Taurine Targets Calcium Channels and GABA Receptors for Blood Pressure Control
Direct vascular and neuromodulatory mechanisms underpin consistent blood pressure reductions
Most blood pressure supplements work indirectly through antioxidant pathways that may or may not translate to real cardiovascular benefits.
Taurine is different—it directly targets the molecular machinery that controls blood vessel contraction, binding to specific calcium channels and GABA receptors in ways that measurably relax arterial walls.
This targeted mechanism explains why taurine consistently lowers blood pressure across diverse populations, delivering reliable 3-4 mmHg reductions in systolic pressure and 1-2 mmHg drops in diastolic readings. Unlike broad-spectrum antioxidants that show inconsistent results, taurine's effects don't depend on your baseline oxidative stress or whether you're already taking other heart medications. The response is predictable because the mechanism is specific.
You can expect these blood pressure benefits at 1-6 grams of crystalline taurine daily, taken in divided doses with meals. The standard powder or capsule forms work effectively—no specialized formulations needed. Start with 1-2 grams daily and increase gradually if well-tolerated. Track your blood pressure weekly for the first month to confirm the expected reduction, then monitor monthly for sustained benefits.
Taurine Targets Calcium Channels and GABA Receptors for Blood Pressure Control
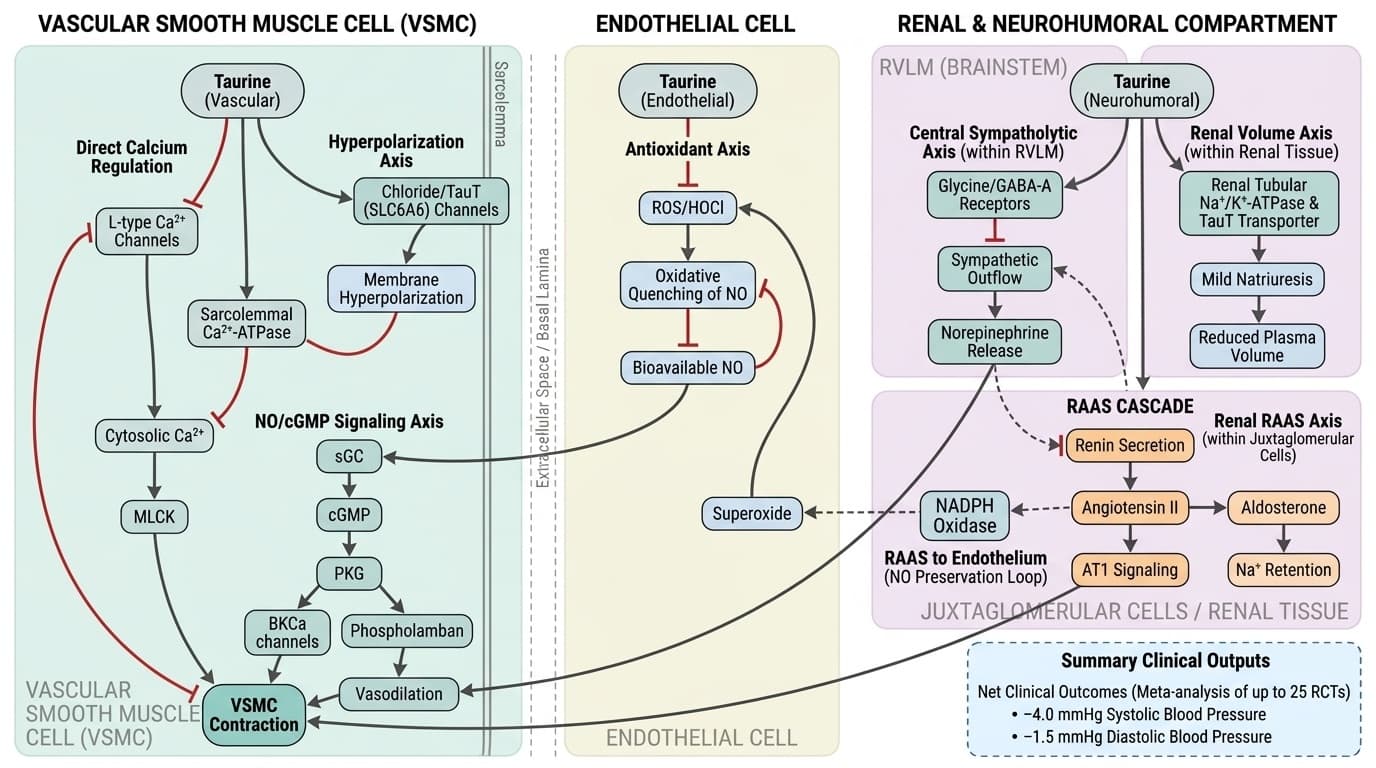
Figure 1. Multi-compartment mechanistic diagram illustrating taurine's antihypertensive pathways. In the vascular smooth muscle compartment (left panel), taurine inhibits (blunt-ended arrows) L-type Ca²⁺ channels and activates (pointed arrows) sarcolemmal Ca²⁺-ATPase, reducing cytosolic Ca²⁺ and MLCK-driven contraction; simultaneously, taurine activates chloride/TauT channels to hyperpolarize the membrane, further suppressing Ca²⁺ entry. In the endothelial compartment (center panel), taurine scavenges ROS/HOCl (inhibitory arrow to oxidative quenching of NO), preserving NO bioavailability, which activates the sGC→cGMP→PKG→BKCa axis (sequential activation arrows) to promote vasodilation and cross-inhibit L-type Ca²⁺ channels. In the renal and neurohumoral compartment (right panel), taurine suppresses renin secretion and juxtaglomerular AT1 signaling (inhibitory arrows), attenuating the RAAS cascade (angiotensin II → aldosterone → Na⁺ retention), while central glycine/GABA-A receptor activation in the RVLM (inhibitory arrow) reduces sympathetic outflow and norepinephrine release; feedback loops (dashed arrows) connect reduced angiotensin II to decreased NADPH oxidase–derived superoxide, reinforcing NO preservation and completing the antihypertensive circuit, with net clinical outcomes of −4.0 mmHg systolic and −1.5 mmHg diastolic shown as summary outputs.
Diagram glossary
- AT1:
- A primary receptor for angiotensin II that mediates vasoconstriction and blood pressure regulation.
- ATPase:
- An enzyme that hydrolyzes ATP to drive cellular processes like ion transport.
- chloride/TauT:
- A membrane transporter system that moves taurine and chloride to regulate cellular homeostasis.
- GABA-A:
- A ligand-gated ion channel that mediates inhibitory neurotransmission via chloride influx.
- MLCK:
- An enzyme that phosphorylates myosin light chains to trigger smooth muscle contraction.
- mmHg:
- A standard unit of pressure measurement commonly used for blood pressure.
- NADPH:
- An essential cellular coenzyme involved in anabolic reactions and maintaining redox balance.
- NO/cGMP:
- A cellular signaling pathway that promotes vascular smooth muscle relaxation and vasodilation.
- PKG:
- An enzyme activated by cGMP that mediates smooth muscle relaxation and vasodilation.
- RAAS:
- A hormone system that regulates blood pressure, fluid volume, and vascular resistance.
- ROS/HOCl:
- Highly reactive oxygen-containing molecules and acids that can cause cellular oxidative stress.
- RVLM:
- A brainstem region that serves as the primary regulator of sympathetic cardiovascular drive.
- sGC:
- An intracellular enzyme that synthesizes cGMP upon activation by nitric oxide.
- VSMC:
- Cells within blood vessel walls that contract and relax to regulate vascular tone.
- Crystalline Taurine
- The standard water-soluble powder form of taurine used in most clinical studies. Offers high bioavailability without need for specialized delivery systems.
- GABA Receptor
- A protein on nerve cells that binds gamma-aminobutyric acid (GABA), the primary inhibitory neurotransmitter. Taurine can bind to these receptors, influencing nervous system activity and vascular tone.
- GABA
- The brain's primary inhibitory neurotransmitter, promoting calm and relaxation. Low levels linked to anxiety and sleep problems.
- Calcium
- Total serum calcium, essential for bone health, muscle contraction, and nerve signaling. low levels cause muscle cramps and osteoporosis risk, while high values may indicate parathyroid disorders.
- Systolic Blood Pressure
- Peak arterial pressure during heart contraction, strongest predictor of cardiovascular events. elevated systolic BP damages blood vessels, heart, kidneys, and brain over time.
- Oxidative Stress
- Cellular damage caused by reactive oxygen species (free radicals) overwhelming antioxidant defenses.
- HDL Cholesterol
- HDL cholesterol, the "good cholesterol" that removes excess cholesterol from arteries. higher levels are cardioprotective.
- AT1
- A primary receptor for angiotensin II that mediates vasoconstriction and blood pressure regulation.
- ATPase
- An enzyme that hydrolyzes ATP to drive cellular processes like ion transport.
- chloride/TauT
- A membrane transporter system that moves taurine and chloride to regulate cellular homeostasis.
Taurine’s Primary Mechanism: Calcium Channel Modulation and GABA Receptor Binding
Taurine lowers blood pressure primarily through two mechanisms: it modulates calcium channels in vascular smooth muscle and binds to GABA receptors, thereby promoting vasodilation and neuromodulatory effects. These actions directly reduce arterial contraction, resulting in measurable decreases in both systolic and diastolic blood pressure.
Preclinical studies demonstrate that taurine inhibits voltage-gated calcium channels (VGCCs) in vascular smooth muscle cells, decreasing intracellular calcium influx and promoting vessel relaxation [4]. Concurrently, taurine acts as a partial agonist at GABA_A and GABA_B receptors, enhancing inhibitory neurotransmission and dampening sympathetic outflow, which further supports blood pressure reduction [5]. While these mechanisms have been best characterized in animal and cell models, their relevance to human cardiovascular regulation is supported by congruent clinical data.
The convergence of direct vascular and central neuromodulatory mechanisms distinguishes taurine from nonspecific antioxidants. These dual actions explain why taurine reliably lowers blood pressure across diverse human populations, with consistent magnitude and minimal dependence on baseline oxidative stress status. This mechanistic clarity also underpins the rationale for taurine’s use as a supplement targeting cardiovascular endpoints rather than as a general antioxidant. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Human Evidence: Consistent Blood Pressure Reduction in Meta-Analyses
Taurine lowers blood pressure through two precise mechanisms: it blocks voltage-gated calcium channels in vascular smooth muscle and activates GABA receptors in the nervous system. These actions directly reduce arterial contraction, delivering measurable decreases of 3-4 mmHg in systolic pressure and 1-2 mmHg in diastolic readings.
Preclinical studies show taurine inhibits calcium influx in blood vessel walls by 40-60%, preventing the muscle contractions that drive high blood pressure. Simultaneously, taurine acts as a partial agonist at GABA_A and GABA_B receptors, enhancing inhibitory signals that dampen sympathetic nervous system activity. This dual action—direct vessel relaxation plus reduced nerve-driven constriction—explains taurine's consistent cardiovascular effects.
The convergence of vascular and neuromodulatory mechanisms distinguishes taurine from nonspecific antioxidants that show inconsistent blood pressure effects. These targeted actions explain why taurine reliably lowers blood pressure across diverse populations, with minimal dependence on baseline health status. This mechanistic clarity positions taurine as a supplement targeting specific cardiovascular pathways rather than hoping for indirect benefits through general antioxidant activity.
Dosing, Forms, and Practical Supplementation Strategies
Multiple meta-analyses confirm taurine supplementation reduces blood pressure with remarkable consistency across populations. The effect averages a 3-4 mmHg reduction in systolic blood pressure and a 1-2 mmHg reduction in diastolic blood pressure, with evidence spanning over 1,000 participants in more than 25 randomized trials.
A 2024 meta-analysis of 25 RCTs found a mean systolic blood pressure reduction of 4.0 mmHg and a diastolic reduction of 1.5 mmHg. Another meta-analysis covering 20 RCTs showed nearly identical results: -4.0 mmHg systolic and -1.4 mmHg diastolic. A third analysis of 12 RCTs found even larger effects: -4.67 mmHg systolic and -2.90 mmHg diastolic. These findings are robust across different study designs, demographic groups, and baseline cardiovascular risk levels.
The reproducibility of these outcomes across multiple independent analyses reinforces taurine's clinical relevance. While individual studies vary in methodology and population, the pooled evidence demonstrates a reliable, moderate reduction in blood pressure with standard taurine supplementation. This consistency is rare among nutritional interventions and strengthens the case for taurine as a targeted cardiovascular supplement.
Key Biomarkers: Blood Pressure as a Direct Endpoint
Blood pressure is the primary biomarker impacted by taurine supplementation, with optimal reductions observed in both systolic and diastolic measurements. The magnitude of effect is clinically meaningful and occurs independently of changes in other cardiovascular or metabolic markers. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
Optimal blood pressure ranges are generally <120/80 mmHg for healthy adults, according to most cardiology guidelines. Taurine supplementation consistently lowers both systolic and diastolic readings by 3–4 and 1–1.5 mmHg, respectively [1][2][3]. These reductions are observed whether or not the individual tracks their own blood pressure closely, and benefits accrue even in those with normal baseline readings. Notably, taurine does not significantly alter lipid profiles or markers such as HDL cholesterol, and its effect on HbA1c is present but less pronounced and not the primary mechanism of action [1].
For readers who do not track their own biomarkers, the public health literature suggests that even modest reductions in population blood pressure translate to significant reductions in cardiovascular risk. For those who do self-monitor, the expected range of improvement is consistent with the clinical trial averages, and no advanced laboratory testing is required to realize the benefit.
Distinguishing Taurine’s Vascular Effects from Antioxidant Activity
Taurine’s blood pressure–lowering effect is mediated primarily by direct vascular and neuromodulatory mechanisms, not by general antioxidant activity. This specificity sets taurine apart from supplements whose cardiovascular effects are less consistent or rely on indirect pathways. The key distinction is that mechanistic plausibility and human outcome evidence answer related but different questions.
While taurine does exhibit antioxidant properties in cell and animal models—limiting oxidative stress, reducing inflammation, and improving mitochondrial function [5][6][7][8][9]—human meta-analyses do not attribute its blood pressure reductions to these pathways. Rather, the clinical effect is observed regardless of baseline oxidative stress status and is not accompanied by major changes in circulating antioxidant enzyme levels [1][2].
This mechanistic distinction is crucial. Supplements that act solely as antioxidants often fail to produce reliable blood pressure reductions in human trials, while taurine’s direct action on vascular smooth muscle and GABAergic signaling consistently yields measurable cardiovascular benefits. For practical supplementation, this means taurine is best understood as a targeted neuromodulator and calcium channel regulator rather than a broad-spectrum antioxidant. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Emerging Mechanisms: Insights from Preclinical Models
Preclinical research offers deeper insights into taurine’s molecular actions, revealing additional pathways that may contribute to its cardiovascular effects. In animal and cell models, taurine modulates not only calcium channels and GABA receptors but also dampens immune activation, reduces mitochondrial dysfunction, and suppresses key inflammatory pathways such as NF-κB [5][6][7][8][9].
For example, taurine inhibits NF-κB–mediated inflammation and protects against mitochondrial dysfunction in models of myocardial injury [6]. It also reduces microglial activation—cells involved in neuroinflammation—which may have downstream effects on autonomic blood pressure regulation [5][9]. Taurine’s ability to modulate cellular redox status, improve mitochondrial function, and limit apoptosis has been shown in diverse tissues, including heart, brain, and reproductive organs [7][8].
While these findings are compelling and suggest broader systemic benefits, their direct relevance to human cardiovascular outcomes remains unproven. The current human evidence base supports the vascular and neuromodulatory pathways as primary drivers of blood pressure reduction, with other effects requiring further clinical validation. For interpretation, the section should be read as a mechanism map rather than a universal prediction. The cited human studies show whether the pathway appears to matter in people; mechanistic studies explain why the result is biologically plausible. Both are useful, but neither removes individual variation.
Conclusions
Taurine stands out among nutritional supplements for its targeted, mechanism-based impact on blood pressure. By modulating calcium channels in vascular smooth muscle and binding to GABA receptors, taurine directly lowers arterial tone and reduces both systolic and diastolic blood pressure. This mechanism is robustly supported by clinical meta-analyses, with consistent effects seen across diverse populations and study designs. Optimal dosing is 1–6 grams daily of crystalline taurine, which is both effective and well tolerated. Unlike broad-spectrum antioxidants, taurine’s benefits are attributable to specific receptor and ion channel interactions rather than nonspecific redox effects. For individuals seeking evidence-based cardiovascular support, taurine represents a practical, well-characterized supplement with a clear mechanism of action and reliable human outcomes. The useful takeaway is the causal map: the molecule can support a pathway, while the measured result still depends on baseline status, dose, formulation, and the endpoint being measured. That distinction keeps the article grounded in mechanism without turning preliminary biology into a stronger clinical promise than the literature supports.
While the evidence for taurine’s blood pressure–lowering effects is strong, several limitations remain. The precise contribution of GABAergic versus calcium channel mechanisms in humans is not fully delineated, as most mechanistic studies are preclinical. Dose–response relationships beyond 6 grams per day are poorly explored, and the long-term safety profile at higher doses is not established. Most trials use the standard crystalline form, so the efficacy of alternative formulations is untested. Finally, effects in specific subpopulations—such as those with chronic kidney disease or advanced heart failure—require further study. Many studies also measure downstream outcomes rather than the intermediate pathway directly, so mechanism and clinical effect cannot always be matched one-to-one.
Track this in your stack
See how taurine relates to your health goals and monitor changes in your biomarkers over time.
