The Insulin Resistance Spectrum: Why 'Normal' Glucose Can Hide a Decade of Metabolic Risk
Uncovering the Hidden Years Before Blood Sugar Rises
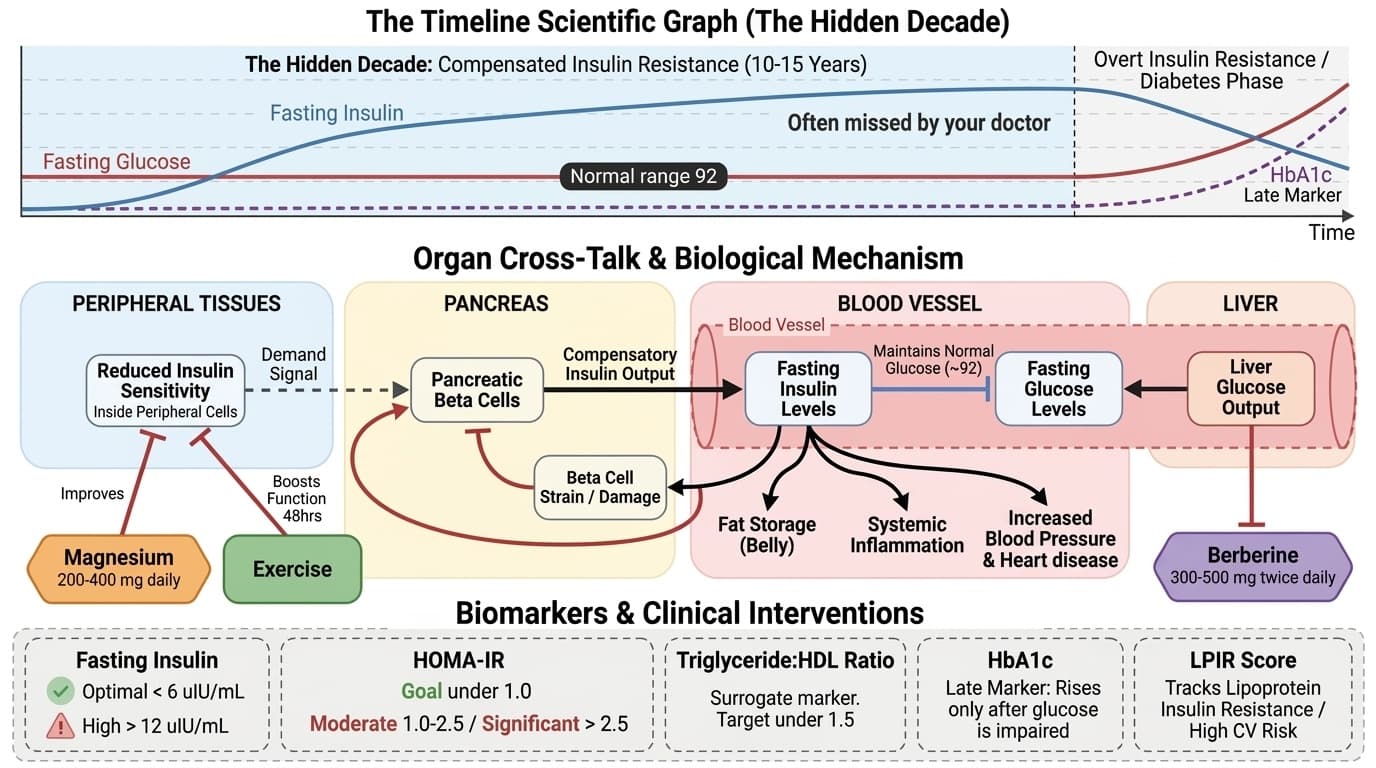
You might be surprised to learn that your "normal" fasting glucose of 92 could be hiding a decade of metabolic damage.
Most doctors only check blood sugar, not insulin levels. This means you could have insulin resistance for 10-15 years before anyone notices a problem. Your pancreas works overtime pumping out extra insulin to keep glucose normal. But this extra effort eventually fails.
This matters because you're missing early warning signs. High insulin levels predict diabetes, heart disease, and metabolic syndrome years before blood sugar rises. You can catch these problems early with the right tests. Ask for fasting insulin (keep it under 6), HOMA-IR (goal under 1.0), and triglyceride-to-HDL ratio (target under 1.5).
Take action now with targeted supplements. Berberine 300-500 mg twice daily reduces liver glucose output. Magnesium 200-400 mg daily improves insulin sensitivity. Exercise boosts insulin function for up to 48 hours after each session. The earlier you look beyond glucose, the better you can protect your health.
The Insulin Resistance Spectrum: Why 'Normal' Glucose Can Hide a Decade of Metabolic Risk
Uncovering the Hidden Years Before Blood Sugar Rises
Diagram glossary
- Berberine:
- A plant-derived supplement used to reduce liver glucose output and improve metabolic health.
- Glucose:
- A simple sugar that serves as the primary energy source for the body's cells.
- HDL:
- High-density lipoprotein is a beneficial blood cholesterol that helps remove other forms of cholesterol.
- HOMA:
- A mathematical model used to quantify insulin resistance and beta-cell function from fasting blood.
- Insulin:
- A hormone produced by the pancreas that regulates glucose levels in the blood.
- LPIR:
- A clinical laboratory score that measures insulin resistance using lipoprotein particle sizes and concentrations.
- Triglyceride:
- A type of fat found in the blood that stores unused calories for energy.
- Metabolic Syndrome
- A group of risk factors—including high blood pressure, high blood sugar, and abnormal cholesterol—that raise your risk of heart disease and diabetes.
- Magnesium Glycinate / Magnesium Citrate
- These are specific formulations of magnesium supplements where the mineral is bound to glycinate (an amino acid) or citrate (an organic acid) to improve absorption and reduce digestive side effects.
- Glucose
- Blood sugar level, the primary energy source for cells. Fasting glucose is normal, prediabetes, ≥126 suggests diabetes.
- Berberine
- A natural plant-derived compound found in herbs like barberry and goldenseal that has been studied for its ability to improve insulin sensitivity and lower blood sugar by activating a key metabolic enzyme called AMPK.
- HOMA-IR (calc)
- Insulin resistance by combining fasting glucose and insulin levels.
- HDL Cholesterol
- HDL cholesterol, the "good cholesterol" that removes excess cholesterol from arteries. higher levels are cardioprotective.
- Triglyceride-to-HDL Ratio
- A simple calculation dividing your triglyceride level by your HDL cholesterol level, used as an indirect marker of insulin resistance; a ratio above 1.5 suggests poor metabolic health and possible ear
- HDL
- High-density lipoprotein is a beneficial blood cholesterol that helps remove other forms of cholesterol.
- HOMA
- A mathematical model used to quantify insulin resistance and beta-cell function from fasting blood.
- Insulin
- A hormone produced by the pancreas that regulates glucose levels in the blood.
- LPIR
- A clinical laboratory score that measures insulin resistance using lipoprotein particle sizes and concentrations.
- Triglyceride
- A type of fat found in the blood that stores unused calories for energy.
The Opening: Normal Glucose, Hidden Risk
Picture this: you go for your annual physical. Your fasting glucose comes back at 92, right in the so-called normal range. Your doctor glances at the number and tells you everything looks great. But what if another test—fasting insulin—had also been ordered? Imagine that result comes back at 14, more than double the optimal range.
Most doctors don’t routinely check fasting insulin, so this early sign goes unnoticed. But your pancreas is already working harder than ever, pumping out extra insulin to keep your blood sugar normal. This is not just a hypothetical. This is what happens to millions of people during the early stages of insulin resistance. Your fasting glucose number may look reassuring, but underneath, your body is already shifting into a higher gear to compensate for cells that are not responding well to insulin.
This matters because missing these early warning signs means you lose out on years of prevention. By the time glucose rises, insulin resistance is well established and much harder to reverse. This sets the stage for metabolic syndrome, type 2 diabetes, and increased cardiovascular risk. Understanding what’s happening beneath the surface is crucial for protecting your long-term health. Next, let’s look at how the hidden decade of compensated insulin resistance unfolds.
The Hidden Decade: Compensated Insulin Resistance
For most people, insulin resistance develops quietly, sometimes for more than ten years, before blood sugar starts to rise. During this phase, called compensated insulin resistance, your pancreas responds to reduced insulin sensitivity by making more and more insulin. This keeps your blood glucose in a normal range, but at a cost.
Research going back to the 1970s, like Kraft's work, showed that people can have double or triple the normal insulin output during an oral glucose tolerance test—even when their glucose is still considered normal [2]. Tabak and colleagues later confirmed that insulin levels start to rise about 13 years before fasting glucose moves out of the typical range [1]. In practical terms, this means your body is working harder behind the scenes for years before any doctor would see a problem on your standard metabolic panel.
Why does this matter? Because elevated insulin itself causes problems. High insulin promotes fat storage, especially around the belly. It increases inflammation and raises blood pressure. It also gradually damages the insulin-producing cells in your pancreas. The longer your insulin stays high, the more strain you put on these beta cells. Over time, they start to lose their ability to keep up. Early intervention here can make a huge difference. But you can't intervene if you don't know there's a problem. That's why looking at fasting insulin, not just glucose, is so important.
The Spectrum in Numbers: Biomarkers and Their Meaning
To truly understand where you are on the insulin resistance spectrum, you need to look at more than just fasting glucose. There are several key biomarkers that track different stages of the process:
1. Fasting Insulin: Optimal is under 6 uIU/mL. Levels between 6 and 12 are concerning. Anything above 12 suggests overt insulin resistance. Insulin can rise for years before glucose does, reflecting your pancreas’s extra effort to keep things in balance.
2. HOMA-IR: This score combines fasting glucose and fasting insulin. Under 1.0 is optimal; 1.0–2.5 suggests moderate resistance, and above 2.5 means significant insulin resistance. HOMA-IR is especially useful for tracking change over time. If you make diet or lifestyle changes, you can usually see a shift in 3–6 months.
3. Triglyceride:HDL Ratio: This is a surrogate marker for insulin resistance, with a goal of less than 1.5. High triglycerides and low HDL are hallmark features of the metabolic syndrome. This ratio moves with diet, exercise, and targeted supplements, often improving within weeks to months.
4. HbA1c: This is a late marker, reflecting average glucose over 2–3 months. It only rises after fasting glucose is already impaired, meaning you miss the early stages of the spectrum if you rely on it alone.
5. LPIR Score: Not always measured, but tracks lipoprotein insulin resistance. High scores here correlate with cardiovascular risk, even before diabetes develops [1].
Each of these markers catches a different phase of the insulin resistance spectrum. For demographic context, younger adults and athletes tend to have lower baseline insulin and HOMA-IR, but even in these groups, family history or higher body fat can push numbers up. For postmenopausal women, insulin resistance often accelerates, so regular monitoring is essential. Next, let’s dig into what’s actually happening inside your cells to drive these changes.
What's Actually Happening in Your Cells: Mechanisms of Insulin Resistance
Insulin resistance is not just a number—it’s a process happening in your muscle, liver, and fat cells. In muscles, when insulin receptors become less responsive, glucose uptake drops. That means you have a harder time clearing sugar from your blood after meals. This is especially important for athletes or anyone with higher muscle mass, since muscle is a major site of glucose disposal.
In the liver, insulin normally tells the organ to stop making glucose. When liver cells become resistant, they keep pumping out sugar, even when insulin levels are high. This drives up fasting glucose over time. It also raises triglyceride production, which is why high triglycerides are a tip-off that your liver is struggling with insulin signaling.
Adipose, or fat tissue, plays a third role. Insulin-resistant fat cells start to release more free fatty acids into the bloodstream. These fatty acids make insulin resistance worse in both muscle and liver, creating a vicious cycle. This is why losing even a small amount of fat can have such a dramatic impact on improving insulin sensitivity.
All of these changes interact—when one tissue becomes resistant, it puts more demand on the others. Over time, the strain on your pancreas grows. For some groups, like people with a family history of diabetes or those who gain weight around the belly, these cycles can accelerate. Understanding these mechanisms shows why early action matters, and sets the stage for what happens when your pancreas can’t keep up anymore.
The Decompensation Cliff: When Beta Cells Can’t Keep Up
There comes a tipping point when your pancreas just can't keep increasing insulin production. This stage is called beta-cell decompensation. When this happens, your fasting glucose starts to climb from the 90s into the 100s, and your HbA1c rises above 5.7%. Finally, the problem shows up on routine blood work—but by then, the underlying process has been going on for years.
Research shows that by the time glucose is impaired, beta cell function may have already declined by 50 to 80 percent [1]. This loss is often not recoverable. That's why relying on fasting glucose alone is so risky—it's a late marker that can't warn you about the approaching cliff.
The transition from compensated resistance to decompensation often accelerates. In most people, the compensated phase lasts a decade or more. But certain risk factors speed things up: genetics, aging, sedentary lifestyle, chronic stress, and weight gain around the midsection. Early warning signs during compensation can include fatigue after meals, stubborn belly fat, or trouble losing weight despite healthy habits—especially in older adults and those with a family history.
For prevention, the key is to monitor insulin and HOMA-IR before glucose rises. Catching the shift early gives you the best chance to preserve beta cell function and reverse the process before permanent damage occurs.
What You Can Do Right Now: Testing, Supplements, and Interventions
If you want to spot problems early, ask your healthcare provider to check your fasting insulin. Aim for a result under 6 uIU/mL. Next, use your fasting glucose and insulin to calculate HOMA-IR. This metric helps you track your metabolic health over time and can guide your prevention plan.
Supplements play a role at different points on the spectrum. Berberine, for example, primarily reduces hepatic glucose output. Studies suggest a dose of 300–500 mg twice daily can improve fasting insulin and glucose, especially in people with elevated HOMA-IR [1]. Magnesium works at the cellular level, improving insulin sensitivity at the receptor. For most adults, 200–400 mg of elemental magnesium is effective. The form matters—magnesium glycinate and citrate are well absorbed and less likely to cause digestive upset.
Exercise is the most powerful intervention of all. Just one session of moderate to intense activity can boost insulin sensitivity for up to 48 hours. This is true regardless of age, but older adults and those with higher baseline insulin resistance see especially large benefits. Stacking these interventions—supplements plus exercise—creates synergy, addressing insulin resistance in both the liver and muscle.
If you’ve read earlier lessons on berberine and HbA1c, you now see why those targets matter. Berberine reduces liver glucose output, while HbA1c tracks the late stage of the process. Combining these markers and interventions lets you address insulin resistance at every step. Next, let’s look at why two people with the same fasting glucose can be in very different metabolic states.
The Bottom Line: Two People, Two Realities
Here’s the take-home message: two people can have a fasting glucose of 95, but their metabolic health could not be more different. One person may have a fasting insulin of 5, signaling excellent insulin sensitivity and little risk. The other could have a fasting insulin of 18, meaning a decade of compensated insulin resistance is already underway.
This spectrum is invisible if you only look at glucose. You must measure insulin to see the full picture. Research linking insulin resistance to cardiovascular disease, even in people with normal glucose, makes this even more important [1]. Early detection means timely intervention—preserving your beta cells, preventing metabolic syndrome, and reducing your long-term risk.
No matter your age or fitness level, understanding and tracking these markers empowers you to take charge of your metabolic health. The earlier you act, the more options you have to stay healthy and prevent future problems.
Conclusions
Insulin resistance is a slow-moving process that can develop for over a decade before your blood sugar rises. Fasting glucose alone misses the early stages, when your pancreas is working overtime and your risk for metabolic disease is quietly increasing. By tracking key markers like fasting insulin (under 6), HOMA-IR (under 1.0), and triglyceride:HDL ratio (under 1.5), you can uncover problems early and take targeted action. Supplements like berberine (300-500 mg twice daily) and magnesium (200-400 mg elemental daily), combined with regular exercise, offer multiple ways to improve insulin sensitivity at different points in the spectrum. The most important step is to move beyond glucose and get a complete picture of your metabolic health before permanent damage occurs.
Most studies referenced were observational or used surrogate markers rather than direct clinical outcomes. Research on the long-term effects of supplements like berberine and magnesium is still emerging, and optimal dosing may differ by individual. Demographic-specific data—especially for women, older adults, and diverse populations—remains limited in some areas. Not all clinical labs offer fasting insulin or LPIR scores, which may affect access to early detection for some readers. Finally, lifestyle interventions work best when tailored to personal risk factors and under medical supervision.
Track this in your stack
See how insulin resistance spectrum relates to your health goals and monitor changes in your biomarkers over time.
