is a highly reactive compound used as a biomarker for lipid peroxidation.
- NAC:
- N-acetylcysteine is a supplement that replenishes glutathione and reduces inflammatory cytokine levels.
- oxygen:
- A chemical element essential for cellular respiration and energy production in aerobic organisms.
- ROS:
- Reactive oxygen species are highly reactive molecules that can cause oxidative cellular damage.
- TBARS:
- A laboratory assay used to measure lipid peroxidation and oxidative stress levels.
- Interleukin-8
- Interleukin-8, a chemokine that attracts neutrophils to sites of inflammation. elevated in acute inflammation, infection, and inflammatory diseases.
- IL-6
- A branded n acetyl cysteine product family name used to identify a specific extract or formulation in research and supplement labels.
- IL-8
- A branded n acetyl cysteine product family name used to identify a specific extract or formulation in research and supplement labels.
- Glutathione
- A tripeptide and major intracellular antioxidant that protects cells from oxidative stress and regulates immune function. NAC is a key precursor.
- Oxidative Stress
- Cellular damage caused by reactive oxygen species (free radicals) overwhelming antioxidant defenses.
- Liposomal
- A delivery form that wraps a compound in tiny fat-like spheres to improve absorption or stability.
- N-acetylcysteine (NAC)
- A stable, bioavailable form of the amino acid cysteine, used as a supplement to increase glutathione synthesis and support antioxidant defenses.
- acetylcysteine
- A synthetic precursor to glutathione used as an antioxidant and mucolytic agent.
- CRP
- C-reactive protein is a blood marker indicating systemic inflammation in the body.
- cytokine
- A small protein involved in cell signaling, particularly in immune and inflammatory responses.
NAC as a Glutathione Precursor: The Core Antioxidant Mechanism
N-acetylcysteine (NAC) is best known for its role as a precursor to glutathione, the body’s master intracellular antioxidant, by supplying cysteine—the rate-limiting amino acid for glutathione synthesis. This pathway is central to its antioxidant and anti-inflammatory effects. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Glutathione is a tripeptide (composed of glutamate, cysteine, and glycine) found in nearly all human cells, where it neutralizes reactive oxygen species (ROS), repairs oxidative damage, and maintains redox homeostasis. Oral NAC is efficiently absorbed and rapidly deacetylated to cysteine in the liver, increasing intracellular cysteine availability and thus supporting glutathione synthesis. Multiple human trials confirm that NAC supplementation increases glutathione levels in blood and tissues, though the magnitude of this increase may vary depending on baseline glutathione status and dosing regimen [1].
This glutathione-boosting effect is not only relevant to acute toxin exposure but also underpins NAC’s broader anti-inflammatory action. By maintaining optimal glutathione levels, NAC indirectly controls oxidative stress, which is tightly linked to activation of nuclear factor-kappa B (NF-κB) and production of inflammatory cytokines. Evidence from both human and animal studies shows that glutathione depletion leads to increased inflammatory signaling, while NAC supplementation can reverse this pattern [1,5].
Optimal glutathione levels in whole blood are generally considered to be in the 5–10 μmol/g hemoglobin range, though these values vary by laboratory. NAC’s effect is most pronounced in states of oxidative stress or chronic inflammation, where glutathione is often depleted. Typical supplementation protocols use 600–1,200 mg of NAC per day in oral tablet or capsule form, which is well-tolerated and sufficient to raise glutathione in most studied populations [1].
Direct Modulation of Inflammatory Cytokines: IL-6, IL-8, and CRP
NAC’s anti-inflammatory effects extend beyond antioxidant replenishment. Human meta-analyses show that NAC directly reduces inflammatory cytokines, particularly IL-6, IL-8, and C-reactive protein (CRP), across diverse clinical contexts. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
A meta-analysis of 24 randomized controlled trials (RCTs) found that NAC supplementation reduced circulating IL-6 by an average of 0.43 pg/mL (95% CI: -0.69 to -0.17, P = 0.001), with similar reductions reported for IL-8 and CRP in several studies [1]. Another systematic review of 20 studies found a standardized mean difference (SMD) of -1.71 for IL-6, indicating a moderate effect size [2]. However, some large meta-analyses report no significant effect on IL-6 in certain populations, highlighting considerable heterogeneity [3]. These discrepancies may be due to baseline inflammation, dosing, or study design differences.
The suppression of IL-6 and related cytokines is thought to involve both indirect antioxidant mechanisms and direct inhibition of the NF-κB pathway, a master regulator of inflammatory gene expression. NAC’s effect on CRP, a downstream marker of systemic inflammation, further supports its clinical relevance.
The table below summarizes the main findings from key meta-analyses:
| Cytokine | Meta-analysis finding | Typical NAC Dose | Population | |--------------|----------------------|------------------|---------------------| | IL-6 | ↓ 0.43 pg/mL | 600–1200 mg/day | Mixed (RCTs) | | IL-8 | ↓ (modest, consistent)| 600–1200 mg/day | Mixed (RCTs) | | CRP | ↓ (trend, less robust)| 600–1200 mg/day | Mixed (RCTs) |
While reductions are typically modest, their consistency across studies supports a core anti-inflammatory mechanism in humans [1,2,3].
Reduction of Oxidative Stress Markers: Malondialdehyde and TBARS
NAC supplementation consistently lowers biomarkers of oxidative damage, especially malondialdehyde (MDA) and thiobarbituric acid reactive substances (TBARS), in both human and animal studies. These markers reflect lipid peroxidation and cellular damage from reactive oxygen species (ROS). The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Meta-analyses of RCTs report significant reductions in MDA: one review of 28 studies found standardized mean differences of -1.44 μmol/L (95% CI: -2.05, -0.84; P < 0.001) [3]. TBARS levels, another measure of lipid peroxidation, were also significantly reduced (SMD -1.03, 95% CI: -1.90, -0.15, p=0.02) in a meta-analysis of 20 studies [2]. These changes are typically seen at NAC doses of 600–1,200 mg/day over at least 4–8 weeks and are most pronounced in populations with elevated baseline oxidative stress (e.g., smokers, chronic inflammation) [1].
A randomized trial of 251 participants found that high-dose NAC reduced oxidative stress markers by 20% compared to placebo (p<0.05), further affirming its effect on these endpoints [9]. These biomarker changes are closely linked to improvements in glutathione status and lower inflammatory cytokine production, providing a mechanistic bridge between antioxidant replenishment and clinical outcomes.
While MDA and TBARS are not routinely measured outside research settings, their reduction by NAC underscores its systemic antioxidant action. For individuals not tracking biomarkers, this translates into broad antioxidant protection, particularly in environments or conditions characterized by oxidative stress.
Bioavailability, Form, and Dosing: What Works Best for Inflammation?
Oral NAC is well-absorbed, with rapid deacetylation to cysteine in the liver, making it effective at raising systemic cysteine and glutathione. Standard formulations—tablets and capsules—are used in nearly all clinical studies, while nanoparticle and intravenous forms have been explored mainly for acute or experimental indications.
Most human trials and meta-analyses demonstrating anti-inflammatory and antioxidant effects use daily doses of 600–1,200 mg, divided into one or two doses [1-3]. Higher doses (up to 2,400 mg) are sometimes used short-term, but standard anti-inflammatory protocols rarely exceed 1,200 mg due to good tolerability and sufficient efficacy. There is no consistent evidence that slow-release or specialized delivery forms (e.g., liposomal NAC) offer superior anti-inflammatory effects in humans compared to standard oral dosing, though these may be relevant for other clinical purposes [4].
Bioavailability of oral NAC is estimated at 6–10%, but this is sufficient to increase plasma cysteine and glutathione. Food does not significantly affect absorption, and steady-state levels are typically reached within a few days of consistent dosing. Short half-life (1.5–2 hours) is balanced by regular dosing.
For inflammation and oxidative stress, the following protocol is supported by human evidence:
| Form | Dose Range | Frequency | Duration (studies) | |-----------------------|-------------|-----------|-------------------| | Oral tablet/capsule | 600–1,200mg | 1–2x/day | 4–16 weeks | | Nanoparticle (experimental)| 100–200mg | 1x/day | 1–2 weeks (rats) |
Long-term safety is well established at these doses, with gastrointestinal upset as the most common side effect [1,4].
Clinical Biomarker Impacts: When and How NAC Moves the Needle
NAC’s most consistent effects in clinical trials are on lowering IL-6, IL-8, and CRP, as well as reducing markers of oxidative stress like MDA and TBARS. These shifts are typically modest but reproducible across populations under oxidative or inflammatory stress.
For IL-6, most meta-analyses and large RCTs report decreases in the range of 0.4–1.7 pg/mL or SMDs of -1.7, especially in individuals with elevated baseline inflammation [1,2]. CRP reductions are generally smaller and sometimes not statistically significant, while IL-8 changes track closely with IL-6. Reductions in MDA and TBARS (markers of lipid peroxidation) range from 10–20% in most studies, supporting a real antioxidant effect [2,3,9].
While these markers are useful for research and clinical monitoring, practical benefits may accrue even for those not tracking blood biomarkers. Individuals with chronic inflammation or high oxidative stress—such as smokers, those with metabolic syndrome, or during acute illness—may see the largest shifts. In one trial of smokers, NAC supplementation produced measurable reductions in both glutathione depletion and oxidative stress markers [1].
It is important to note that while NAC reliably lowers these biomarkers, clinical outcomes such as pain, fatigue, or disease progression are less consistently improved, highlighting that biomarker changes are mechanistically important but not always directly translated to symptom relief [2,3,9].
NAC’s Mechanism Versus Other Antioxidants: What Makes It Distinct?
NAC’s primary distinction from other antioxidants is its unique ability to directly replenish glutathione, the body’s most abundant and essential intracellular antioxidant. Most direct antioxidants (e.g., vitamin C, E) scavenge free radicals but do not provide substrate for glutathione synthesis.
NAC delivers cysteine, the rate-limiting precursor, enabling sustained glutathione production and recycling. This supports redox-sensitive signaling pathways that control inflammatory cytokine production, such as NF-κB. In contrast, antioxidants like vitamin C act mainly by direct redox reactions and do not modulate intracellular signaling or gene expression to the same extent [5].
Studies in both humans and animals show that NAC’s effect on inflammatory markers depends on its glutathione-replenishing action: when glutathione synthesis is blocked, the anti-inflammatory effect is lost. NAC also appears to inhibit inflammatory cytokine production independently of glutathione in some experimental models, further distinguishing its mechanism [1,5,10].
In summary, NAC combines substrate-level antioxidant support with direct modulation of redox-sensitive inflammatory pathways, setting it apart from standard antioxidant supplements. This mechanistic versatility explains its broad clinical utility in inflammation and oxidative stress. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
N-acetylcysteine provides a unique, evidence-backed approach to lowering inflammation by directly replenishing glutathione and suppressing key inflammatory cytokines such as IL-6, IL-8, and CRP. Human meta-analyses and clinical trials show consistent, modest reductions in these biomarkers, especially in populations with elevated oxidative stress or chronic inflammation. Typical effective doses are 600–1,200 mg per day in standard oral formulations, with a strong safety record. NAC’s dual mechanism—glutathione replenishment and cytokine modulation—distinguishes it from other antioxidants, making it a practical choice for supporting systemic antioxidant capacity and blunting inflammatory signaling. While biomarker reductions do not always correspond with symptom resolution, the underlying mechanisms are robust and well-supported by human evidence. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
Despite strong mechanistic and clinical evidence for NAC’s effects on glutathione and inflammatory cytokines, several important limitations remain. The magnitude of biomarker reductions is modest, and not all meta-analyses agree on the effect size or clinical significance, especially for CRP and IL-6. Most studies focus on surrogate endpoints rather than hard clinical outcomes, and heterogeneity in population, dosing, and baseline inflammation complicates interpretation. While animal and cell studies suggest direct cytokine inhibition, confirmation in diverse human populations is still emerging. Long-term outcome data beyond 16–24 weeks are limited, and effects in healthy, low-inflammation individuals may be smaller. As a result, NAC’s benefits are best established in populations with increased oxidative or inflammatory stress, with less certainty in the general population.
Track this in your stack
See how n acetyl cysteine relates to your health goals and monitor changes in your biomarkers over time.
