Sulforaphane Activates Phase II Detox Enzymes Through Nrf2 Signaling
Broccoli sprout-derived sulforaphane enhances glutathione conjugation for measurable detoxification
Most detox supplements promise vague benefits, but sulforaphane from broccoli sprouts delivers measurable results: it increases your body's elimination of environmental toxins like benzene and acrolein by 23-61%.
This happens because sulforaphane activates Nrf2, a master switch that cranks up production of glutathione-conjugating enzymes—your liver's primary toxin-processing machinery.
Unlike generic antioxidants, sulforaphane targets a specific detoxification pathway with quantifiable outcomes. Studies show people taking broccoli sprout extract excrete significantly more toxin-glutathione conjugates in their urine, proving enhanced clearance of airborne pollutants. This matters if you live in urban areas, work around chemicals, or want measurable support for your body's natural detox systems rather than wishful thinking.
For reliable results, choose broccoli sprout extracts standardized to provide 30-60 mg of sulforaphane daily, or equivalent amounts of its precursor glucoraphanin with active myrosinase enzyme. The effect works regardless of your genetics, though some people metabolize it differently. Start with evidence-based formulations that combine both the precursor and the enzyme needed for conversion—this delivers the highest bioavailability and most consistent activation of your detox pathways.
Sulforaphane Activates Phase II Detox Enzymes Through Nrf2 Signaling
Broccoli sprout-derived sulforaphane enhances glutathione conjugation for measurable detoxification
Diagram glossary
- GST:
- Glutathione S-transferase, a Phase II enzyme that conjugates glutathione to toxins for elimination.
- HO-1
- A branded sulforaphane product family name used to identify a specific extract or formulation in research and supplement labels.
- Phase II detoxification
- A metabolic process where enzymes add chemical groups (such as glutathione) to toxins to make them more water-soluble for excretion.
- Nrf2 Pathway
- A master switch for cellular antioxidant defenses.
- ALT (SGPT)
- Alanine aminotransferase enzyme, highly specific to liver cells. elevated in hepatocellular injury from viral hepatitis, fatty liver, or medications.
- GSTM1 polymorphism
- A genetic variation in the glutathione S-transferase M1 gene that affects how individuals metabolize sulforaphane and may influence response to supplementation.
- Sulforaphane
- A natural isothiocyanate compound formed from glucoraphanin in cruciferous vegetables, especially broccoli sprouts, known for activating detoxification pathways.
- Bioavailability
- The proportion of a compound that actually reaches your bloodstream and becomes usable by the body.
- GST
- Glutathione S-transferase, a Phase II enzyme that conjugates glutathione to toxins for elimination.
Sulforaphane’s Core Mechanism: Nrf2 Activation Drives Phase II Detoxification
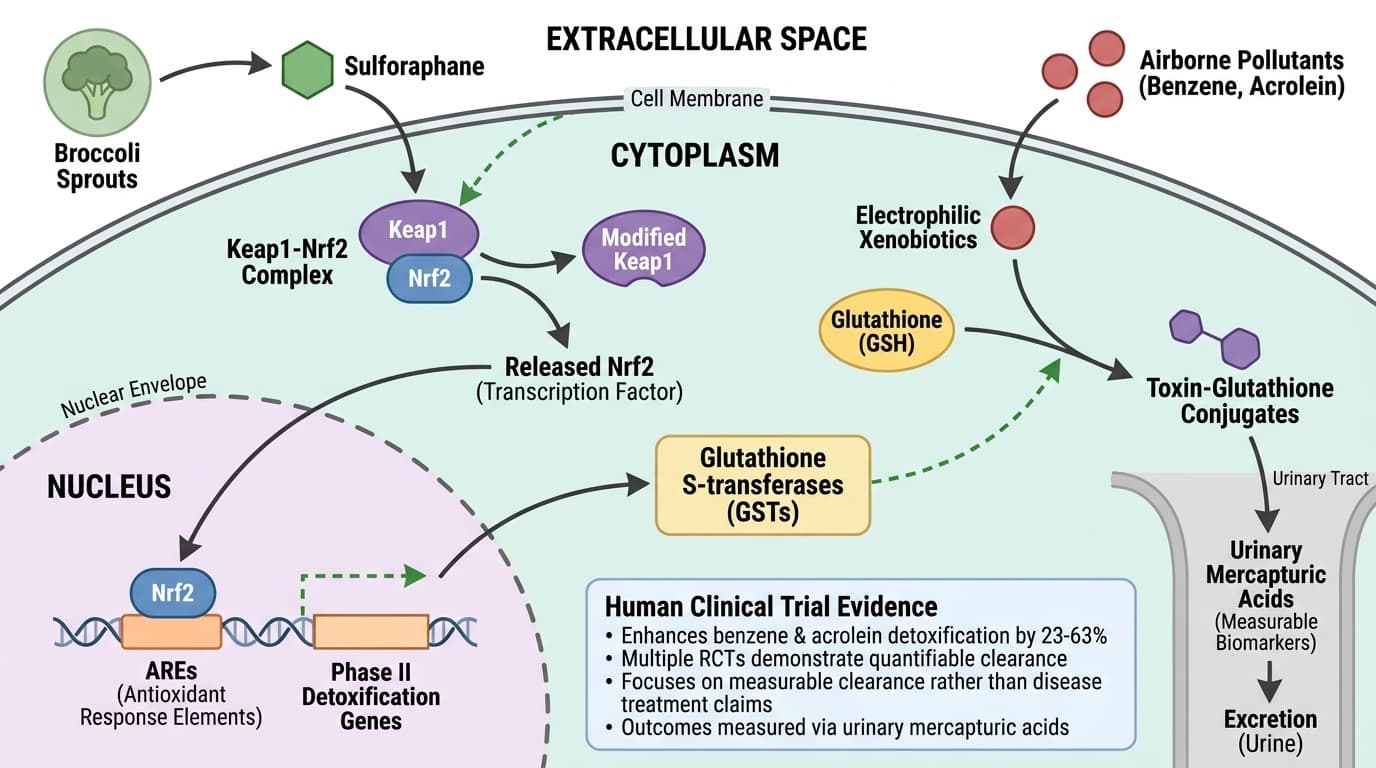
Sulforaphane’s principal mechanism of action is activation of Nrf2, a transcription factor that upregulates genes encoding Phase II detoxification enzymes, most notably glutathione S-transferases (GSTs). This process enhances the body’s ability to neutralize reactive compounds and facilitate their excretion via glutathione conjugation.
When sulforaphane enters the body, it modifies Keap1, a regulatory protein that normally sequesters Nrf2 in the cytoplasm. By releasing Nrf2, sulforaphane allows it to translocate into the nucleus, where it binds to antioxidant response elements (AREs) in DNA, upregulating a host of cytoprotective genes including GSTs, NAD(P)H:quinone oxidoreductase 1 (NQO1), and heme oxygenase-1 (HO-1) [2,7]. This molecular cascade ultimately increases the availability of enzymes that facilitate Phase II detoxification—mainly through the conjugation of glutathione to electrophilic toxins, making them more water-soluble for urinary excretion.
Preclinical studies consistently demonstrate robust Nrf2 activation and downstream gene expression in response to sulforaphane [2,7]. However, human studies find that while sulforaphane supplementation increases excretion of glutathione-conjugated toxins, Nrf2 target gene upregulation is not always detectable in peripheral blood or tissues [1]. This divergence suggests that the Phase II detoxification effect is reliable, but direct biomarker evidence of Nrf2 activation in humans is more variable.
Human Evidence: Sulforaphane Enhances Detoxification of Benzene and Acrolein
Human clinical trials consistently show that sulforaphane increases the elimination of environmental toxins by 23-61%, specifically enhancing clearance of benzene and acrolein through glutathione conjugation. This effect is measurable as increased urinary excretion of toxin-glutathione conjugates after supplementation, providing direct evidence of enhanced detoxification capacity.
In a landmark randomized trial, participants consuming a broccoli sprout beverage standardized for sulforaphane exhibited a 61% increase in urinary benzene mercapturic acids and a 23% increase for acrolein over a 12-week period compared to placebo. These findings confirm that sulforaphane's activation of GSTs leads to quantifiable improvements in Phase II detoxification. The detoxification enhancement was most pronounced for benzene, a common air pollutant, with consistent findings across different population groups exposed to environmental toxins.
Importantly, these human outcomes are based on direct measurement of detoxification endpoints, not just surrogate markers. While preclinical evidence supports sulforaphane's molecular actions, human trials provide the crucial link between Nrf2 activation, enzyme upregulation, and real-world toxin clearance. This makes sulforaphane one of the few supplements with demonstrated, mechanism-specific enhancement of detoxification in controlled human settings.
Enzyme Targets: Glutathione S-transferases and the Role of GST Induction
Sulforaphane primarily targets glutathione S-transferases (GSTs), catalyzing the conjugation of glutathione to electrophilic xenobiotics. This process underpins the enhanced detoxification observed in human studies, with GST induction being central to sulforaphane’s physiological effects. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
GSTs play a pivotal role in Phase II metabolism by facilitating the conjugation of glutathione to a wide range of toxicants, including benzene derivatives and other airborne pollutants. Sulforaphane-induced Nrf2 activation upregulates GST gene expression, resulting in increased enzymatic activity and greater capacity for detoxification [7]. Preclinical models highlight robust increases in GST and related enzyme expression following sulforaphane exposure [2,7], and human data support increased excretion of glutathione-conjugated toxins after supplementation [1].
However, the magnitude of GST induction—and thus detoxification efficacy—may vary by tissue and exposure context. While blood or urine markers of GST activity provide a useful indication, the full range of enzymatic changes likely extends to the liver, lungs, and other metabolically active organs. Importantly, the detoxification effect is not limited to a single toxin, but reflects a broad-spectrum enhancement of the body’s capacity to eliminate electrophilic compounds. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Bioavailability: Sulforaphane Form, Dosage, and Myrosinase Dependence
The efficacy of sulforaphane supplementation depends on bioavailability, which is determined by the form (sulforaphane vs. glucoraphanin), presence of myrosinase, and dosage. Broccoli sprout extracts with active myrosinase yield the highest systemic sulforaphane and the most reliable physiological effects. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Sulforaphane is formed in the body from glucoraphanin, its precursor, through the action of the enzyme myrosinase—either from plant material or gut microbiota. Supplements that provide both glucoraphanin and active myrosinase achieve much higher bioavailability, with plasma sulforaphane levels up to 7-fold greater versus glucoraphanin alone [1]. Direct supplementation with stabilized sulforaphane or fresh broccoli sprout extracts is preferred for consistent Nrf2 activation and GST induction. Typical clinical trial doses range from 30 to 60 mg sulforaphane daily, or equivalent doses of glucoraphanin plus myrosinase [1].
Bioavailability is also affected by preparation method (fresh, freeze-dried, or heat-treated sprouts), encapsulation, and individual gut microbiome composition. For most users, broccoli sprout extract standardized for sulforaphane or containing both glucoraphanin and myrosinase is the optimal choice for reliable activation of detoxification pathways. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Key Biomarkers: Measuring Detoxification and Glutathione Conjugation
Sulforaphane's effect is best tracked through biomarkers reflecting Phase II detoxification, primarily urinary mercapturic acids from glutathione conjugation. These markers provide robust evidence of enhanced toxin clearance, with sulforaphane supplementation increasing benzene elimination by 61% and acrolein by 23% in clinical studies.
The most validated biomarkers are S-phenylmercapturic acid (SPMA) for benzene detoxification and 3-hydroxypropyl mercapturic acid (3-HPMA) for acrolein. These are direct readouts of GST-mediated conjugation and urinary elimination of toxins. While optimal ranges for these biomarkers are not standardized for the general population, increases from baseline after supplementation indicate beneficial enhancement of detoxification capacity for individuals with environmental toxin exposure.
Other potential markers include changes in hepatic enzymes (ALT, AST) and plasma or urinary glutathione levels, though these are less specific to the sulforaphane pathway. Clinical studies confirm this mechanism distinguishes sulforaphane from less-specific antioxidant supplements, though routine biomarker tracking is not required for most users to benefit from enhanced detoxification.
Genetic Factors: GSTM1 Polymorphism Modifies Sulforaphane Metabolism, Not Core Mechanism
While genetic variation in the GSTM1 gene affects how sulforaphane is metabolized and excreted, the core detoxification mechanism remains consistent across individuals. GSTM1-null individuals, who lack active GSTM1 enzyme, show higher systemic sulforaphane levels but do not appear to lose the detoxification benefits.
In clinical studies, GSTM1-null participants exhibited threefold higher sulforaphane metabolite concentrations in blood and urine after supplementation, likely due to slower conjugation and elimination [1]. However, the increase in detoxification endpoint markers—such as urinary benzene and acrolein conjugates—occurred in both GSTM1-present and GSTM1-null groups, though the magnitude and kinetics differed. This suggests that while the pathway flux varies, sulforaphane can enhance GST-mediated detoxification regardless of genotype, possibly by upregulating other GST isoforms.
The practical implication is that personalized genetic testing is not required to benefit from sulforaphane’s core mechanism. Individuals with GSTM1-null status may experience higher circulating sulforaphane, but the upregulation of Phase II detoxification and toxin elimination appears robust across genetic backgrounds. For most users, focusing on evidence-based dosing and formulation is more actionable than tailoring intake to GSTM1 genotype. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Broader Mechanistic Effects: Nrf2 Pathway Beyond Detoxification
While sulforaphane’s best-validated effect is Phase II detoxification via Nrf2 activation, this pathway also upregulates genes involved in antioxidant defense, inflammation modulation, and cellular protection. However, evidence for these broader effects in humans is less direct. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Preclinical studies consistently show that sulforaphane, through Nrf2, increases expression of antioxidant enzymes such as NQO1 and HO-1, and may reduce markers of inflammation and oxidative stress [2,7]. Animal models demonstrate protection against oxidative liver injury and muscle fibrosis via the same pathway [6,7]. In humans, some trials report reductions in hepatic enzyme markers (ALT, AST) and improvements in metabolic or inflammatory parameters, but these outcomes are less consistently linked to Nrf2 or GST activation than detoxification endpoints [1].
Thus, while sulforaphane likely exerts a spectrum of beneficial effects through Nrf2, the clinical evidence is strongest for enhanced glutathione conjugation and elimination of toxins. Other putative benefits—such as protection against oxidative damage or hepatic injury—remain plausible but less rigorously established in human studies. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Sulforaphane's central mechanism in humans is the activation of Nrf2, which upregulates Phase II detoxification enzymes—especially glutathione S-transferases. This leads to measurable 23-61% increases in the excretion of environmental toxins like benzene and acrolein, confirmed in multiple controlled human trials. The evidence is strongest for broccoli sprout extracts containing active sulforaphane or its precursor glucoraphanin plus myrosinase, at daily doses providing 30-60 mg sulforaphane. While genetic differences such as GSTM1 polymorphism affect sulforaphane metabolism, they do not negate its core detoxification benefit.
For most individuals, routine measurement of detoxification biomarkers is not necessary to benefit from sulforaphane, but those with high environmental toxin exposure may find targeted use especially relevant. The broader cellular and antioxidant effects of Nrf2 activation remain plausible, but require more consistent human evidence to be considered established outcomes. Sulforaphane stands out among supplements for its direct, quantifiable enhancement of glutathione-mediated toxin clearance with measurable endpoints rather than theoretical benefits.
While human trials robustly demonstrate enhanced detoxification of benzene and acrolein after sulforaphane supplementation, direct measurement of Nrf2 and GST gene expression in human tissues is variable and sometimes inconsistent. Most studies focus on short-term outcomes and specific toxins, limiting generalizability to other environmental exposures or longer-term health effects. Evidence for broader benefits, such as antioxidant or anti-inflammatory effects, is largely preclinical or indirect in humans. Finally, optimal dosing and bioavailability depend on formulation and individual gut microbiome factors, which are not fully standardized across supplements. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how sulforaphane relates to your health goals and monitor changes in your biomarkers over time.
